The bone cancer drugs market is estimated to be valued at USD 1,500.2 Mn in 2026 and is expected to reach USD 2,167.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.4% from 2026 to 2033.
To learn more about this report, Request Free Sample
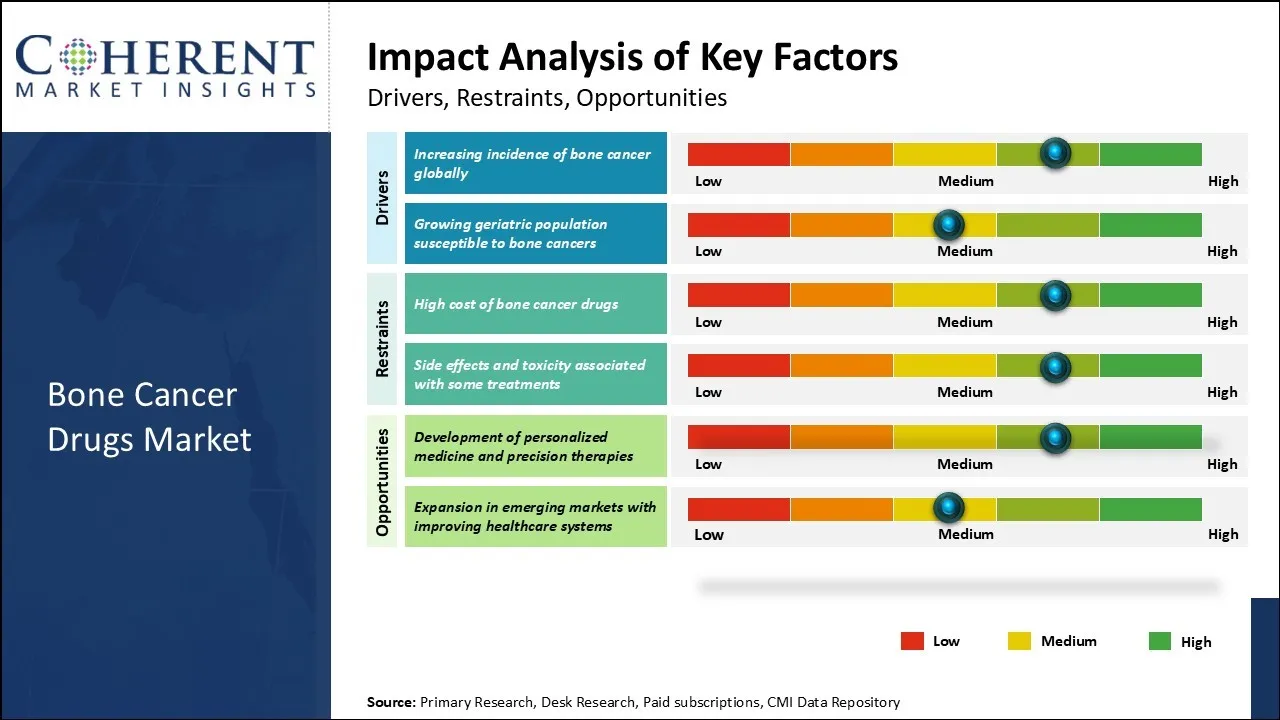
The global bone cancer drugs market is witnessing steady growth driven by the increasing prevalence of bone cancers such as osteosarcoma, Ewing sarcoma, and chondrosarcoma, alongside advancements in targeted therapies and immunotherapies. Rising awareness, improved diagnostic capabilities, and supportive governmental initiatives further amplify the demand for innovative treatment options in this niche but critical therapeutic area.
However, limited availability of effective treatments for rare bone cancer types is one of the factors restraining the market growth.
|
Current Events |
Description and its impact |
|
Regulatory Advancements in Oncology Drug Approvals |
|
|
Geopolitical and Trade Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing incidence of bone cancer globally is a significant factor propelling the demand for bone cancer drugs, thereby influencing the overall expansion of the bone cancer drugs market. Bone cancer, although relatively rare compared to other cancers, has witnessed a noticeable rise in diagnosed cases, attributed to improved diagnostic techniques and greater awareness among healthcare professionals and patients.
For instance, according to the American Cancer Society's Cancer Facts & Figures 2025, primary bone and joint cancers are projected to account for approximately 3,770 new cases and 2,190 deaths in the U.S. in 2025.
In terms of drug type, the antimetabolites segment is estimated to contribute the highest market share of 37.2% in 2026, owing to their mode of action, and strong backing from well-established treatment protocols.
Antimetabolites act primarily by interfering with DNA and RNA synthesis, thereby inhibiting the proliferation of cancerous cells, which is a crucial mechanism given that bone cancers such as osteosarcoma and Ewing sarcoma typically involve rapidly dividing malignant cells. The widespread adoption of drugs like methotrexate and cytarabine in chemotherapy regimens has solidified the position of antimetabolites in treatment strategies globally.
To learn more about this report, Request Free Sample
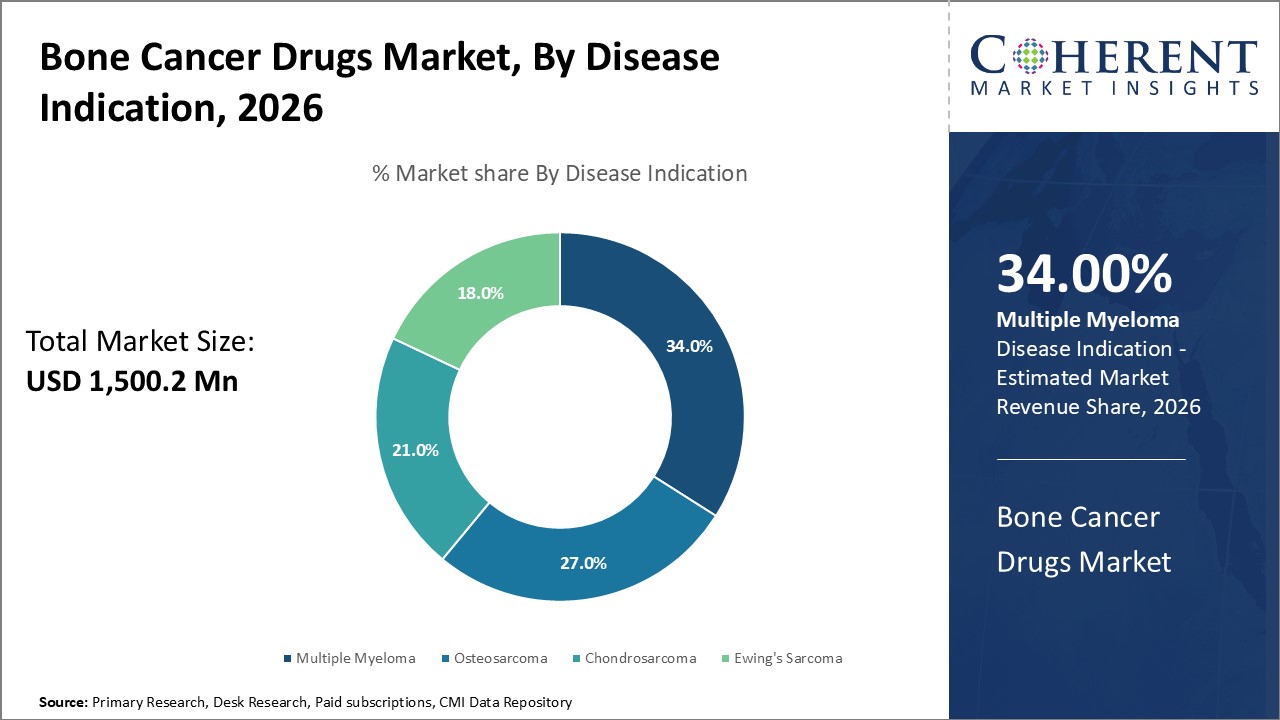
In terms of disease indication, the multiple myeloma segment is estimated to contribute the highest market share in 2026, owing to the increasing prevalence of multiple myeloma across the globe.
The chronic and relapsing nature of multiple myeloma has created persistent demand for innovative drugs that can manage the disease over extended periods, thereby increasing the market share of drug therapies addressing this condition. Breakthroughs in Immunomodulatory Drugs (IMiDs), proteasome inhibitors, and monoclonal antibodies have been instrumental in transforming the treatment landscape for multiple myeloma, encouraging sustained investment and a focus by pharmaceutical companies.
In terms of distribution channel, the hospital pharmacies segment is estimated to contribute the highest market share in 2026, largely attributable to their critical role within healthcare infrastructure and direct integration with oncology treatment protocols.
Hospital pharmacies serve as key touchpoints in the management and dispensing of bone cancer medications, primarily because of the specialized and often complex nature of bone cancer treatment. Unlike many other medicinal areas, bone cancer typically requires highly coordinated care, including chemotherapy, radiotherapy, surgery, and targeted drug therapy, all of which are frequently administered or monitored in hospital settings.
By integrating AI into drug discovery, development, and patient management, companies are accelerating innovation while improving outcomes.
To learn more about this report, Request Free Sample
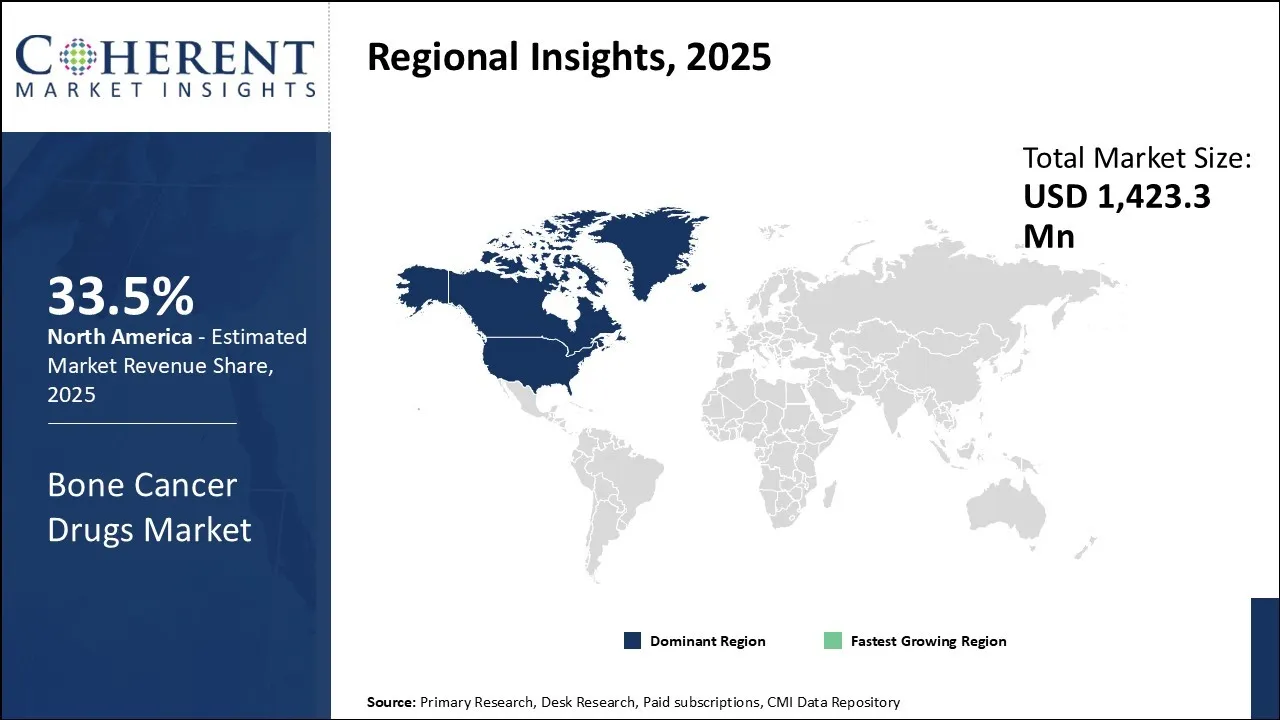
North America is estimated to hold a dominant position in the global bone cancer drugs market over the forecast period. North America is estimated to hold 33.5% of the market share in 2026. Global bone cancer drugs market is expected to witness significant growth in the near future due to high prevalence of bone cancer, favorable health reimbursement, and increased awareness. Increasing prevalence of neurological disorders is contributing to growth of the bone cancer drugs market in North America region.
For instance, as per the American Cancer Society, primary bone cancers (cancers that start in the bones) are uncommon, accounting for less than 1% of all cancers. In adults, cancers that spread to the bones from other organs (known as bone metastasis) are much more common than primary bone cancers.
The U.S. bone cancer drugs market is driven by its robust healthcare infrastructure and significant investments in cancer research. The U.S. Food and Drug Administration (FDA) plays a major role by providing expedited approvals for innovative therapies, including orphan drugs for rare cancers. This regulatory support accelerates the availability of new treatments.
Germany's bone cancer drugs market is significantly bolstered by its comprehensive healthcare system and emphasis on preventive care. The country's well-established referral pathways ensure timely access to specialized care, which is crucial for effective bone cancer treatment. Germany's focus on health education as well as early detection programs leads to higher awareness among the population, promoting early diagnosis and intervention.
India's bone cancer treatment landscape is characterized by distinct challenges, notably the prevalence of advanced-stage diagnoses and constrained access to specialized care in rural regions. These issues are compounded by a shortage of medical professionals in underserved areas, leading to delays in diagnosis and treatment. However, concerted efforts are underway to address these disparities.
China is swiftly advancing in the realm of bone cancer treatment, driven by substantial investments in healthcare infrastructure and research. The National Medical Products Administration's approval of innovative therapies, such as radionuclide-drug conjugates for bone metastases, underscores the nation's commitment to expanding treatment options. Collaborations between Chinese biotech firms and Western pharmaceutical companies are fostering the development of competitive therapies, including PD-1 and PD-1 VEGF inhibitors.
|
Drug Category |
Price Range per Treatment Cycle/Month (USD) |
|
Chemotherapy |
USD 300 – USD 1,800 |
|
Targeted Therapies |
USD 3,500 – USD 13,000 |
|
Immunotherapy |
USD 10,000 – USD 15,000 per dose |
|
Bone-Modifying Agents |
USD 500 – USD 4,200 per dose |
|
Supportive Care |
USD 20 – USD 3,000 |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,500.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.4% | 2033 Value Projection: | USD 2,167.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Advaxis, Inc., Cellectar Biosciences, Inc., OPKO Health, Inc., Pfizer Inc., Amgen Inc., Novartis AG, Eli Lilly and Company, Debiopharm Group, Merck & Co, Bayer AG, Bristol-Myers Squibb Company, Takeda Pharmaceutical, F. Hoffmann-La Roche Ltd, and Teva Pharmaceutical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Bone cancer develops in bone or cartilage cells. Primary bone cancer is another name for it. A mass of cancer cells known as a cancerous (malignant) tumour is capable of invading neighboring tissue and wreaking havoc on it. It has the ability to "metastasize" to other body regions. Rising number of patient population with bone cancer is expected to boost demand for bone cancer treatment across the globe. Thus, it offers the opportunity for various market players to offer cost effective treatment.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients