The pharmaceutical market consists of sales of pharmaceuticals and related services by entities (organizations, sole traders, and partnerships) that produce pharmaceuticals used in treating diseases such as diabetes, cancer, cardiovascular disease, chronic kidney diseases, and others. Pharmaceuticals can be any type of drugs that are used for medicinal purposes, in the treatment of diseases . This industry includes organizations or companies that produce biologics and pharmaceutical drugs. Pharmaceutical companies are offering drugs for customized individual treatment for various diseases as listed above. Personalized medicine, also referred to as precision medicine, aims to provide medical care according to the patient's individual characteristics and genetic makeup. Precision therapies are increasingly being adopted as firms increasingly let go of the one-size-fits-all model for common medical conditions such as lung cancer, melanoma (skin cancer), colon cancer, and pancreatic cancer. It can also help with some rare childhood illnesses, cystic fibrosis, and HIV.
The India & Oman pharmaceutical industry market is estimated to be valued at US$ 47,179.5 million in 2021 and is expected to exhibit a CAGR of 12.7% over the forecast period (2021-2030).
Figure 1. India and Oman Pharmaceutical Industry Market Share (%) in Terms of Value, By Country, 2021
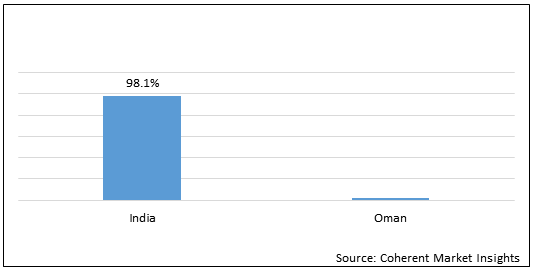
To learn more about this report, Request Free Sample
Increasing number of product launches in the India pharmaceutical industry is expected to drive the market growth over the forecast period.
The increasing number of product launches in the India pharmaceutical industry is expected to drive growth of the India pharmaceutical industry market over the forecast period. For instance, in February 2021, Glenmark Pharmaceuticals, a pharmaceutical company in India, announced the launch of SUTIB, a generic version of Sunitinib oral capsules indicated for the treatment of kidney cancer in India.
India & Oman Pharmaceutical Industry Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 47,179.5 Mn |
| Historical Data for: | 2011 to 2020 | Forecast Period: | 2021 to 2030 |
| Forecast Period 2021 to 2030 CAGR: | 12.7% | 2030 Value Projection: | US$ 138,308.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., Bristol Myers Squibb, Sanofi S.A., F. Hoffmann-La Roche AG, Bayer AG, Novartis International AG, Merck & Co., Inc., AbbVie, GlaxoSmithKline plc, Eli Lilly and Company, Zydus Cadila, Aurobindo Pharma Ltd., Cipla Ltd., Dr. Reddy's Laboratories Ltd., Lupin Ltd., Sun Pharmaceutical Industries Limited, Serum Institute of India Pvt. Ltd., Biocon Limited, Strides Pharma Science Limited, and Unichem Laboratories |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. India & Oman Pharmaceutical Industry Market Share (%), by Drug, 2020
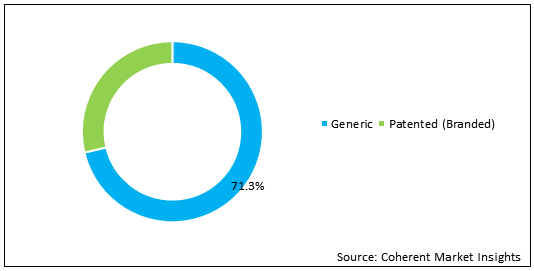
To learn more about this report, Request Free Sample
The increasing prevalence of chronic diseases in Oman is expected to drive the market growth over the forecast period.
For instance, according to The Ministry of Health (MOH) report 2017, the number of recorded tumor cases in 2017 reached 2,101 including 1,892 (90.05%) cases among Omanis, 188 (8.95%) cases among residents, and 19 (0.9%) cases of Carcinoma in situ among Omanis. Furthermore, according to the similar source, the number of cancer cases among Omani males reached 887 (46.9%) compared to 1,005 (53.1%) cases among Omani females. 127 (6.7%) cases were registered among Omani children at the age of 14 and under in 2017.
India & Oman Pharmaceutical Industry market– Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread to over 100 countries across the globe and the World Health Organization declared it a public health emergency in 2020. According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, the number of COVID-19 cases reported in India as of July 8, 2021 are approximately 30,663,665 confirmed cases and 404,211 deaths. Moreover, according to the same source, the number of COVID-19 cases reported in Oman as of July 8, 2021 are approximately 278,560 confirmed cases of COVID-19 and 3,339 deaths. The major challenges faced by key players include supply of raw materials required for manufacturing instruments and reagent kits due to irregularities in transportation facility, irregular demand for products from the retailers due to increasing number of patients suffering from COVID-19 and other life threatening disorders.
Thus, to overcome the dependency of active pharmaceutical ingredient (API) and raw materials, the Indian government is focused on developing and manufacturing of raw materials and active pharmaceutical ingredients. For instance, in 2020, the government of India announced the incentive package of INR 13.76 billion (US$ 18.39 Bn) for the promotion of domestic manufacturing of critical key starting materials, drug intermediates, APIs, and medical devices in India. Moreover, according to the Directorate-General of Pharmacy and Drug Control Oman 2020, in Oman there was prohibition on the export of masks, hand sanitizers, and medical supply products to the other countries due to shortage of the pharmaceutical appliances and products in Oman.
India & Oman Pharmaceutical Industry Market: Restraint
High number of banned products in India and lack of national health insurance plan in Oman are expected to hinder growth of the India & Oman pharmaceutical industry market. For instance, according to the Central Drugs Standard Control Organization and Directorate General of Health Services Ministry of Health & Family Welfare Government of India, banned drugs in 2018 include combinations of Nimesulide + Levocetrizine, Ofloxacin + Ornidazole Injection and Glucosamine + Ibuprofen due to their side effects and drug abuse situations. Whereas, lack of national health insurance plan in Oman leads to the expenses of the public healthcare system to be depended on the government, perpetuating reliance on state provisions and straining the government budget and is expected to hinder the growth of the market.
Key Players
Major players operating in the India & Oman pharmaceutical industry market include Pfizer, Inc., Bristol Myers Squibb, Sanofi S.A., F. Hoffmann-La Roche AG, Bayer AG, Novartis International AG, Merck & Co., Inc., AbbVie, GlaxoSmithKline plc, Eli Lilly and Company, Zydus Cadila, Aurobindo Pharma Ltd., Cipla Ltd., Dr. Reddy's Laboratories Ltd., Lupin Ltd., Sun Pharmaceutical Industries Limited, Serum Institute, Biocon Limited, Strides Pharma Science Limited, and Unichem Laboratories
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients