The dextrose injection market is anticipated to grow at a CAGR of 5.7% with USD 6.14 Bn in 2026 and is expected to reach USD 9.05 Bn in 2033. Increasing incidence of diabetes (900 million) and conditions associated with diabetes such as hypoglycemia, increasing launch and approval of products, and rising government initiatives for the promotion of sports among individuals suffering from disabilities are expected to drive the market growth over the forecast period.
To learn more about this report, Request Free Sample
|
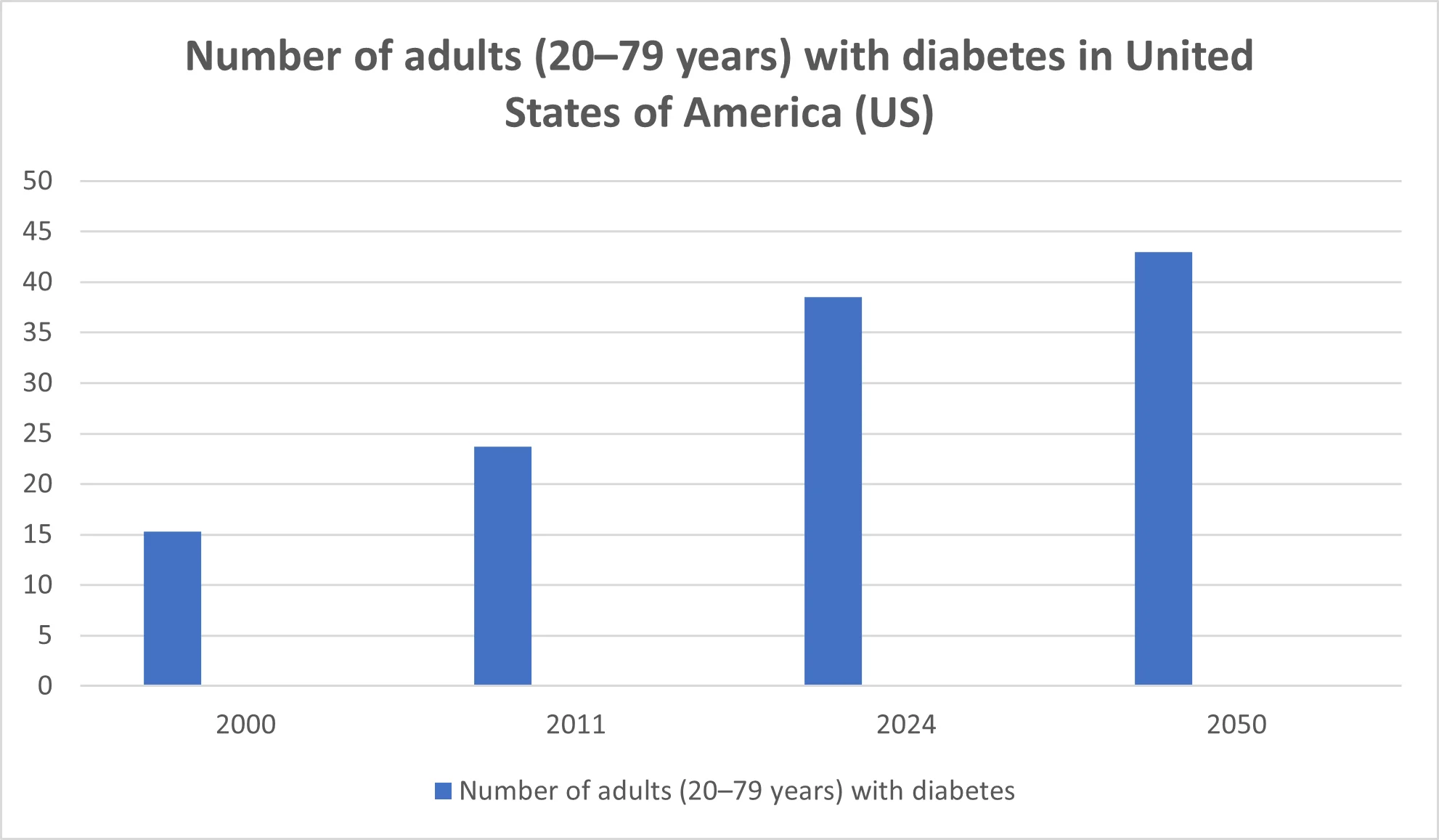
Year |
People Affected (Millions) |
|
2000 |
15.3 million |
|
2011 |
23.7 million |
|
2024 |
38.5 million |
|
2050 |
43.0 million |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Smart infusion pumps are becoming highly integrated with dextrose injection solutions. These pumps come equipped with programmable controls as well as safety features such as automated dosage limits and alarms, which mitigate the risks associated with improper administration. The combination of smart pumps as well as ready-to-use formulations ensures better compliance with treatment protocols, reduces the workload on healthcare professionals, and fosters improved therapeutic outcomes.
In December 2025, Baxter International Inc., an innovative leader in infusion therapies and technologies, released data in collaboration with The University of Texas Medical Branch (UTMB Health) demonstrating the positive impact of smart infusion pump integration with hospital electronic medical records (EMRs) on patient safety, clinicians' bedside productivity as well as programming compliance. The analyses of more than one million infusions on Baxter’s Spectrum IQ large volume infusion pumps (LVP) at UTMB were presented at the American Society of Health-System Pharmacists (ASHP) 2025.
Prefilled Syringes and Infusion Systems, Prefilled syringes containing precise doses of dextrose reduce medication errors and preparation time. Advanced infusion pumps enable controlled delivery rates to patients with major needs. Pre-filled syringes cut down substantially on steps from preparation to administration, with a 40% reduction in the overall process. According to the Institute of Packaging Professionals (IOPP), pre-filled syringes represented 6.5% of global pharmaceutical products.
Becton Dickinson (BD) has released prefilled syringe technologies as well as smart infusion pumps connecting with hospital IT systems for tracking doses.
|
Current Event |
Description and its Impact |
|
U.S. Hospital IV Shortages Amid Surge in Cardiac (2025) |
|
|
India’s Drug Price Control Expansion Including IV Solutions (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
50% Dextrose Injection is projected to account for the largest share in 2026, representing approximately 50% of the total volume. due to its extensive applications in clinical and hospital settings, fueled by its versatility and potency. The prevalence of hypoglycemia was determined as 58.44%. Nearly 11.5% of the patients encounter severe hypoglycemia. Which needs to be treated by 50% Dextrose Injection due to its severity.
The World Health Organization (WHO) has long recommended 50% Dextrose Injection as a rapid-response treatment in emergency protocols for diabetic patients experiencing hypoglycemic crises, which substantiates its widespread clinical adoption. Due to the concentrated nature of this injection, it efficiently provides immediate glucose supplementation to critically ill patients, making it indispensable in trauma centers and intensive care units.
Based on application, First-aid treatment dominates the market, accounting for a significant 40% share in 2026. The broad as well as established use of dextrose injections in hospital emergency rooms, ambulances, and urgent care centers worldwide largely drives the prevalence of this segment. According to guidelines from the American Diabetes Association, intravenous dextrose is recommended as the immediate treatment of choice for severe hypoglycemia, a common emergency in diabetic patients. This clinical backing reinforces widespread utilization by healthcare providers, ensuring a steady demand for dextrose injections as a reliable life-saving intervention.
Furthermore, beyond hypoglycemia treatment, dextrose injections are utilized in fluid replacement therapies to manage dehydration, electrolyte imbalances, as well as nutritional support in critical care settings, elevating their importance in first-aid scenarios. In 2025, a total of 1057 electrolyte abnormalities were recorded, with hypocalcemia (263 episodes, 25%) being the most common, followed by hypokalemia (192 episodes, 18%) as well as hyponatremia (172 episodes, 16%).
To learn more about this report, Request Free Sample
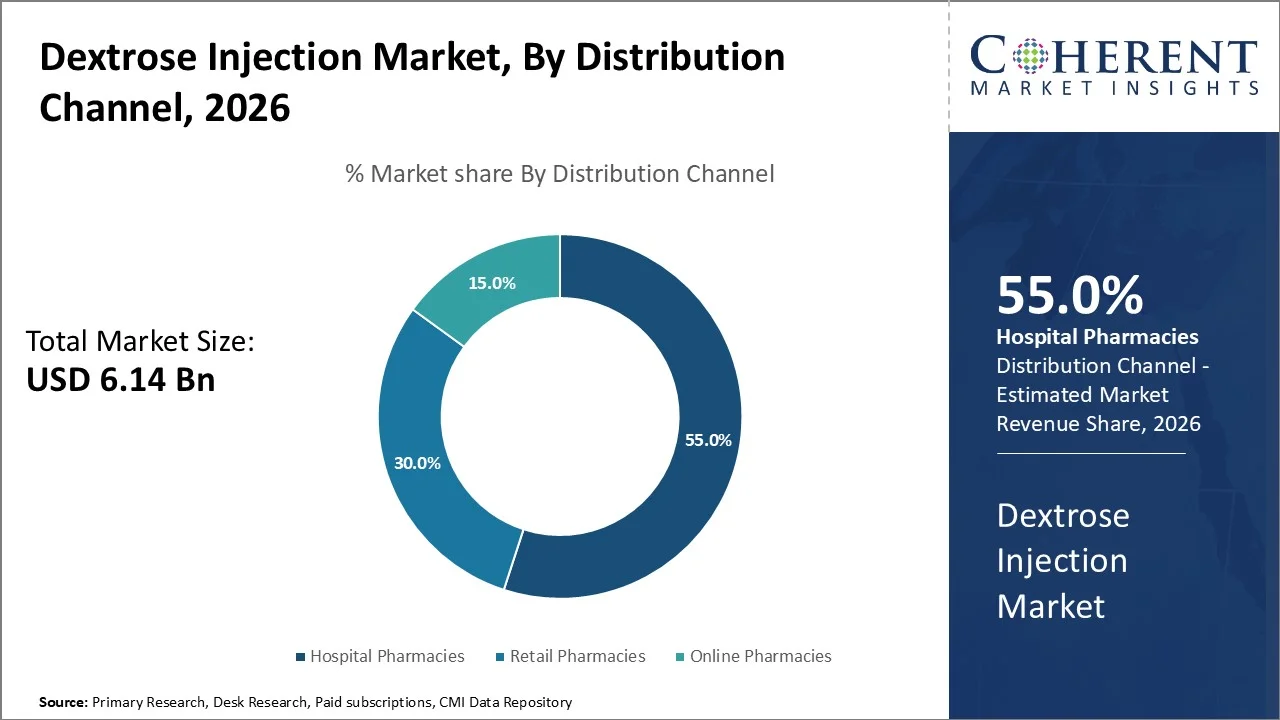
Hospital pharmacies account for the largest share of 55% in 2026, this dominance can be attributed primarily due to the critical role that hospitals play in acute and chronic patient care, where dextrose injections are usually prescribed as part of intravenous therapy for patients requiring instant replenishment of glucose levels. According to the U.S. CDC 2025, hospitals account for 55% of all intravenous glucose administrations in inpatient settings, reflecting the high demand for dextrose injections in acute care.
Hospital pharmacies are connected deeply within healthcare infrastructure, ensuring timely availability as well as stringent quality control of injectable medicines, which further reinforces their predominant market share. hospital pharmacies usually operate under regulated guidelines mandated by national health authorities such as the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA), making sure adherence to safety standards that build trust in these channels.
To learn more about this report, Request Free Sample
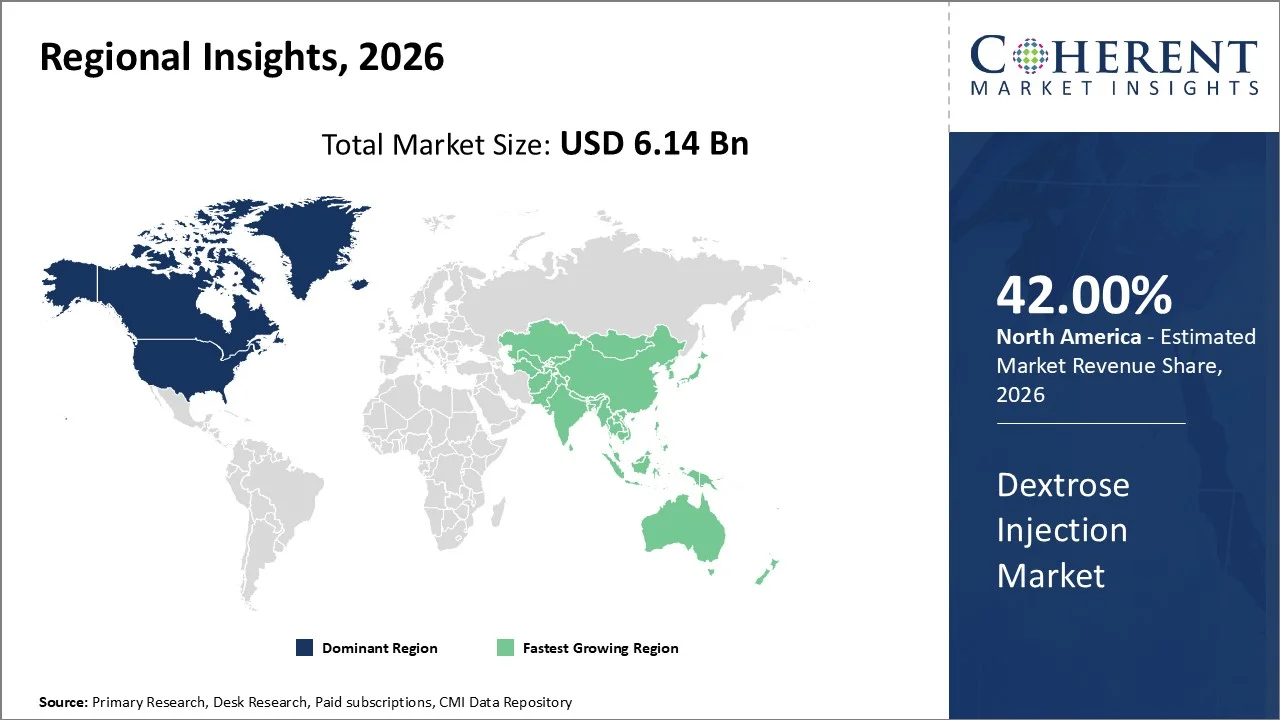
North America account 42% market share in 2026, the region's dominance in the dextrose injection market stems from several critical factors including the presence of world-class healthcare institutions, extensive hospital networks, and a growing medicine spending. Over the next five years, medicine spending will grow between 5–8% on a list price basis and 3–6% after discounts and rebates.
The American Hospital Association reported that there are over 6,090 hospitals in the United States alone, creating an enormous demand base for essential medical supplies including dextrose injections. Furthermore, the region benefits from sophisticated supply chain management systems as well as regulatory oversight by the Food and Drug Administration (FDA), which maintains rigorous quality standards for injectable pharmaceuticals.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.74%. Growth is driven by more hospitals being built, higher government spending on healthcare, and more patients needing nutrition through injections. ADB committed USD 4.26 billion in sovereign lending to India in 2025 for projects including healthcare, with an overall regional growth forecast of 5.1% to support development.
The accelerated growth is particularly evident in advancing economies such as India, Vietnam, and Indonesia, where government initiatives to improve healthcare accessibility have resulted in substantial increases in hospital capacity and medical facility upgrades. India's National Health Mission has led to the establishment of numerous new healthcare centers as well as the evolving of existing facilities, creating unprecedented demand for essential medical supplies including dextrose injections.
The United States healthcare system's advanced emergency medical services and critical care facilities create significant demand for dextrose injections across multiple therapeutic applications including hypoglycemia treatment, parenteral nutrition support, and perioperative care. The American Hospital Association reports that the U.S. operates over 6,090 hospitals with more than 919,000 staffed beds, showing a vast network requiring consistent dextrose injection supplies for emergency departments, intensive care units, as well as surgical suites.
The Centers for Disease Control and Prevention indicates that approximately 37.3 million people in United States have diabetes, creating a substantial patient population at risk for hypoglycemic episodes requiring immediate dextrose intervention. Furthermore, strict rules from the U.S. Food and Drug Administration (FDA) ensure high-quality production, making U.S. pharmaceutical companies’ major suppliers of dextrose injections in the U.S. and to Canada.
China contributes the highest share in the region owing to its robust healthcare infrastructure development, massive patient population, as well as comprehensive pharmaceutical manufacturing capabilities. The country's healthcare system has undergone major modernization over the past decade, with substantial government investments in hospital construction and medical equipment procurement. China has more than 35,000 hospitals as of 2023, says the National Health Commission, which creates a big need for medical supplies like dextrose injections.
China has over 280 million people aged 60 and older, and many need more medical care. Dextrose injections are often used for these treatments. Furthermore, China's position as a global pharmaceutical manufacturing hub significantly contributes to its market leadership.
Some of the major key players in dextrose injection are Pfizer Inc., Amphastar Pharmaceuticals Inc., Shangai Haixin group Co. Ltd., Seqirus GmbH, Baxter International Inc., Shandong Qidu Pharmaceutical, Sanctus drugs and pharmaceuticals Pvt., Hospira Inc., B Braun Medical Ltd., Aurobindo Pharmaceuticals Ltd., and SteriMAx Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.14 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.7% | 2033 Value Projection: | USD 9.05 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Amphastar Pharmaceuticals Inc., Shangai Haixin group Co. Ltd., Seqirus GmbH, Baxter International Inc., Shandong Qidu Pharmaceutical, Sanctus drugs and pharmaceuticals Pvt., Hospira Inc., B Braun Medical Ltd., Aurobindo Pharmaceuticals Ltd., and SteriMAx Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients