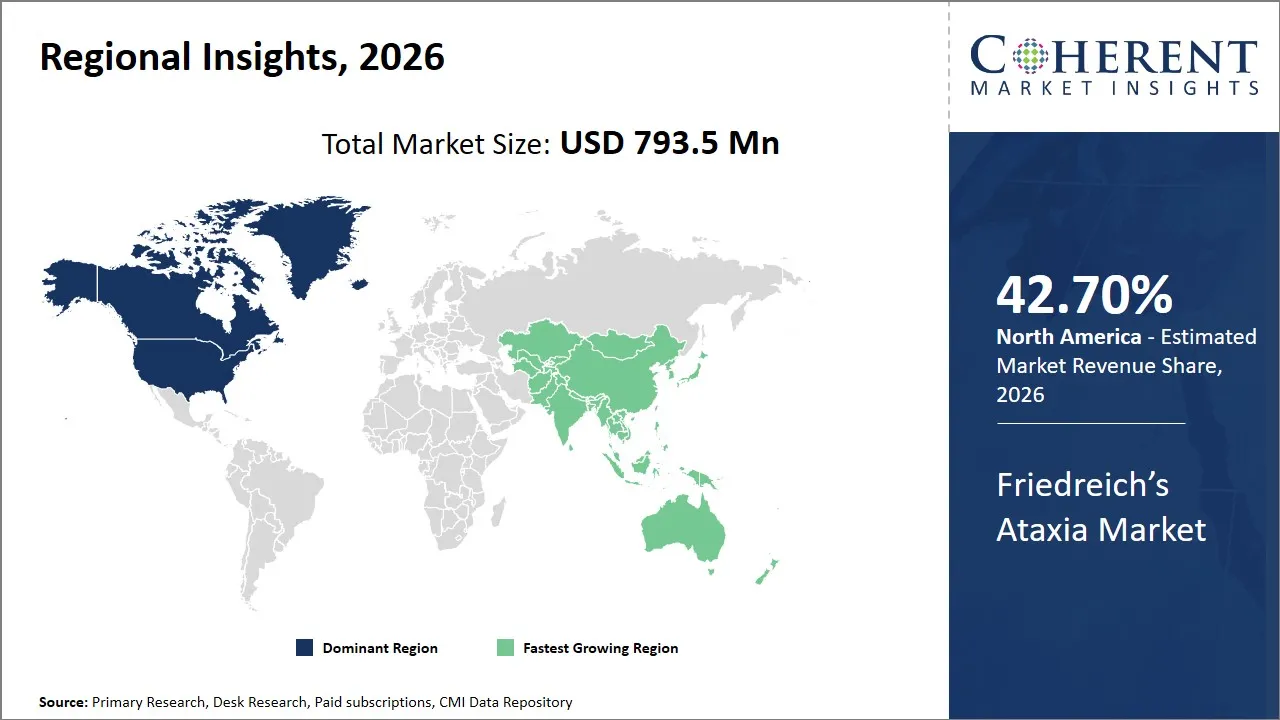
The Friedreich’s Ataxia market was valued at USD 793.5 Mn in 2026 and is forecast to reach a value of USD 1,513.1 Mn by 2033 at a CAGR of 10.5% between 2026 and 2033.
The market for Friedreich’s Ataxia therapies is expanding due to increasing disease awareness, improved genetic diagnostics, and growing research investments in rare neurological disorders. Advances in gene therapy, disease-modifying treatments, and supportive care options are driving market growth. Regulatory incentives for orphan diseases and ongoing clinical trials by biotechnology companies are further supporting innovation, creating opportunities for new therapeutic developments and improved patient outcomes in the coming years.
|
Current Event |
Description and its Impact |
|
Advancements in Gene Therapy for Friedreich’s Ataxia |
|
|
Strategic Partnerships and Funding Initiatives |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug class, the ACE inhibitors segment is expected to lead the market with 40% share in 2026, owing to their proven efficacy in managing cardiac complications associated with Friedreich’s Ataxia (FA), particularly cardiomyopathy, which is a leading cause of mortality in FA patients. The progressive cardiac hypertrophy and fibrosis common in FA lead to heart failure, arrhythmia, and other cardiovascular issues, making ACE inhibitors a cornerstone in therapeutic intervention.
For instance, ongoing developments in heart failure therapies, such as Novartis’ Entresto (LCZ696) pediatric safety survey in chronic heart failure patients (September 2025), highlight continued innovation in cardiac care. While not FA-specific, these studies reinforce the central role of ACE inhibitors in managing heart complications and indicate potential future alternatives that could complement standard FA treatment.
In terms of route of administration, the oral segment is expected to hold 72.8% share of the market in 2026. This dominance of the oral segment can be attributed to several key factors intrinsic to patient preferences, therapeutic convenience, and treatment adherence. Oral medications offer ease of administration, greater patient comfort, and reduced need for professional healthcare settings, making them the preferred mode of therapy for chronic conditions such as Friedreich’s Ataxia.
For instance, Minoryx Therapeutics has announced the ongoing development of Leriglitazone (MIN-102), a novel, orally bioavailable, and selective PPAR gamma agonist with potential best-in-class properties for central nervous system (CNS) diseases. As a metabolite of pioglitazone, Leriglitazone demonstrates sufficient brain penetration and a favorable safety profile in humans, enabling effective engagement of PPAR gamma in the CNS beyond levels achievable with traditional glitazones.
In terms of distribution channel, the hospital pharmacies segment is projected to account for 48.1% share of the market in 2026. This dominance of the Oral segment can be attributed to several key factors intrinsic to patient preferences, therapeutic convenience, and treatment adherence. Oral medications offer ease of administration, greater patient comfort, and reduced need for professional healthcare settings, making them the preferred mode of therapy for chronic conditions such as Friedreich’s Ataxia.
For many patients, managing this progressive neurodegenerative disorder involves long-term treatment regimens, and oral formulations significantly enhance compliance by allowing self-administration without frequent clinical visits or invasive procedures. This is particularly important since Friedreich’s Ataxia patients often face mobility challenges, making in-clinic injectable administration logistically difficult. International non-profit organizations like the Friedreich’s Ataxia Research Alliance (FARA) stress the importance of patient-centered therapies that minimally disrupt daily life, a factor that strongly favors oral medications.
To learn more about this report, Request Free Sample
North America is expected to dominate the Friedreich’s Ataxia market with 42.70% share in 2026, owing to several critical factors tied to the healthcare infrastructure, research initiatives, and the presence of pivotal patient advocacy groups. The United States, in particular, stands out due to its advanced genetic research capabilities and an established healthcare system that prioritizes rare disease management.
For instance, in June 2025, Design Therapeutics, Inc., a biotech company working on treatments for serious genetic diseases, announced that the first Friedreich’s Ataxia (FA) patient has received an intravenous (IV) dose of DT-216P2. This is part of their RESTORE-FA Phase 1/2 clinical trial, which tests increasing doses of the therapy to evaluate safety and effectiveness.
Asia Pacific is expected to exhibit the fastest growth, due to increasingly sophisticated healthcare infrastructure, expanding patient awareness, and rapidly advancing biotechnology sectors, especially in countries such as Japan, China, South Korea, and India. Recent developments illustrate the rising emphasis on rare genetic disorders, including Friedreich’s Ataxia, as public health priorities in these countries evolve from merely infectious disease control toward managing chronic and genetic illnesses.
In particular, China’s National Health Commission has issued specific policy frameworks supporting rare disease diagnosis and treatment pathways that catalyze research and clinical care for Friedreich’s Ataxia. For example, the China Rare Diseases Registry System, established to collect and analyze patient data for over 70 rare diseases, has progressively included neurodegenerative disorders such as Friedreich’s Ataxia, thus enabling more precise epidemiological understanding and accelerating drug development access.
the U.S. contributes the highest share in the Friedreich’s Ataxia market, owing to a combination of extensive research infrastructure, advanced healthcare systems, and a well-established network of clinical trial centers specializing in rare neurodegenerative disorders. The U.S. leads largely because it houses a concentration of dedicated research institutions such as the National Ataxia Foundation (NAF), which actively funds and supports Friedreich’s Ataxia research and patient advocacy.
For instance, in February 2025, PTC Therapeutics, Inc. announced that the U.S. FDA has accepted its application to approve vatiquinone, a treatment for Friedreich’s Ataxia (FA) in children and adults. The FDA has given the application Priority Review, with a decision expected by August 19, 2025. The company is currently valued at $3.82 billion, and its stock has increased by over 90% in the past year.
The China subsegment contributes the highest share in the Friedreich’s Ataxia market owing to several critical factors rooted in its extensive healthcare infrastructure developments, growing governmental focus on rare diseases, and expanding research initiatives in neurodegenerative disorders. China’s significant patient population base and rising awareness about rare inherited ataxias underpin its leadership in the region.
For instance, the China Rare Diseases Diagnosis and Treatment Alliance, established under the National Health Commission, has been pivotal in consolidating patient registries and facilitating multi-center clinical research across prominent Chinese hospitals. These efforts have improved diagnostic accuracy and treatment protocols tailored for Friedreich’s Ataxia, helping China maintain its dominant position within APAC.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 793.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.5% | 2033 Value Projection: | USD 1,513.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Reata Pharmaceuticals, Inc., Retrotope Inc., Minoryx, PTC Therapeutics, Design Therapeutics, Inc., Larimar Therapeutics, Inc., Jupiter Neurosciences, Inc., Lexeo Therapeutics, Zydus Lifesciences Ltd., Cipla Limited, GlaxoSmithKline Plc., Aurobindo Pharma Ltd., Sun Pharmaceutical Industries Ltd., Torrent Pharmaceuticals Ltd., and Intas Pharmaceuticals Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of Friedreich’s Ataxia (FA) globally is a significant factor propelling the expansion of the Friedreich’s Ataxia market. FA, a rare genetic disorder characterized by progressive damage to the nervous system, leading to symptoms such as gait disturbance, speech problems, and heart disease, has seen a rise in diagnosed cases partly due to improved genetic screening methods and awareness initiatives.
The increasing investment in research and development (R&D) activities is a pivotal factor driving the advancement and growth of the Friedreich’s Ataxia market. Friedreich’s Ataxia, a rare inherited neurodegenerative disorder characterized by progressive damage to the nervous system and impaired muscle coordination, has long been an area with limited therapeutic options. However, heightened funding and focus on innovative research programs are fostering the discovery and development of novel treatment candidates and diagnostic tools.
The development of novel gene editing and therapy techniques presents a transformative opportunity within the Friedreich’s Ataxia (FA) market, fundamentally reshaping treatment paradigms for this rare neurodegenerative disorder. Friedreich’s Ataxia is primarily caused by a genetic mutation resulting in the deficiency of frataxin, a protein vital to mitochondrial function. Traditional therapeutic approaches have largely focused on symptom management and slowing disease progression, often with limited efficacy.
The Friedreich’s Ataxia (FA) market is witnessing significant momentum driven by growing awareness of this rare genetic disorder and advancements in therapeutic research. Key drivers include increasing investments in gene therapy and innovative drug development focused on symptom management and disease modification. Additionally, rising diagnosis rates due to improved genetic testing contribute to greater patient identification, fueling demand for targeted treatments.
However, the market faces notable challenges such as the complex pathophysiology of Friedreich’s Ataxia and limited understanding of its progression, which hinder rapid drug development. Furthermore, high treatment costs and a relatively small patient population limit widespread commercial viability, posing restraint on market expansion.
On the opportunity front, ongoing clinical trials exploring novel molecules and the potential of gene-editing technologies offer promising avenues to address unmet medical needs. Collaborations between pharmaceutical companies, research institutions, and patient advocacy groups are also enhancing innovation and facilitating faster regulatory approvals.
Geographically, North America dominates the Friedreich’s Ataxia market, driven by advanced healthcare infrastructure, availability of funding, and a strong presence of key market players. Europe follows closely, benefiting from robust rare disease frameworks and increasing government support. Meanwhile, the Asia-Pacific region is emerging as the fastest-growing market due to improving healthcare access and rising awareness.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients