Crispr And Cas Gene Market is estimated to be valued at USD 2.85 Bn in 2026 and is expected to reach USD 12.15 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 23.0% from 2026 to 2033.
The CRISPR and Cas gene market is witnessing growth, driven by rising demand for precision medicine, gene therapy, and other emerging biotechnology applications. The rising incidence of genetic diseases, advancements in genome sequencing technologies, and increasing research on regenerative medicine are major drivers of market growth.
For example, in 2024, Vertex Pharmaceuticals and CRISPR Therapeutics gained U.S. FDA approval for Casgevy, the first CRISPR/Cas9-gene therapy for sickle cell disease and transfusion-dependent beta thalassemia. This approval was a watershed moment in the therapeutic application of gene editing and heralded the arrival of CRISPR treatments to market.
|
Current Events |
Description and its impact |
|
Casgevy Approval for Sickle Cell Disease and Beta Thalassemia
|
|
|
Patent Dispute Between UC Berkeley and Broad Institute
|
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial Intelligence (AI) is serving as a revolutionary force in propelling the CRISPR and Cas gene-editing industry forward with increased precision, efficiency, and scalability of genetic research and therapeutic development. AI technologies—specifically machine learning and deep learning—are being utilized to sift through enormous genomic datasets, determine gene targets, and forecast the results of gene edits with unprecedented precision. These abilities are important in reducing off-target effects, one major challenge in CRISPR technology, thus enhancing the efficacy and safety of gene-editing treatments.
One of the most important advances of AI lies in guide RNAs (gRNAs) design and optimization that is crucial for targeting Cas enzymes to precise DNA sequences. AI programs are able to screen thousands of possible gRNAs quickly to determine those that have the maximum on-target activity and minimum chance of causing off-target mutations. This speeds up the drug development pipeline and improves the consistency of CRISPR interventions.
AI helps drive CRISPR research through real-time imaging analysis, automated monitoring of cellular behaviour, and modelling of intricate gene interactions. Applications such as these cut experiment time and cost, allowing researchers to investigate more genetic targets efficiently.
Additionally, AI platforms are facilitating the personalization of gene therapies by incorporating personal genetic profiles, disease phenotypes, and environmental influences to develop customized treatment regimens. With further advancements in AI, its intersection with CRISPR technologies is likely to open new doors in genomics, making gene editing more accessible, accurate, and relevant across a wider range of diseases.
Increasing product launches by the market players are expected to aid in the market growth over the forecast period. For instance, on June 21, 2021, QIAGEN, a provider of sample and assay technologies for molecular diagnostics, applied testing, academic, and pharmaceutical research, announced the launch of QIAprep& CRISPR Kit and CRISPR Q-Primer Solutions that will help researchers to analyse edited gene material.
In September 2024, QIAGEN expanded its QI Acuity digital PCR platform by adding over 100 new validated assays for applications in cancer research, inherited genetic disorders, infectious disease surveillance, and other areas.
Increasing research and developmental activities by the market players is expected to propel the market growth over the forecast period. For instance, on April 27, 2023, a team of University of Florida researchers developed a promising new CRISPR-powered method for non-invasive blood tests that could help clinicians diagnose cancer at earlier stages. On December 2, 2019, Cyrus Biotechnology, Inc., a biotech company headquartered in Seattle, U.S. and the institute of MIT and Harvard launched multi-target collaboration to develop optimized Crispr gene editing technology.
The market players are focusing on launching technologically advanced products, thus driving the market growth over the forecast period. In May 2024, Profluent, a U.S.-based AI-first protein design company, released the sequence of OpenCRISPR-1, a novel Cas9 gene editor developed using artificial intelligence. This AI-driven approach utilized a large language model trained on a database of 5.1 million Cas9-like proteins to design a gene editor with improved off-target editing capabilities.
By product type, the vector-based cas is expected to hold a dominant position in the global crispr and cas gene market during the forecast period, and this is attributed to the increasing development of innovative products.
To learn more about this report, Request Free Sample
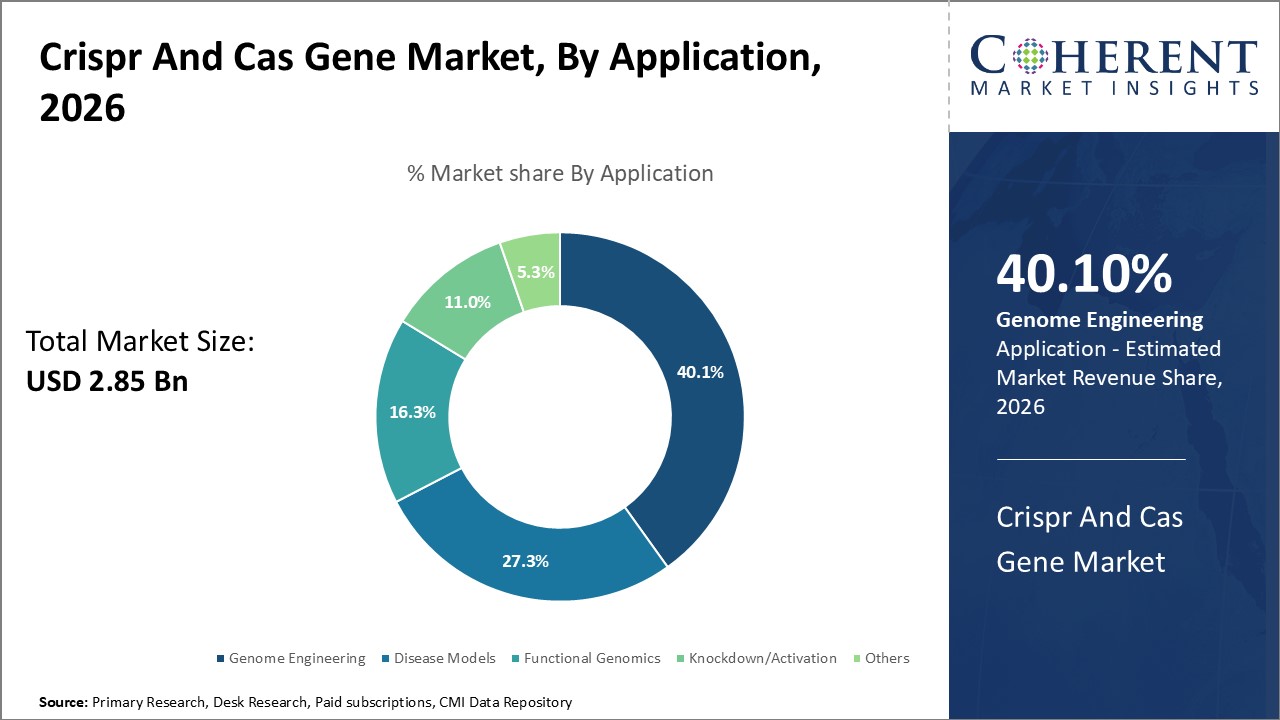
By application, the genome engineering is expected to dominate the market over the forecast period, and this is attributed to increasing product launches.
By end user, the biotechnology and pharmaceutical companies is expected to dominate the market over the forecast period, and this is attributed to the increasing research and development activities by biotechnology companies.
Key players are focusing on inorganics strategies, such as collaborations, for the development of products is expected to drive the market growth over the forecast period. For instance, on August 18, 2023, CrisprBits, a technology company based in India, announced a strategic collaboration with Molbio Diagnostics, a molecular diagnostic company. This collaboration intends to focus on point of care (POC) diagnostics by the introduction of crispr in POC tests.
To learn more about this report, Request Free Sample
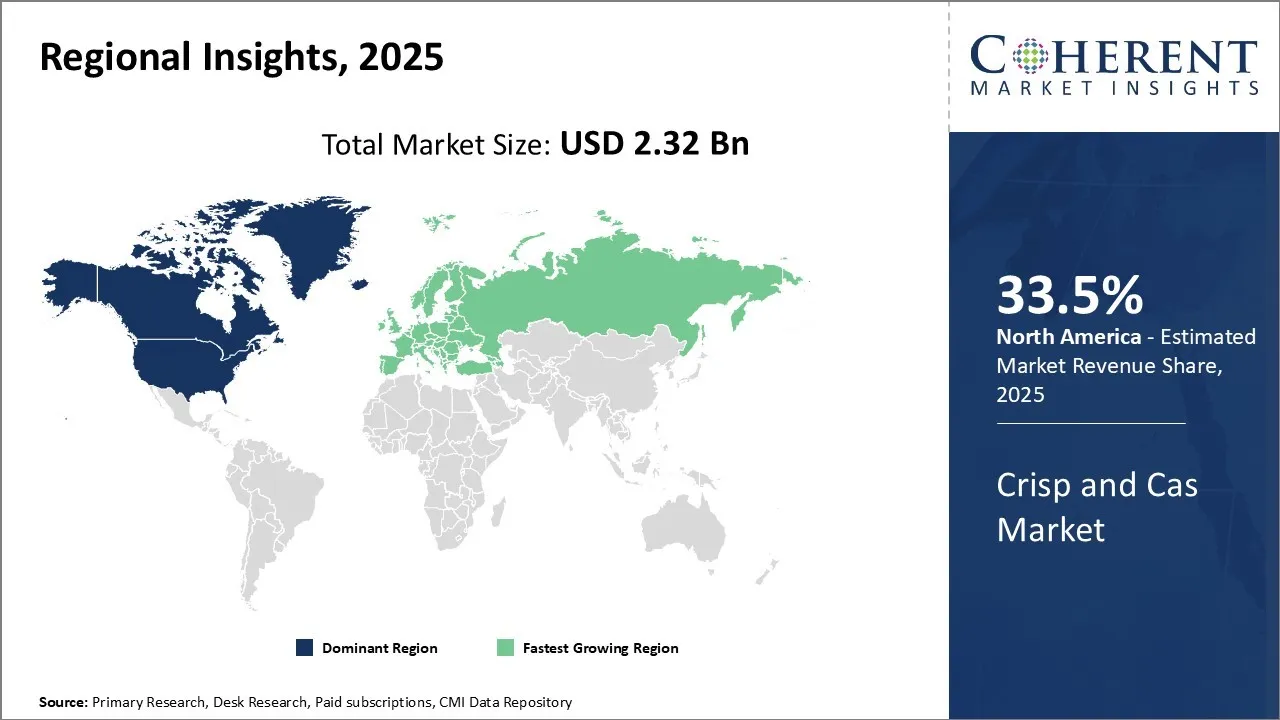
Among regions, North America is estimated to hold a dominant position in the global crispr and cas gene market over the forecast period. North America is estimated to hold 33.5 % of the market share in 2026. Global crispr and cas gene market is expected to witness significant growth in the near future, driven by increasing research and development in the field by the market players.
The United States dominates the CRISPR and Cas gene space because of its strong biotechnology landscape, research institutions, and favorable regulatory environment. With the FDA's approval of Casgevy in 2024, the United States became the first nation to bring a CRISPR-based gene therapy to market. Major players like CRISPR Therapeutics, Editas Medicine, and Intellia Therapeutics have their headquarters here, advancing innovation through cutting-edge clinical trials and collaborations.
China dominates the CRISPR market, with significant investment in gene-editing research and agri-biotech. Chinese researchers have been among the first to adopt CRISPR for research on human embryos, and the nation has rapidly scaled up the application of gene editing to plant improvement and animal breeding. The support of the government and more lenient regulatory environments allow quicker adoption of CRISPR technologies than in most Western countries.
Germany is one of the leaders of CRISPR research in Europe and has a robust pharmaceutical industry and life science sector. The Max Planck Institute and Bayer are some of the players developing CRISPR technologies for gene therapy and precision medicine. High-quality inputs to the international gene-editing scene from Germany are promoted due to the nation's dedication to ethical research and clinical testing.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.85 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 23.0%2033 Value Projection: | USD 12.15 Bn | |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., Merck KGaA (Sigma-Aldrich), OriGene Technologies, Inc., New England Biolabs, Cellecta, Inc., Agilent Technologies, Inc., Applied StemCell, Inc., Synthego, Genscript, Mirus Bio LLC, Integrated DNA Technologies, Inc., and Mammoth Biosciences, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The major players operating in the global crispr and cas gene market includes Thermo Fisher Scientific Inc., Merck KGaA (Sigma-Aldrich), OriGene Technologies, Inc., New England Biolabs, Cellecta, Inc., Agilent Technologies, Inc., Applied StemCell, Inc., Synthego, Genscript, Mirus Bio LLC, Integrated DNA Technologies, Inc., and Mammoth Biosciences, Inc.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients