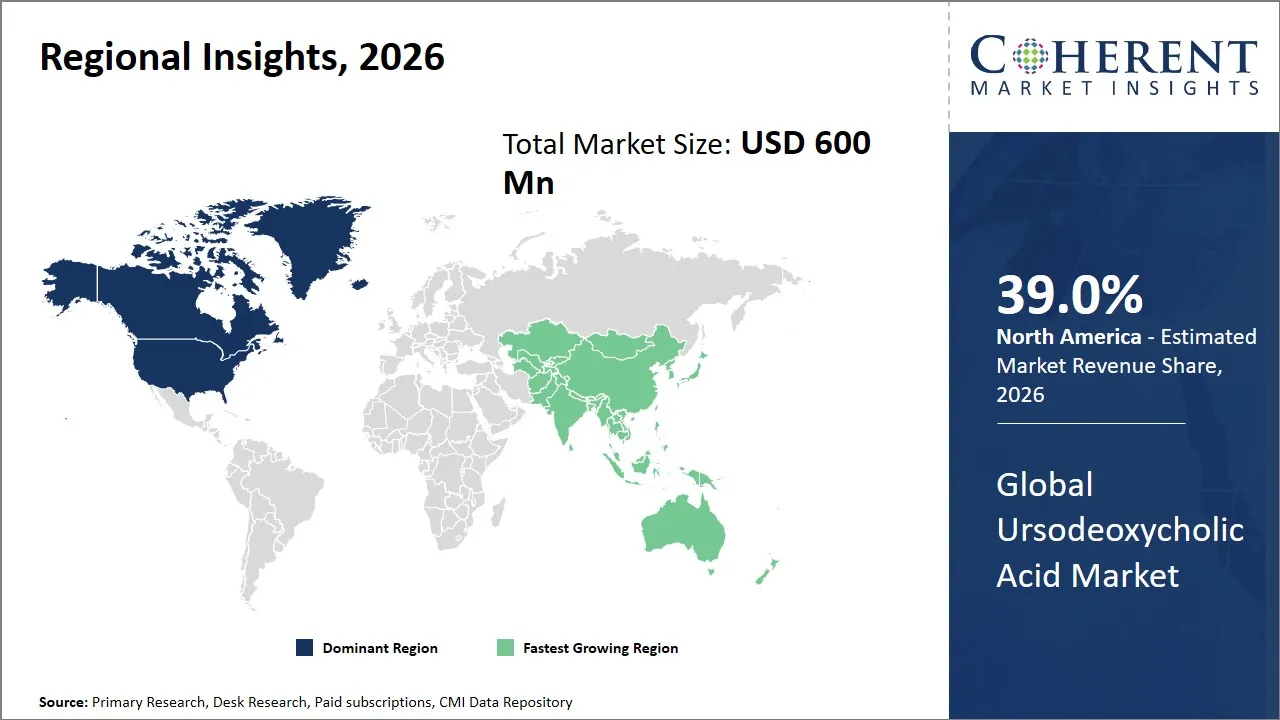
The global ursodeoxycholic acid market is estimated to be valued at USD 600 Mn in 2026 and is expected to reach USD 1,050 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033. This steady growth is a result of the rising adoption of ursodeoxycholic acid for the treatment of various liver-related diseases, advancements in the drug formulations and the rising awareness among patients across the globe. The growth of the market is also fueled by the rising incidence of gallstone disease and primary biliary cholangitis.
|
Current Events |
Description and its Impact |
|
Patent Expiry & Generic Competition |
|
|
Growing Clinical Research on UDCA Applications |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The generics segment is anticipated to hold a market share of 68.4% in 2026. This can be attributed to the increased accessibility of low-cost generics, which is a result of the loss of patent protection for branded UDCA products. Ursodeoxycholic acid can be used to treat chronic diseases. These include primary biliary cholangitis, the breakdown of gallstones, and various cholestatic liver diseases.
Patients may require lifelong therapy to manage their health. Therefore, the affordability of the drug plays a critical role in deciding the type of medication to be used. Generic UDCA products have shown similar efficacy and safety to their branded counterparts at a fraction of the price, thus encouraging healthcare professionals to use them to reduce the financial burden of the disease.
In addition, the increasing number of generic pharmaceutical manufacturers in regions like Asia Pacific, Europe, and Latin America will also boost the supply of Ursodeoxycholic Acid products. These manufacturers will benefit from their existing manufacturing capabilities and competitive strategies to meet the requirements of price-sensitive markets. Moreover, regulatory authorities in different countries have also shown their support to the use of generics by providing streamlined approval and substitution processes.
For Instance, in October 2025, research presented at the American College of Gastroenterology (ACG) Annual Scientific Meeting showed that using fibrates with ursodeoxycholic acid (UDCA) therapy improved biochemical results and reduced healthcare costs for primary biliary cholangitis. This finding further supported the clinical use of treatments based on UDCA, including the generic versions that are available.
The tablets segment is expected to hold 54% share in 2026. Tablets are the most common way to give UDCA. This is because they are easy to take, allow for precise dosing, and are generally well-accepted by patients. As a traditional dosage form, tablets are a well-established type of oral medication. This has made them the preferred choice for chronic disease treatments like Ursodeoxycholic Acid. The solid form of a medication allows for accurate dosing, helps it stay stable, and extends its shelf life. This, in turn, boosts patient assurance that the drug will retain its potency for an extended period.
From the point of view of patient compliance, tablets are easy to consume, convenient, and have low chances of spillage compared to oral suspensions, which are not only messy but also difficult to transport, especially over different geographic regions, including remote areas. Many medical practitioners prefer tablets of UDCA due to its high compatibility with established prescribing.
Another reason that makes tablets a preferred option is that they are cost-effective in terms of manufacturing, which in turn makes them cheaper. In addition, tablets can be manufactured in different strengths and sizes, thus providing personalized therapy that can meet the specific requirements of different people. Unlike capsules, tablets can also be scored to provide flexibility in adjusting the dosage.
The 150 mg segment is expected to dominate the ursodeoxycholic acid market, holding a estimated market share of 42% in 2026. The 150 mg dosage of Ursodeoxycholic Acid (UDCA) has been commonly used, as it fits well into the general clinical guidelines that have been established for the treatment of liver-related diseases, including primary biliary cholangitis and gallstones. This particular dosage has been commonly used as a baseline in the majority of clinical guidelines, which allows clinicians to easily adjust the dosages as required. This particular dosage of UDCA has been considered flexible, as it is of moderate strength, making it easy to use in personalized clinical practices.
The 150 mg dosage of UDCA has been commonly used by clinicians, as it has been considered to have an optimal balance between safety and efficacy, which makes it easy to manage patients with UDCA, especially over an extended period of time. This particular dosage of UDCA has been commonly used, as it has been available in tablet formulations that have been commonly used in clinical practices. This particular dosage of UDCA has been considered to be cost-effective, as it can be easily adjusted without resulting in the wastage of UDCA.
Global manufacturing capabilities for Ursodeoxycholic Acid (UDCA) APIs are dominated by Asia Pacific, especially China and India. Their competitive edge stems from both lower production costs and the strength of their respective pharmaceutical industries. As a result, these two countries are the main suppliers of UDCA APIs to pharmaceutical companies worldwide.
On the other hand, Europe and North America have been focusing more on the pharmaceutical processing, formulation, and compliant manufacturing of pharmaceuticals. Pharmaceutical companies in Europe are renowned for their high-end technology in pharmaceutical production and the implementation of stringent quality standards, thus catering to the market. The increasing global market for UDCA in the treatment of various liver diseases, including primary biliary cholangitis and gallstones, is encouraging manufacturers to enhance their manufacturing capacity and global UDCA APIs supply chain.
|
Region |
Manufacturing Characteristics |
Key Countries |
|
Asia Pacific |
Large-scale API production, cost-efficient manufacturing, strong export capacity |
China, India, Japan |
|
Europe |
Advanced pharmaceutical manufacturing and strict regulatory compliance |
Italy, Germany |
|
North America |
Focus on drug formulation, distribution, and regulatory-approved manufacturing |
U.S., Canada |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
The North America region is projected to lead the market with a 39% share in 2026, due to a strong pharmaceutical industry, a well-developed healthcare system, and a supportive regulatory environment. The market for UDCA-based products has seen significant growth, owing to the high involvement of major pharmaceutical players like Mallinckrodt Pharmaceuticals and Actavis, now a Teva company. These companies have improved both the availability and the formulation of the product.
Consequently, governmental initiatives, in conjunction with the FDA's regulatory framework, have contributed to the preservation of rigorous quality and safety benchmarks, thereby bolstering consumer trust. This encompassed the provision of designations and incentives to pharmaceutical entities, with the objective of stimulating the creation and introduction of novel therapeutic interventions. These interventions were specifically targeted towards conditions where ursodeoxycholic acid (UDCA) remains the primary therapeutic modality for Primary Biliary Cholangitis.
Moreover, the number of people with liver-related diseases, along with the general understanding of how effective UDCA is in treating these conditions, such as primary biliary cholangitis, has increased. This has positively affected the ursodeoxycholic acid market.
The Asia Pacific region is expected to exhibit the fastest growth of 9.3% in the ursodeoxycholic acid market share in 2026, propelled by the surge is fueled by rising healthcare expenditures, a growing patient population, and more frequent diagnoses of liver ailments. Nations like China, India, and Japan are witnessing increased government investment in healthcare.
Furthermore, regulatory changes are streamlining drug approval processes, which should further accelerate the growth of the UDCA market in the region. The presence of multinational as well as domestic players such as Lupin Limited, Zhejiang Huahai Pharmaceutical, and Takeda Pharmaceutical Company Limited is increasing product offerings, making UDCA available at affordable rates.
For instance, in May 2025, Zhejiang Huahai Pharmaceutical announced plans to increase the production capacity for its active pharmaceutical ingredients, such as bile acid derivatives, to meet global and regional pharmaceutical markets, indicating an increase in production capabilities for UDCA availability in the Asia Pacific region. In addition, favorable trade agreements, increasing distribution networks, and healthcare awareness among the population are fueling UDCA product adoption in emerging markets in the Asia Pacific region.
(Source: Zhejiang Huahai Pharmaceutical)
The U.S. benefits economically from both advanced research and a strong system of regulations. This ensures the safety and efficacy of UDCA products. Mallinckrodt Pharmaceuticals and Teva Pharmaceuticals are key players, actively shaping the landscape by pushing forward drug formulations and expanding the uses of UDCA. The favorable environment for reimbursement policies, as well as the high rate of clinical adoption for liver-related diseases, clearly indicates the maturity level of the United States market. Furthermore, partnerships between research centers and companies facilitate the development of new delivery systems, thus maintaining the leadership position for the U.S. market.
The China market for UDCA is expanding significantly due to the rising rate of liver diseases and the government’s efforts to upgrade the healthcare infrastructure. The major companies, such as Zhejiang Huahai Pharmaceutical and Fosun Pharma, are investing strategically to upgrade the manufacturing and distribution capabilities of UDCA. The new rules are also reducing the time taken to approve new products, which will allow new products to be brought to market more quickly. This will be complemented by increased awareness of UDCA among healthcare professionals and patients, which will drive market adoption. The production of UDCA locally will make the product more affordable, thereby increasing its accessibility to the wider population and increasing China’s influence on the market.
Japan is still at the forefront in terms of clinical research and innovative treatments for hepatic diseases. Organizations such as Takeda Pharmaceutical and Kissei Pharmaceutical have played a vital role in developing advanced UDCA drugs according to patient requirements. Japan has a robust healthcare infrastructure, and the government is supporting chronic disease management. This has resulted in continuous demand for UDCA drugs. Japan is also known for maintaining high standards in terms of product quality and intellectual rights. This has attracted major pharmaceutical companies to invest in Japan, resulting in a strong domestic market for UDCA drugs.
India, as a market, has an increasing cost-conscious but expanding accessibility to UDCA products. The country has some of the largest pharmaceutical companies, such as Lupin, Sun Pharmaceutical, and Cipla, which have helped increase accessibility to UDCA at affordable costs. The government has also introduced initiatives such as Ayushman Bharat, which aims at increasing accessibility to primary medications, including UDCA. India, as a country, has an increasing export infrastructure for pharmaceuticals, which makes it a significant player in the global ursodeoxycholic acid market as both a consumer and supplier.
|
Compound |
Trial Phase |
Trial Indication |
Trial Objective |
|
Nor-ursodeoxycholic acid (norUDCA / norucholic acid) |
Phase III |
Primary Sclerosing Cholangitis (PSC) |
Evaluate efficacy in reducing alkaline phosphatase levels and slowing disease progression in PSC patients |
|
Nor-ursodeoxycholic acid (norUDCA) |
Phase II |
Metabolic dysfunction-associated steatotic liver disease (MASLD / NASH) |
Assess improvements in liver inflammation and fibrosis markers |
|
Nor-ursodeoxycholic acid (norUDCA) |
Phase II |
Cholestatic liver diseases |
Investigate hepatoprotective effects and bile-acid metabolism improvement |
|
UDCA-based combination therapies (e.g., UDCA + fibrates) |
Phase II / III studies |
Primary Biliary Cholangitis (PBC) |
Evaluate improved biochemical response in patients with inadequate response to UDCA monotherapy |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Investing in R&D for enhancing UDCA products, global infrastructure, and partnerships with pharmaceutical companies and healthcare organizations |
Dr. Falk Pharma GmbH markets UDCA-based products such as Ursofalk globally and continues investing in bile-acid therapeutics research, including derivatives like norUDCA. |
|
Mid-Level Players |
Cost-effective production and availability of UDCA therapies for price-sensitive markets, often through partnerships with CMOs and local pharmaceutical companies |
Daewoong Bio and Grindeks manufacture UDCA formulations and expand regional market presence through distribution agreements in Asia and Eastern Europe. |
|
Small-Scale Players |
Opportunity in niche markets through specialized UDCA products, drug delivery systems, and partnerships with local companies. |
Octavius Pharma and Symbiotec Pharmalab supply UDCA APIs and collaborate with regional pharmaceutical companies for formulation development and local market entry. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 600 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 1,050 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
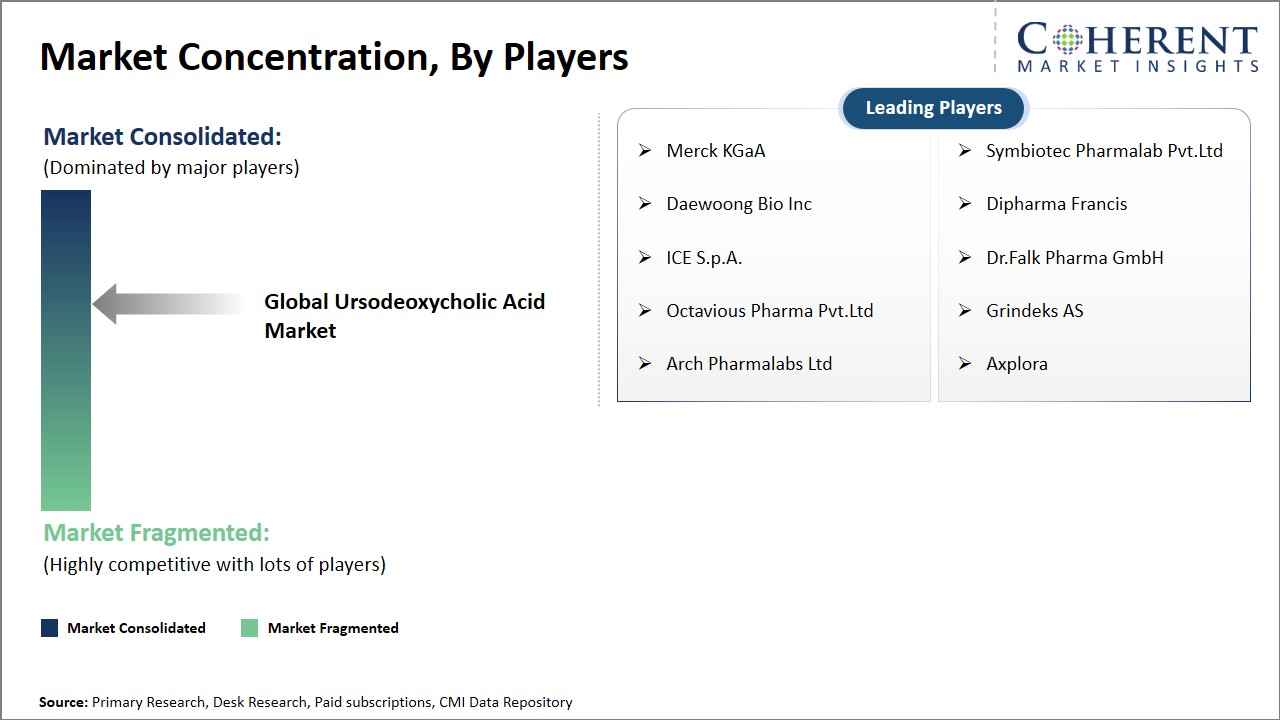
Merck KGaA, Symbiotec Pharmalab Pvt.Ltd, Daewoong Bio Inc, Dipharma Francis , ICE S.p.A., Dr.Falk Pharma GmbH, Octavious Pharma Pvt.Ltd, Grindeks AS, Arch Pharmalabs Ltd, and Axplora |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
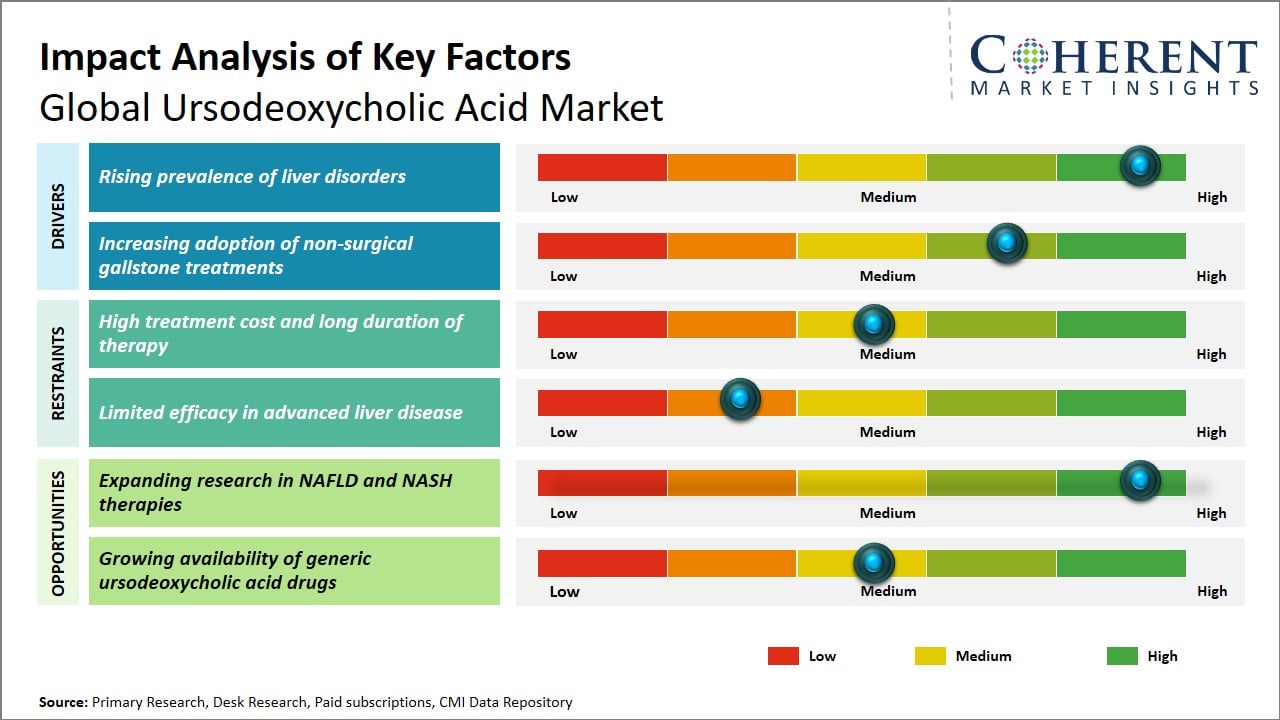
The growing prevalence of liver diseases, including primary biliary cholangitis and non-alcoholic fatty liver disease, is a major factor driving the growth for ursodeoxycholic acid market. The increasing prevalence of liver-related health issues is due to various factors, such as changes in lifestyle, diet, obesity, and excessive alcohol consumption. Therefore, the need for effective treatments for these health problems is growing.
The surging demand for ursodeoxycholic acid, a drug known to treat liver problems, has made it a key treatment for liver-related health issues, particularly cholestatic liver diseases. Furthermore, the growing recognition of liver health concerns among both medical professionals and patients has fueled the escalating demand for ursodeoxycholic acid. This heightened focus on liver-related health problems, coupled with the rising prevalence of these conditions, has substantially driven the increasing need for ursodeoxycholic acid consequently fostering its expansion.
For instance, in February 2025, a research study published by EurekAlert highlighted that ursodeoxycholic acid is effective in the treatment of drug-induced liver injury, also known as DILI, as it helps in lessening the symptoms of cholestasis, oxidative stress, and cell death in the liver. The research study proved that the drug has the capability of preventing and treating DILI when taken along with hepatotoxic drugs.
(Source: EurekAlert)
The increasing rate of demand for therapeutic interventions in treating NAFLD and NASH, as well as ongoing studies and trials conducted to evaluate the efficiency of UDCA in treating liver conditions by reducing liver inflammation and fibrosis, represents a tremendous opportunity in the market for UDCA, which is increasingly becoming a viable option in treating NAFLD and NASH. For instance, in December 2025, the researchers from the Medical University of Vienna published their study and highlighted the therapeutic efficiency of nor-ursodeoxycholic acid (norUDCA) in treating metabolic dysfunction-associated steatotic liver disease (MASLD), in which liver inflammation and fibrosis were reduced, thus providing a boost to the market for UDCA in treating liver conditions such as NAFLD and NASH.
(Source- National Library of Medicine)
In addition, progress in the pathophysiology of NAFLD and NASH has led to an increase in investments in targeted therapies, and UDCA is an important candidate in this regard, given its favorable safety profile and its ability to modulate bile acid composition. Pharmaceutical companies and research institutions are increasingly joining hands to conduct studies on the potential benefits of UDCA in combination therapies, which is expected to improve treatment outcomes. Furthermore, the hepatology therapeutic segment is expected to grow as healthcare professionals become more aware of the potential benefits of UDCA in treating NAFLD and NASH.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients