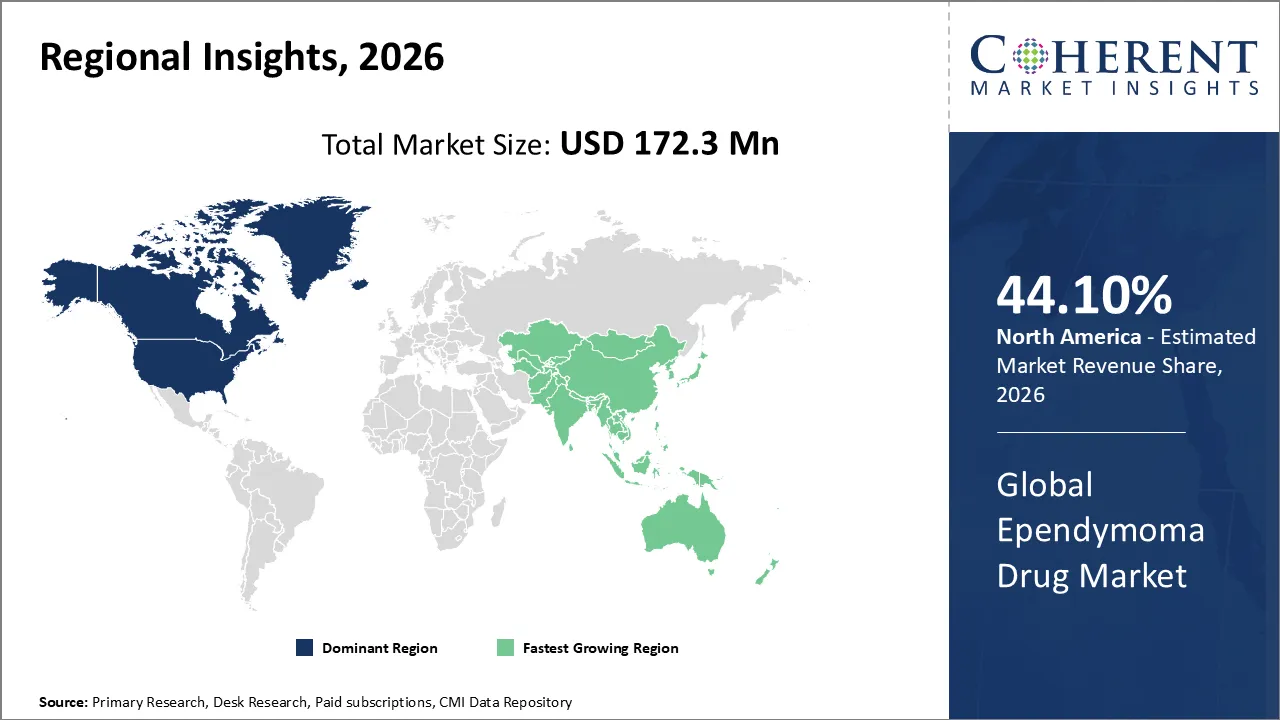
The global ependymoma drug market was valued at USD 172.3 Mn in 2026 and is forecast to reach a value of USD 239.4 Mn by 2033 at a CAGR of 4.8% between 2026 and 2033.
Ependymoma is a type of tumor that can form in the brain or spinal cord. Ependymoma rises from the ependymal cells that line the ventricles of the brain and the center of the spinal cord. Ependymoma can occur at any age, but most often occurs in children under 5 years of age. Children with ependymoma may experience headaches and seizures.
The development of drugs for ependymoma is progressing through systemic therapies, with chemotherapy, targeted agents, and immunotherapy demonstrating quantifiable clinical results. Recent trials underscore the constraints of traditional regimens and the potential of molecularly guided methodologies.
|
Current Event |
Description and its Impact |
|
Regulatory Framework Evolution |
|
|
Technological Breakthroughs in Oncology |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Category |
Details |
|
Incidence |
Ependymoma accounts for 5–10% of pediatric brain tumors; annual incidence is 0.29–0.6 per 100,000 people worldwide. |
|
5‑Year Progression‑Free Survival (PFS) |
Approximately 40–67% after surgery combined with radiotherapy. |
|
5‑Year Overall Survival (OS) |
Approximately 70–83% following standard treatment. |
|
Relapse |
High recurrence rates observed; posterior fossa group A (PF‑A) subtype has the worst prognosis compared to other molecular subtypes. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug type, the corticosteroids segment is projected to account for 42.7% share of the market in 2026, because they are essential to lowering inflammation and controlling intracranial pressure in people with ependymoma. They are often given as a supportive treatment along with surgery and radiation to help with symptoms quickly. Their low cost and proven clinical use give them the biggest market share.
In terms of disease type, the anaplastic ependymoma segment is expected to lead the market with 50% share in 2026, due to it is more aggressive and more common than other types. Patients need a intensive treatment, which makes drugs like corticosteroids and antiseizure agents in high demand. It is the biggest cause of overall drug use because it is so common and has so many side effects.
In terms of distribution channel, the hospital pharmacies segment is expected to hold 60% share of the market in 2026, due to most of them get treatment at specialized oncology and neurology centers. These facilities ensure controlled access to corticosteroids and antiseizure drugs, which are crucial for short-term care. Most of their drug sales come from their reliability, regulatory oversight, and direct connection to treatment pathways.
To learn more about this report, Request Free Sample
North America is expected to lead the ependymoma drug market with 44.10% share in 2026, due to advanced healthcare infrastructure, strong pharmaceutical research and development, high awareness of rare diseases, and FDA approvals that are good for business. Strong clinical trial activity and early use of new cancer treatments make it even more dominant, which leads to more patients being able to get care and more market growth.
For instance, in June 2025, The FDA gave Plus Therapeutics in Austin, Texas, approval for its investigational drug REYOBIQ™ (rhenium Re186 obisbemeda). The Department of Defense is giving $3 million to support the pediatric trial, which will include U.S. patients with recurrent, refractory, or progressive high-grade glioma and ependymoma.
Asia Pacific is expected to exhibit the fastest growth, due to greater resources going into healthcare, better diagnostic tools, and growing patient awareness. China and India are leading the way in growth by supporting the treatment of rare diseases, increasing the number of clinical trials, and making it easier to get oncology treatments. This is all helping the market grow quickly.
For instance, in August 2025, FivepHusion, a biotech company from Australia, is working on Deflexifol, a new type of co-formulation of 5-FU and leucovorin. It is being tested in a national pediatric trial for recurrent or refractory ependymoma, even though it is meant for colorectal cancer. Deflexifol could be the first drug approved for this aggressive childhood brain tumor, thanks to support from cancer foundations.
The U.S. Ependymoma Drug Market is in high demand in 2026 because of advanced healthcare infrastructure, strong pharmaceutical R&D, and high awareness of rare diseases. Good FDA approvals and a robust clinical trial activity speed up access to new treatments, making sure that patients use them and keeping the country at the top of the list for rare cancer treatments.
For instance, in January 2026, The PNOC-027 trial at Columbia University Irving Medical Center in New York used functional precision medicine to treat relapsed medulloblastoma and ependymoma. A national tumor board examines at the results of testing tumor samples against 231 drugs to suggest personalized treatments. The trial is based in the U.S. and is making progress in treating brain cancer in children.
The ependymoma drug market in Australia will be highly competitive in 2026 because more people are aware of rare diseases, diagnostic tools are getting better, and the government is strongly supporting cancer research. Adoption is driven by more people taking part in clinical trials and having access to better treatments. As a result of these factors, Australia is a major player in the growth of the regional market.
For instance, in February 2026, Australia started Paedneo-Vax, the first trial of a pediatric mRNA cancer vaccine in the world. It is aimed at aggressive brain tumors like ependymoma. Each child's tumor profile is used to make personalized vaccines. Sponsored by rovidence Therapeutics and partners, this groundbreaking study aims to improve the chances of survival and treatment options for kids with brain cancers that have relapsed or fail to respond to treatment.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 172.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 239.4 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Fera Pharmaceuticals, Pfizer, Novartis AG, Merck KGaA, Baxter, Cipla Limited, Zydus Cadila, Lupin Pharmaceuticals, Inc., UCB, Inc., APOTEX INC, and Moleculin Biotech, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing number of ependymoma cases around the world is a major factor the ependymoma drug market growth. According to Cancer Therapy Advisor, about 9% of all central nervous system (CNS) tumors in children are ependymomas, and less than 2% of all CNS tumors in adults are ependymomas. In the United States, the estimated annual incidence rate of ependymoma is between 0.29 and 0.6 cases for every 100,000 people. More people are being diagnosed with rare brain tumors due to better diagnostic tools and more awareness of these tumors. This has led to a greater need for effective treatments.
As cancer rates rise, healthcare systems put more emphasis on oncology solutions, which encourages drug companies to put money into research and development. This growing number of patients gives drug companies long-term chances to make money, which keeps the market growing steadily in areas where healthcare spending is high.
Frequent approvals from agencies like the FDA and EMA greatly increase the ependymoma drug market demand. These approvals show that new treatments are safe and effective, which makes doctors and patients more likely to use them. As more drugs become available, treatment options grow, which leads to better patient outcomes and more competition among drug companies. Regulatory support also speeds up clinical trials, which means that people can get advanced treatments faster. This changing environment makes people more confident in the market, which boosts both demand and long-term growth.
A larger population in Asia-Pacific, Europe, and North America are becoming aware of rare brain tumors like ependymoma. This is a big chance for drug companies. Better diagnostic tools, educational programs, and patient advocacy groups are all helping to find problems earlier and find better ways to treat them. This increased awareness pushes healthcare providers to use more advanced treatments, which makes them available to more patients and encourages new ideas. The ependymoma drug market is expected to expand consistently, fueled by an increasing patient population. This growth stems from a rising global awareness of the specific needs associated with this rare form of cancer.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients