Interferon Beta Drugs Market is estimated to be valued at USD 4.76 Bn in 2026 and is expected to reach USD 6.35 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.2% from 2026 to 2033.
Analysts’ Views on Global Interferon Beta Drugs Market:
An increasing number of research and development and drug discovery activities are expected to boost the growth of the global interferon beta drugs market over the forecast period. For instance, on January 4, 2023, scientists working at Brigham and Women’s Hospital, a hospital that conducts hospital-based research programs, developed a bifunctional whole cancer cell–based therapeutic with direct tumor killing and immunostimulatory roles. The scientists repurposed the tumor cells from interferon-β (IFN-β) sensitive to resistant using CRISPR-Cas9 by knocking out the IFN-β–specific receptor and subsequently engineered them to release the immunomodulatory agents IFN-β and granulocyte-macrophage colony-stimulating factor.
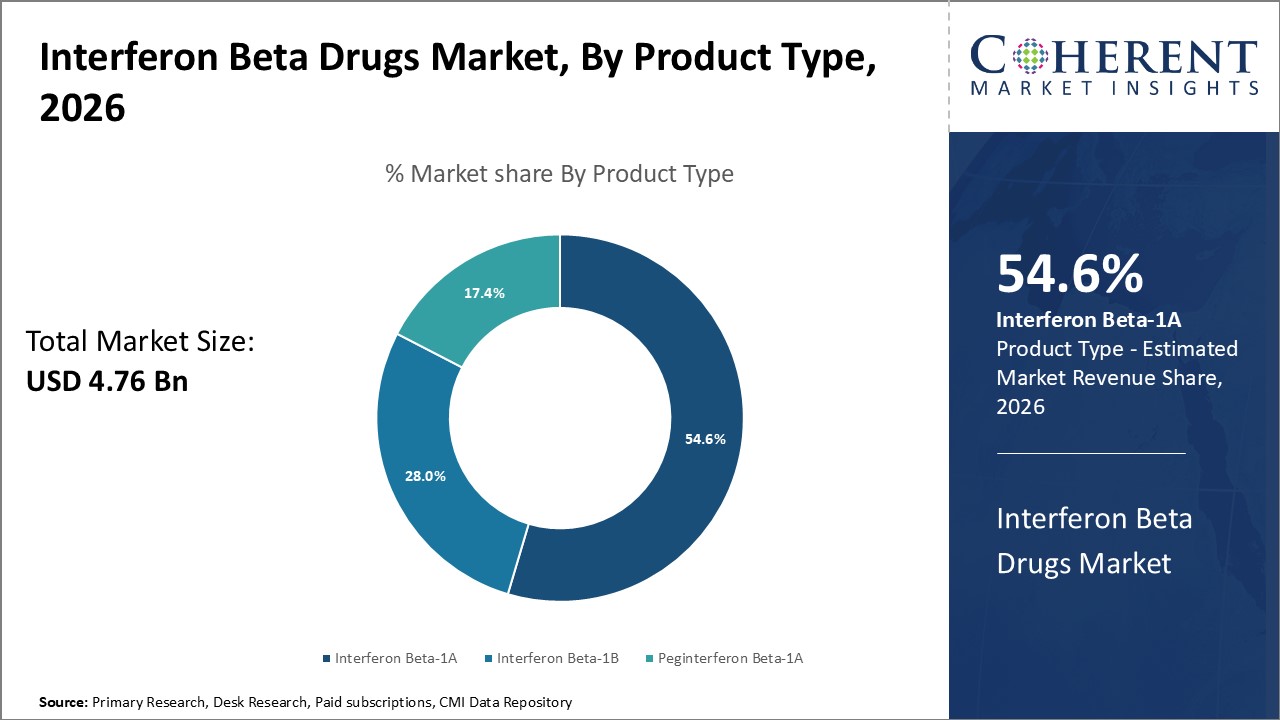
Figure 1. Global Interferon Beta Drugs Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Interferon Beta Drugs Market – Driver
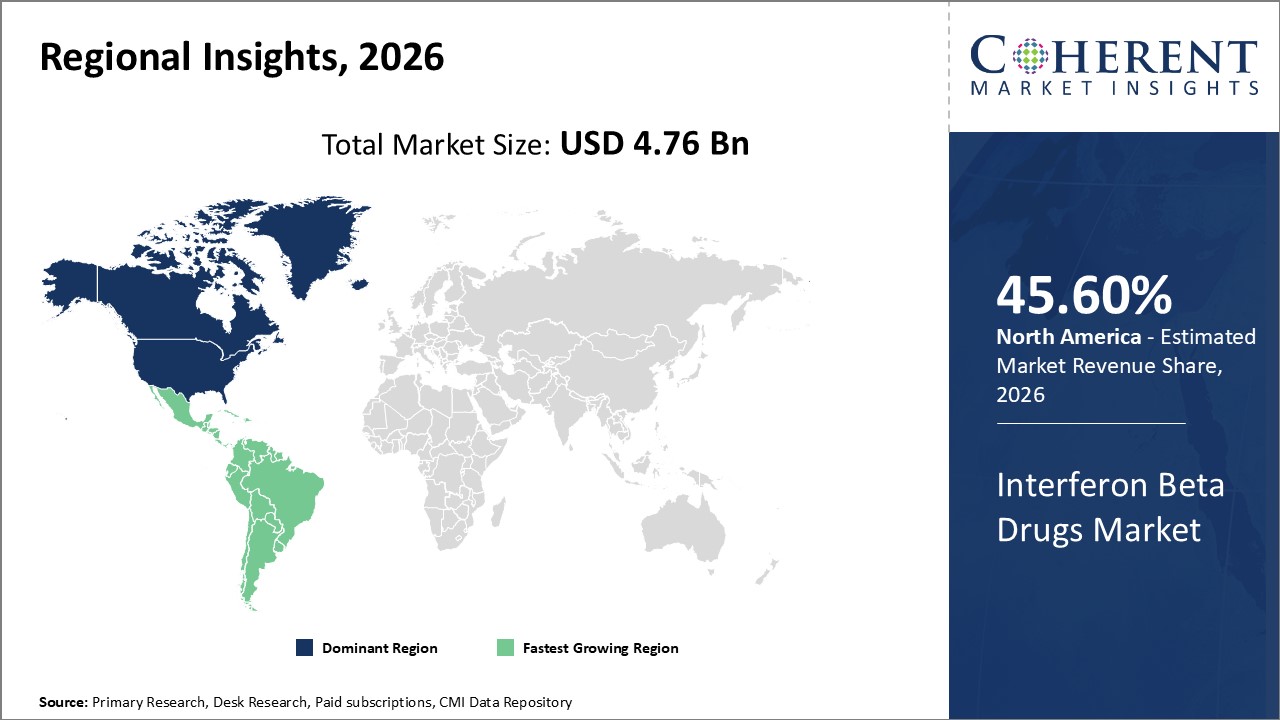
Figure 2. Global Interferon Beta Drugs Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Interferon Beta Drugs Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global interferon beta drugs market over the forecast period. This is due to the increasing research and development using beta interferons in the region. For instance, according to an article by Northwestern Medicine, a non-profit healthcare system affiliated with the Northwestern University Feinberg School of Medicine in Chicago, U.S., in April 2026, scientists have discovered a novel signaling pathway activated by beta interferons, a group of immune system proteins, that suppresses the anti-tumor response of interferons in patients with a particular type of blood cancer.
Global Interferon Beta Drugs Market Segmentation:
The global interferon beta drugs market report is segmented into product type, route of administration, distribution channel, and region.
Among all the segments, the product type segment has the highest potential due to the launches of new products across the world over the forecast period. For instance, in May 2020, EMD Serono, the biopharmaceutical business of Merck KGaA, announced that the U.S. Food and Drug Administration (FDA) had approved the inclusion of new safety data on pregnancy and breastfeeding in the prescribing information for Rebif (interferon beta-1a), in accordance with the FDA's Pregnancy and Lactation Labeling Rule (PLLR).
Global Interferon Beta Drugs Market Cross Sectional Analysis:
Among product type, interferon beta-1A is expected to dominate the market over the forecast period in the Europe region. For instance, on November 19, 2022, according to an article published in Neuro-Oncology, a monthly peer-reviewed medical journal covering cancer of the nervous system, published by Oxford University Press in the U.K., adding interferon beta- 1A to adjuvant temozolomide radiotherapy prolongs survival in high-grade glioma.
Global Interferon Beta Drugs Market: Key Developments
Global Interferon Beta Drugs Market: Key Trends
Interferon Beta Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.76 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.2% | 2033 Value Projection: | USD 6.35 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Biogen, Merck KGaA, Novartis AG and Bayer AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Interferon Beta Drugs Market: Restraint
Global Interferon Beta Drugs Market - Key Players
Major players operating in the global interferon beta drugs market include Biogen, Merck KGaA, Novartis AG and Bayer AG.
*Definition: Interferon beta-1b injection is used to reduce episodes of symptoms in patients with relapsing-remitting (a course of disease where symptoms flare up from time to time) of multiple sclerosis (MS, a disease in which the nerves do not function properly and patients may experience weakness, numbness, loss of muscle coordination, and problems with vision, speech, and bladder control). Interferon beta-1b is in a class of medications called immunomodulators. It is not known exactly how interferon beta-1b works to treat MS.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients