Familial Amyloid Cardiomyopathy Treatment Market Size and Forecast – 2026 – 2033
The Global Familial Amyloid Cardiomyopathy Treatment Market size is estimated to be valued at USD 1.1 billion in 2026 and is expected to reach USD 2.3 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 11.2% from 2026 to 2033.
Global Familial Amyloid Cardiomyopathy Treatment Market Overview
The Familial Amyloid Cardiomyopathy Treatment Market comprises therapies and interventions designed to manage and slow the progression of cardiac amyloidosis caused by genetic mutations. Key products include transthyretin (TTR) stabilizers, such as tafamidis, which prevent misfolded protein accumulation in the heart, and gene-silencing therapies that reduce abnormal protein production. Supportive treatments, including heart failure medications, diuretics, and anticoagulants, help manage symptoms and improve quality of life. Additionally, emerging therapies like RNA interference (RNAi) drugs and monoclonal antibodies are gaining traction in clinical trials. These products collectively aim to enhance cardiac function, reduce disease progression, and improve patient survival outcomes.
Key Takeaways
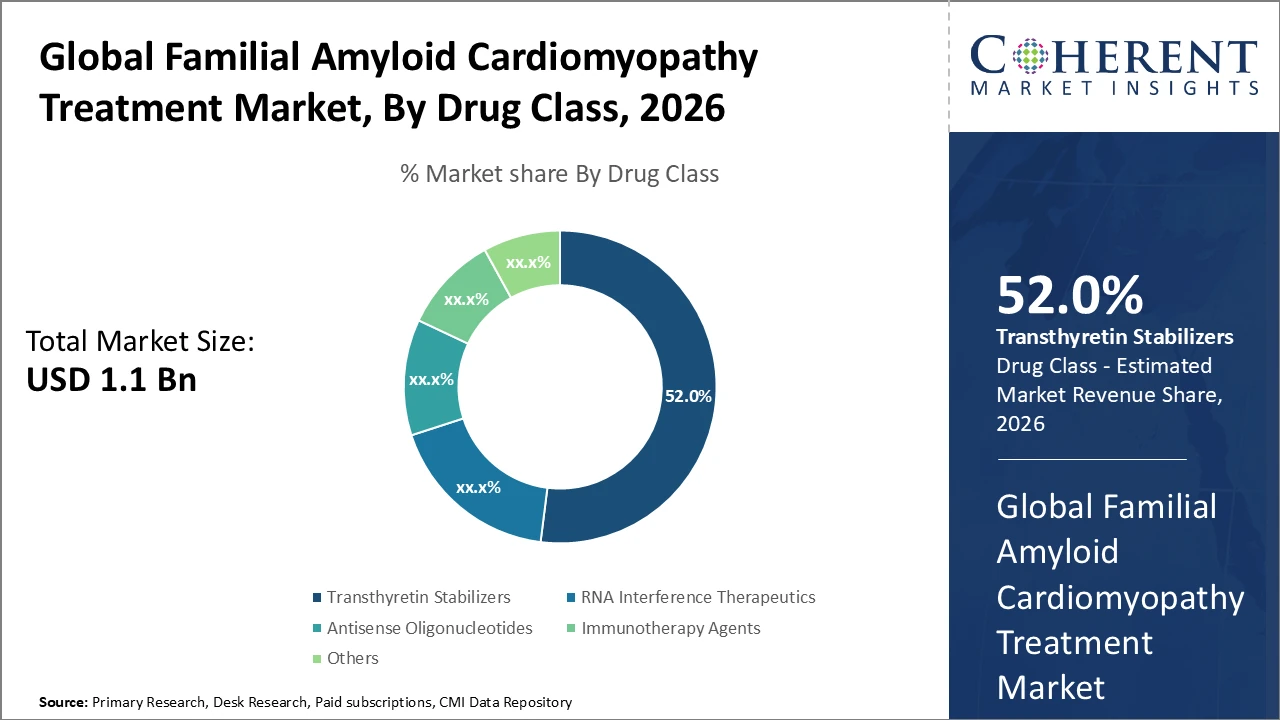
Transthyretin stabilizers dominate treatment segments, holding over half the market share due to proven clinical efficacy and wide regulatory approval in major markets. RNA interference therapeutics, while growing faster, currently contribute a smaller revenue share, indicating potential for future expansion.
Hospital-based treatment remains the primary setting, with ambulatory care models growing rapidly in response to shifts in healthcare delivery and patient management strategies.
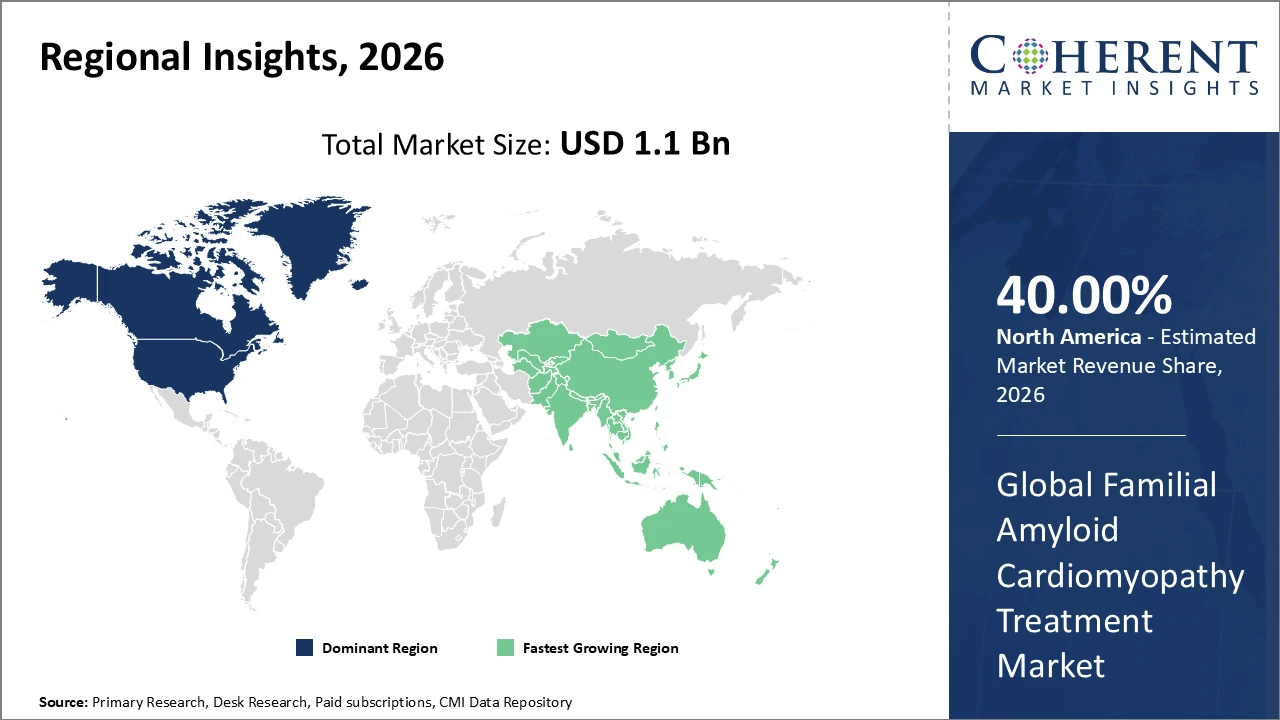
North America leads the market with over 40% share, supported by advanced healthcare infrastructure and favorable reimbursement policies. Asia Pacific is growing at a CAGR of 13.5% due to rising disease awareness and government initiatives, while Europe shows steady growth driven by strong pharmaceutical R&D.
Familial Amyloid Cardiomyopathy Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Familial Amyloid Cardiomyopathy Treatment Market Insights, By Drug Class
Transthyretin stabilizers dominate the market with a 52% share, driven by early regulatory approval, widespread clinical adoption, and proven efficacy in reducing amyloid deposits. They are considered standard care, supported by post-market data demonstrating cardiovascular safety and improved survival. The fastest-growing subsegment is RNA interference therapeutics, which gained traction from 2024 to 2026 clinical trials showing superior amyloid clearance and functional benefits, prompting rapid adoption despite a smaller current market share. Antisense oligonucleotides and immunotherapy agents, though smaller in revenue, target distinct pathogenic pathways with ongoing trials expanding their potential.
Familial Amyloid Cardiomyopathy Treatment Market Insights, By Treatment Settings
Hospital-based treatment remains dominant in the FAC market due to the complexity of managing the disease, which requires multidisciplinary care, advanced diagnostics, and inpatient monitoring. Hospitals generate the majority of market revenue through inpatient administration of complex therapies and post-treatment observation. Ambulatory care settings are the fastest-growing segment, as streamlined administration protocols and patient monitoring technologies allow outpatient management, reducing system burdens and enhancing convenience. Home care is emerging, driven by patient preference for in-home infusions and telemedicine follow-ups, though current revenue is limited.
Familial Amyloid Cardiomyopathy Treatment Market Insights, By Patient Demographics
Hereditary FAC patients constitute the largest segment, supported by increasing genetic screening and family history analysis that enable early diagnosis and treatment eligibility. This group has driven significant market activity, particularly for RNA-targeted therapies addressing specific mutations. Wild-type FAC, representing non-hereditary late-onset cases, is the fastest-growing subsegment due to aging populations in developed markets and heightened awareness among cardiologists for symptom differentiation. Despite diagnostic complexities, expanding clinical trials and real-world evidence have enhanced therapeutic engagement, contributing to revenue growth.
Familial Amyloid Cardiomyopathy Treatment Market Trends
Breakthrough RNA interference therapies approved between 2024 and 2026 are driving market growth, demonstrating the ability to halt disease progression with improved safety profiles.
Phase III trials reported a 50% reduction in cardiovascular-related hospitalizations by 2025, highlighting the clinical impact of these therapies.
Integration of digital health tools, such as remote patient monitoring and adherence tracking, has become an important trend, accelerated by the COVID-19 pandemic and continuing in treatment protocols.
Personalized medicine is gaining prominence, with genetic profiling increasingly guiding therapy selection, as evidenced by a 2026 study across multiple Asia Pacific countries.
Familial Amyloid Cardiomyopathy Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Familial Amyloid Cardiomyopathy Treatment Market Analysis and Trends
In North America, the Familial Amyloid Cardiomyopathy Treatment Market holds over 40% of the regional share, supported by advanced healthcare infrastructure, early adoption of genetic and cardiac diagnostics, and robust reimbursement policies. The United States has benefited from increased government funding for rare diseases and orphan drug initiatives, enhancing market accessibility and revenue potential. Major pharmaceutical companies, including Pfizer and Alnylam, play a pivotal role by developing innovative therapies, expanding patient access, and conducting clinical trials. These factors collectively strengthen North America’s leadership position in the global market, driving adoption of novel treatments and supporting sustained growth in the FAC therapy sector.
Asia Pacific Familial Amyloid Cardiomyopathy Treatment Market Analysis and Trends
Asia Pacific is the fastest-growing region in the Familial Amyloid Cardiomyopathy Treatment Market, with a CAGR exceeding 13%, driven by increasing adoption of genetic testing, government-led healthcare reforms, and growing awareness of disease prevalence. Countries such as Japan and South Korea have established supportive regulatory frameworks and expanded clinical registry programs, facilitating faster patient access to innovative therapies. Key pharmaceutical players, including Akcea Therapeutics and Prothena, have strengthened their regional presence through localized clinical collaborations and strategic partnerships. These initiatives are accelerating market development, improving treatment accessibility, and positioning Asia Pacific as a significant contributor to global FAC therapy growth.
Familial Amyloid Cardiomyopathy Treatment Market Outlook for Key Countries
USA Familial Amyloid Cardiomyopathy Treatment Market Analysis and Trends
The U.S. Familial Amyloid Cardiomyopathy Treatment Market gained significant traction due to a robust rare disease ecosystem and supportive policies, including revisions to the Orphan Drug Act in 2025. Leading companies such as Alnylam Pharmaceuticals have captured substantial market revenue by introducing innovative RNA interference therapies alongside comprehensive patient support programs. Additionally, expanded genetic screening initiatives in cardiology clinics have increased the number of newly diagnosed patients by 18% year-over-year through 2025, improving early treatment access. These developments have strengthened the U.S. market’s share, driving long-term growth potential and reinforcing its leadership position in the global FAC treatment landscape.
Germany Familial Amyloid Cardiomyopathy Treatment Market Analysis and Trends
Germany’s Familial Amyloid Cardiomyopathy Treatment Market is witnessing steady growth, driven by advanced healthcare infrastructure, widespread adoption of genetic testing, and increasing awareness of rare cardiac diseases. Transthyretin stabilizers dominate treatment protocols, while RNA interference therapies are gaining traction through ongoing clinical trials and expanded approvals. Hospital-based treatment remains the primary care setting, complemented by emerging ambulatory and home care models. Government support for rare disease management and reimbursement for innovative therapies further boosts market adoption. Additionally, pharmaceutical companies are investing in localized clinical collaborations and patient support programs, positioning Germany as a key contributor to the European FAC treatment market.
Analyst Opinion
Enhanced diagnostic adoption is driving early therapeutic intervention, with advancements in genetic screening and cardiac imaging increasing early-stage FAC identification by 25% in 2025 compared to 2023, boosting demand for specialized treatments.
Increasing pipeline innovation is accelerating market revenue growth, as next-generation transthyretin stabilizers and RNA-targeting therapeutics gain prominence; over 60% of clinical trial enrollments in 2024 focused on novel modalities, supporting a projected 15% year-over-year revenue increase from 2024 to 2026.
Regional market share shifts reflect policy and reimbursement dynamics, with North America holding over 40% market share, while Asia Pacific grows at a CAGR above 13% due to expanded genetic screening and rising healthcare expenditure.
Growing off-label and combination therapy usage is evident, with a 10% increase in 2026 of physicians combining tafamidis with gene-silencing agents for refractory cases, broadening clinical practice and market scope.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.1 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.2% | 2033 Value Projection: | USD 2.3 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Pfizer Inc., Novartis AG, Vifor Pharma, Moderna Inc., Biogen Inc., Roche Holdings AG, CSL Behring, Sanofi Genzyme, Eidos Therapeutics, Ionis Pharmaceuticals | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Familial Amyloid Cardiomyopathy Treatment Market Growth Factors
The surge in genetic testing and advanced diagnostic technologies has transformed early intervention strategies, driving recent market growth. Accelerated development and approval of RNA interference and antisense therapies between 2024 and 2026 have demonstrated strong clinical efficacy, encouraging broader adoption. Government initiatives supporting rare disease treatment reimbursement, including the U.S. Orphan Drug Act amendments in 2025, have reduced access barriers and enabled faster regulatory approvals. Furthermore, increasing epidemiological awareness among cardiologists and rising patient advocacy efforts are improving diagnosis rates and therapy adherence. Together, these factors create a solid foundation for continued expansion of the Familial Amyloid Cardiomyopathy treatment market.
Familial Amyloid Cardiomyopathy Treatment Market Development
In March 2026, Viz.ai partnered with Alnylam to enhance early detection and streamline diagnosis of cardiac amyloidosis, an underdiagnosed cause of heart failure. Cardiac amyloidosis is a progressive condition caused by misfolded protein buildup in the heart.
Key Players
Leading Companies of the Market
Pfizer Inc.
Novartis AG
Vifor Pharma
CSL Behring
Moderna Inc.
Biogen Inc.
Roche Holdings AG
Eidos Therapeutics
Ionis Pharmaceuticals
Sanofi Genzyme
Competitive strategies in the Familial Amyloid Cardiomyopathy treatment market have focused on mergers, acquisitions, and strategic partnerships to strengthen market position and expand regional reach. In 2025, Alnylam collaborated with Sanofi to accelerate the development of RNA interference therapies while extending their market footprint into emerging regions. Similarly, Pfizer pursued an aggressive licensing and development strategy for transthyretin stabilizers, securing its position as a market leader by 2026 and increasing its market share by approximately 8%. These approaches highlight the importance of strategic alliances and targeted expansion in driving growth and maintaining competitiveness in the FAC treatment landscape.
Familial Amyloid Cardiomyopathy Treatment Market Future Outlook
The future outlook for the Familial Amyloid Cardiomyopathy Treatment Market is highly promising, driven by ongoing innovation in RNA interference, antisense oligonucleotides, and next-generation transthyretin stabilizers. Advances in genetic screening and early diagnostic technologies are expected to expand patient identification, enabling timely therapeutic intervention. Increasing adoption of digital health tools, including remote monitoring and adherence tracking, will enhance treatment outcomes and patient engagement. Supportive government policies and reimbursement frameworks, particularly for rare diseases, will facilitate broader market access. Additionally, growing awareness among cardiologists and patients, coupled with expanding clinical trials, will continue to propel global market growth over the next decade.
Familial Amyloid Cardiomyopathy Treatment Market Historical Analysis
The historical analysis of the Familial Amyloid Cardiomyopathy Treatment Market shows steady growth driven by advances in diagnostics, therapeutics, and rare disease awareness. Initially, transthyretin stabilizers dominated treatment protocols, providing the first effective disease-modifying option for patients. Supportive therapies, including heart failure management and symptom control, complemented early treatment approaches. Between 2020 and 2023, increasing adoption of genetic screening and cardiac imaging improved early diagnosis rates, expanding the patient pool for targeted therapies. The period also saw emerging RNA interference and antisense oligonucleotide therapies progressing through clinical trials, laying the groundwork for innovative treatments that are now accelerating market growth globally.
Sources
Primary Research Interviews:
Executives and operations managers at leading pharmaceutical and biotechnology companies specializing in FAC therapies
Clinical researchers and cardiologists focusing on amyloidosis and hereditary cardiomyopathies
Technology providers for RNA interference, antisense oligonucleotides, and gene-silencing therapeutic platforms
Magazines:
Pharmaceutical Technology – Innovations in Rare Disease Therapies
BioCentury – Drug Development and Clinical Pipeline Updates
Fierce Biotech – Biopharmaceutical Market Developments and Therapeutics
Genetic Engineering & Biotechnology News – Gene Therapy and RNAi Advancements
Journals:
Journal of Cardiac Failure – Management of Familial Amyloid Cardiomyopathy
Orphanet Journal of Rare Diseases – Clinical and Therapeutic Developments in FAC
European Heart Journal – Cardiac Amyloidosis and Genetic Cardiomyopathies
Circulation: Heart Failure – FAC Diagnosis, Treatment, and Patient Outcomes
Newspapers:
The Wall Street Journal – Biopharmaceutical Innovations and Market Trends
Financial Times – Rare Disease Therapy Market Developments
The New York Times Health Section – Updates on Cardiac and Genetic Therapies
BioWorld – News on Rare Disease Drug Approvals and Market Dynamics
Associations:
American Heart Association (AHA) – Cardiac Amyloidosis and Rare Cardiomyopathies
European Society of Cardiology (ESC) – Heart Failure and Amyloidosis Committees
Orphanet – Rare Disease Database and Familial Amyloid Cardiomyopathy Resources
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients