Europe Platelet Rich Plasma Market is estimated to be valued at USD 170.2 Mn in 2026 and is expected to reach USD 475.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 15.8% from 2026 to 2033.
Analysts’ Views on Europe Platelet Rich Plasma Market:
Increasing prevalence of orthopedic disorders such as arthritis is expected to drive the market growth over the forecast period. For instance, in September 2020, according to a report published by the National Center for Biotechnology Information (NCBI), stated that rheumatoid arthritis is the commonest of the inflammatory arthritides. Its annual incidence is 20–50 per 100,000 population in European countries.
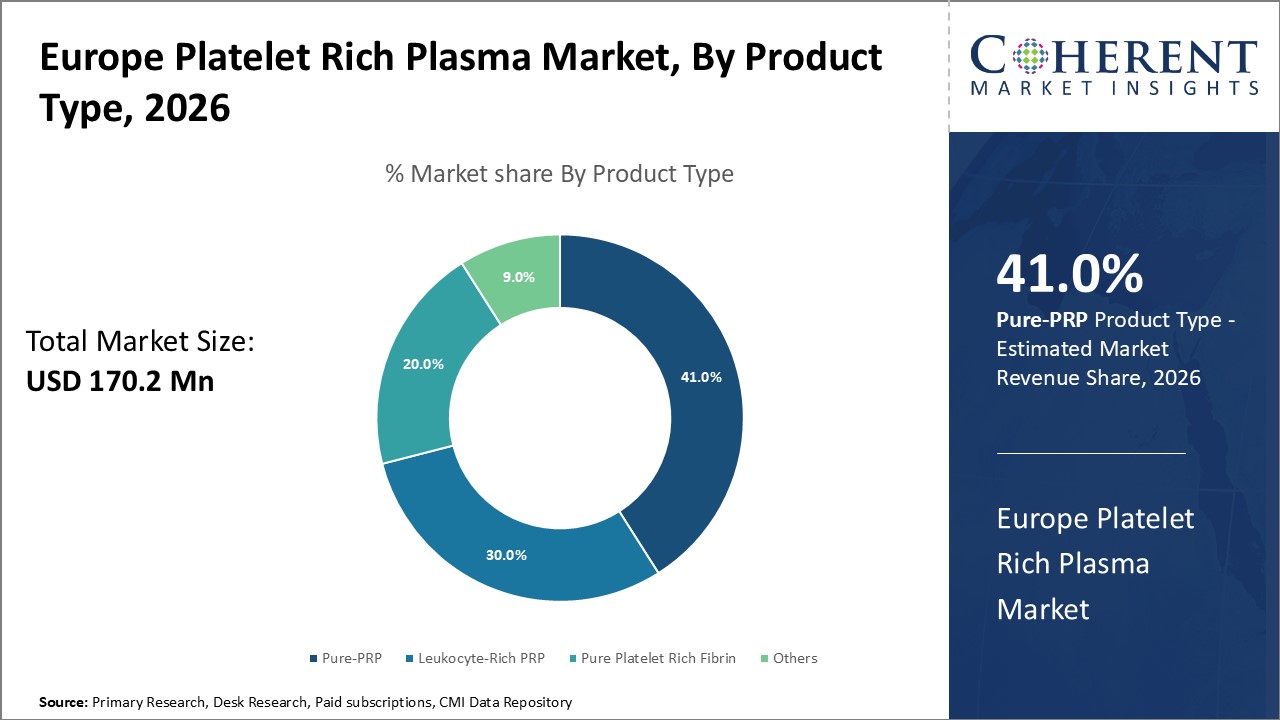
Figure 1. Europe Platelet Rich Plasma Market Share (%), By Product Type, 2026
To learn more about this report, Request Free Sample
Europe Platelet Rich Plasma Market– Drivers
Rising Use of Platelet-Rich Plasma in Various Therapeutic & Clinical Areas
The increasing use of Platelet-Rich Plasma in various therapeutic and clinical areas, is expected to grow the global platelet rich plasma market over the forecast period. For instance, in June 2021, according to the report published in the National Center for Biotechnology Information, platelet-rich plasma (PRP) is becoming more popular as a non-operative treatment option for a broad spectrum of medical disorders. Also, PRP is widely used in orthopedic and sports medicine to relieve pain through the natural promotion of healing in musculoskeletal diseases such as tendonitis, arthritis, ligament sprains, and tears.
High Prevalence of Sports Injuries
The increasing prevalence of sport injuries, is expected to grow the global platelet rich plasma market over the forecast period. For instance, according to the National High School Sports-Related Injury Surveillance Study 2021-2022, 1,385,717 cases of sport-related injuries were recorded in high schools in the U.S.
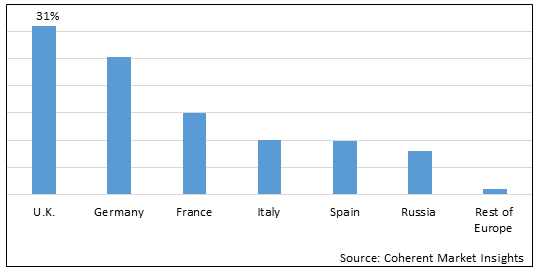
Figure 2. Europe Platelet Rich Plasma Market Value (US$ Million), By Country, 2026
To learn more about this report, Request Free Sample
Europe Platelet Rich Plasma Market- Country Analysis
Among countries, the U.K. is estimated to hold a dominant position in the Europe platelet rich plasma market over the forecast period, due to increasing research and development activities. For instance, on June 5, 2022, a research article published in the Journal of Skin and Steam Cells, where researchers found that under sterile conditions, platelet rich plasma preparation and administration protocol reduced the prevalence of further contamination and complications associated with it. The occurrence of infection after injection was zero, even in patients with hemophilia. When carried out under controlled circumstances, platelet rich plasma therapy is effective for treating a wide range of etiologies and involves both intra-articular and soft tissue operations with no danger of infection.
Europe
Between April and October of 2020, there were 484,415 (17%) fewer total appointments than during the same period in 2019.
Europe Platelet Rich Plasma Market- Segmentation
Europe Platelet Rich Plasma market is segmented into PRP type, origin, application and country.
Based on PRP type, the market is segmented into Pure-PRP, Leukocyte-Rich PRP, Pure Platelet Rich Fibrin, Others. Out of which, the Pure-PRP segment is expected to hold a dominant position in the Europe Platelet Rich Plasma market during the forecast period, owing to increasing use of Pure-PRP.
Based on Origin, the market is segmented into Autologous, Allogeneic/Homologues. Out of which, the Autologous segment is expected to hold a dominant position in the Europe Platelet Rich Plasma market during the forecast period, due to increasing use of autologous platelet rich plama products.
Based on Application, the market is segmented into Orthopaedics, Cosmetic Surgery and Dermatology, Neurosurgery, Cardiothoracic Surgery, General Surgery, Urology, Others. Out of which, the Orthopaedics segment is expected to hold a dominant position in the Europe Platelet Rich Plasma market during the forecast period, due to increasing use of platelet rich plama products for orthopedics.
Based on Country, the market is segmented into U.K., Germany, France, Italy, Spain, Russia, Rest of Europe. Out of which, the U.K. is expected to dominate the market over the forecast period, owing to increasing inorganic growth strategies such as collaboration by key players in the region.
Among all segmentation, the application segment has the highest potential due increasing cosmetic procedures worldwide, which is expected to drive the growth of the segment in the global market over the forecast period. For instance, in 2021, according to Plastic Surgery Statistics Report: 2020, about 16.6 million procedures were carried out, and 13.2 million cosmetic minimally-invasive procedures were carried out.
Europe Platelet Rich Plasma Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 170.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 15.8% | 2033 Value Projection: | USD 475.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Vivostat A/S, Estar Medical, CosmoFrance Inc., Kelly Harris Aesthetics, Arthrex, Inc., Terumo Corporation, T-Biotechnology Ltd. St., EmCyte Corporation, Johnson & Johnson Private Limited, Zimmer Biomet, PRP Concepts, ADVACARE PHARMA, Isto Biologics, Arthrex, Inc., Stryker, GLOFINN, Medira Ltd, Regen Lab SA, Estar Technologies Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Europe Platelet Rich Plasma Market- Cross Sectional Analysis
Among origin, Autologous segment holds a dominant poistion in Europe region due to increasing launch of clinical trials conducted to evaluate the efficiency of PRP extracted by autologous blood in various therapeutic applications such as dermatology. For instance, according to article published by Journal of Experimental and Therapeutics Medicine, in February 2020, a research study was conducted to explore the potential molecular mechanisms for the role of PRP in rejuvenating wrinkled and aged skin and provide evidence for future clinical applications. This study concluded that autologous PRP has the best ratio of growth factors as part of the various functional factors contained in PRP.
Europe Platelet Rich Plasma Market- Key Developments
Increasing inorganic growth strategies such as agreements and acquisitions by key players in the market is expected to drive the market groeth in the forecast period.
In May, 2022, Ester Medical, a pharmaceutical company announced that It had signed an agreement with Aesthetic Management for exclusive long-term marketing and distribution of Eclipse PRP and Tropocells in the U.S. market.
In March 2020, EmCyte Corporation, a manufacturer of medical devices announced the acquisition of all of the assets of Cellmedix Holdings, a biotechnology company, including its Centrepid Platelet Concentrator system. EmCyte’s acquisition of Cellmedix Holdings LLC’s (Cellmedix) assets could expands its intellectual property profile and product development capabilities.
Europe Platelet Rich Plasma Market- Key Trends
Increasing Research and Development Activities By Key Market Players
Increasing research and development activities for PRP by key market players, in order to expand their product portfolio is expected to drive the market growth over the forecast period. For instance, in June 2022, a research article published in the Journal of Skin and Steam Cells, where researchers found that under sterile conditions, platelet rich plasma preparation and administration protocol reduced the prevalence of further contamination and complications associated with it. The occurrence of infection after injection was zero, even in patients with hemophilia. When carried out under controlled circumstances, platelet rich plasma therapy is effective for treating a wide range of etiologies and involves both intra-articular and soft tissue operations with no danger of infection.
Europe Platelet Rich Plasma Market- Restraint
Uncertainty in Efficacy of Platelet Rich Plasma Therapies and Lack of Standardization Increasing recommendations by government bodies against the use of PRP is expected to hinder growth of the market. For instance, in January 2020, the American College of Rheumatology, in partnership with the Arthritis Foundation, released the 2019 ACR/AF Guideline for the Management of Osteoarthritis of the Hand, Hip and Knee, which did not recommend the use of PRP injections for treatment of hip and knee osteoarthritis. Lack of standardization in the preparation and dosage of autologous blood concentrate and platelet-separation techniques is a major factor hindering the market growth. Lack of standardization has led to the lack of strong evidence to support the increasing clinical use of platelet-rich plasma therapy as a treatment modality for musculoskeletal injuries, including orthopedic bone and soft-tissue injuries.
Europe Platelet Rich Plasma Market- Key Players
Major players operating in the Europe platelet rich plama market include Vivostat A/S, Estar Medical, CosmoFrance Inc., Kelly Harris Aesthetics, Arthrex, Inc., Terumo Corporation, T-Biotechnology Ltd. St., EmCyte Corporation, Johnson & Johnson Private Limited, Zimmer Biomet, PRP Concepts, ADVACARE PHARMA, Isto Biologics, Arthrex, Inc., Stryker, GLOFINN, Medira Ltd, Regen Lab SA, Estar Technologies Ltd.
*Definition: Platelet rich plasma (PRP) is a portion of the plasma fraction of analogous blood which contains platelet concentration more than standard concentration. Platelets contain bio proteins that help in healing, tissue regeneration, and blood loss. The use of platelet rich plasma therapy increased after 2009 due to its wide application and no side effects. Platelet rich plasma is used for the treatment of injured tendons, gynecology, muscles, joints, ligaments, and others.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients