Listeria Monocytogenes Infections Treatment Market Size and Forecast – 2026 – 2033
The Global Listeria Monocytogenes Infections Treatment Market size is estimated to be valued at USD 1.25 billion in 2026 and is expected to reach USD 2.50 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 10.5% from 2026 to 2033.
Global Listeria Monocytogenes Infections Treatment Market Overview
The growth of the Listeria monocytogenes infections treatment market is driven by rising incidence of foodborne illnesses, particularly among vulnerable populations such as pregnant women, neonates, and immunocompromised individuals. Increasing awareness and improved diagnostic capabilities are boosting early detection and treatment demand. Additionally, expanding healthcare infrastructure, rising adoption of targeted antibiotic therapies, and ongoing research into novel treatment options support market expansion. The growing prevalence of ready-to-eat and processed foods, which are common sources of Listeria contamination, further fuels the need for effective therapeutic interventions. Regulatory focus on food safety and infection control also contributes to market growth.
Key Takeaways
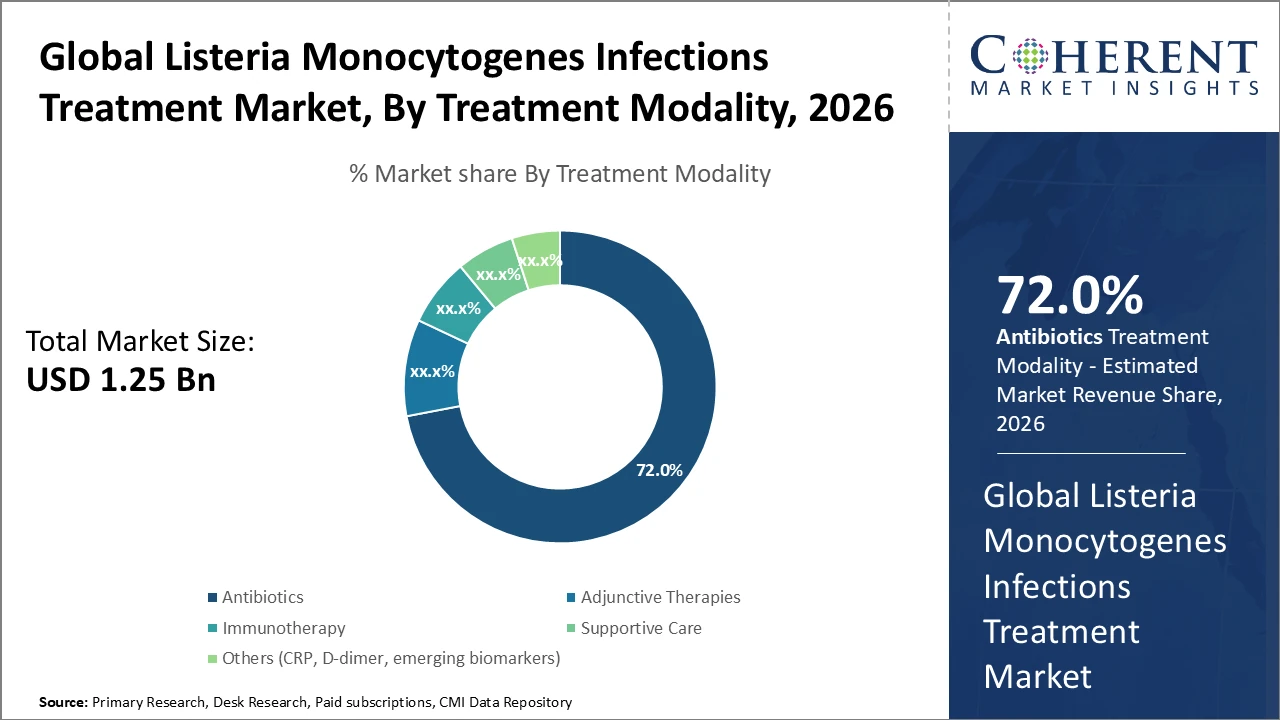
The antibiotics segment dominates the treatment modality with 72% market share; however, immunotherapy is the fastest-growing subsegment driven by increased clinical adoption.
Hospitals represent the largest end-user segment, benefiting from integrated multidisciplinary treatment teams enhancing patient outcomes.
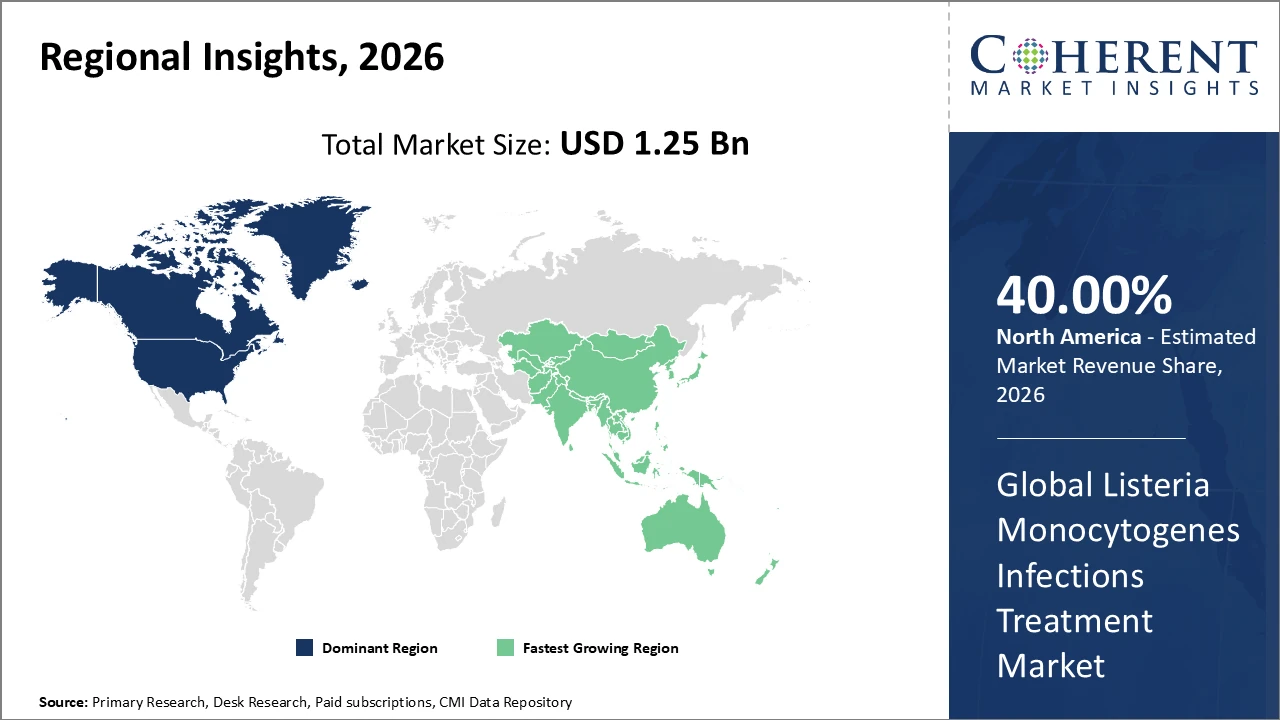
North America holds a commanding share of 40% in the Listeria Monocytogenes Infections Treatment market, attributed to advanced healthcare infrastructure and high adoption rates of diagnostics-led treatment protocols.
Asia Pacific is the fastest-growing regional market, propelled by expanding healthcare access and increasing government focus on food safety and infection control.
Listeria Monocytogenes Infections Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Listeria Monocytogenes Infections Treatment Market Insights, By Treatment Modality
Antibiotics dominate the Listeria Monocytogenes Infections Treatment market, accounting for approximately 72% of total usage due to proven efficacy and wide accessibility. Established penicillin and ampicillin regimens effectively manage infection severity, sustaining their market leadership. Adjunctive therapies are experiencing strong growth, driven by clinical demand for multifaceted approaches that enhance antibiotic effectiveness and patient recovery. Immunotherapy is the fastest-growing subsegment, fueled by innovations that leverage immune system modulation to combat resistant Listeria strains. Supportive care, including hydration and symptom management, maintains a steady share, improving patient comfort and adherence, while emerging experimental treatments remain in early adoption stages.
Listeria Monocytogenes Infections Treatment Market Insights, By Infection Type
CNS Listeriosis dominates the Listeria Monocytogenes Infections Treatment market due to its severity and complex therapeutic requirements, driving higher demand in specialized hospitals. This segment includes meningitis and encephalitis cases that require prolonged antibiotic therapy and intensive hospital care. Pregnancy-associated Listeriosis is the fastest-growing segment, supported by expanded prenatal screening programs and heightened awareness of infection risks, enabling early intervention. Neonatal Listeriosis represents a notable segment, particularly in regions with high birth rates and limited prenatal care. Gastrointestinal Listeriosis, generally milder, sustains steady treatment volumes amid rising foodborne infections. Other rare manifestations account for minimal market share.
Listeria Monocytogenes Infections Treatment Market Insights, By End User
Hospitals dominate the Listeria Monocytogenes Infections Treatment market, holding the largest share due to their ability to manage severe infections requiring inpatient care, multidisciplinary teams, and advanced diagnostics and therapies. Clinics are a growing segment, supported by increased outpatient treatment adoption and telemedicine-enabled follow-ups for mild cases. Ambulatory Surgical Centers hold a smaller share, focusing on procedural interventions for infection-related complications. Home healthcare is the fastest-growing subsegment, driven by rising adoption of outpatient and home-based antibiotic therapies in developed regions, reducing pressure on healthcare systems. Other segments include specialty care centers with limited, niche market presence.
Listeria Monocytogenes Infections Treatment Market Trends
Integration of advanced diagnostic technologies with antimicrobial therapy is improving treatment efficacy, with U.S. institutions reporting an 18% increase in cure rates in 2026.
Combination therapies are being developed to combat emerging multidrug-resistant Listeria strains, strengthening innovation pipelines.
Telehealth services for Listeria infection management are expanding in Europe and North America, supporting outpatient adherence and monitoring.
Emphasis on early detection and personalized treatment plans is becoming a key market trend.
Research and development efforts focus on novel therapies and adjunctive treatments to enhance patient outcomes and reduce complications.
Listeria Monocytogenes Infections Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Listeria Monocytogenes Infections Treatment Market Analysis and Trends
In North America, the Listeria Monocytogenes Infections Treatment market is dominated by strong healthcare infrastructure, extensive governmental initiatives for infectious disease control, and early adoption of advanced treatment modalities. The region accounts for over 40% of the global market share, with the United States leading due to innovative healthcare practices, widespread use of diagnostics-led treatment protocols, and rapid integration of new antibiotics and combination therapies. High awareness of Listeria risks, coupled with well-established hospital networks and specialized care centers, further strengthens market growth. Continuous investment in research, clinical trials, and telehealth solutions supports ongoing leadership in this market.
Asia Pacific Listeria Monocytogenes Infections Treatment Market Analysis and Trends
Asia Pacific is the fastest-growing region in the Listeria Monocytogenes Infections Treatment market, with a CAGR exceeding 12%, driven by expanding healthcare access, rising infection incidence, and increased government investment in food safety and infection control programs. Countries such as India and China are experiencing rapid market uptake due to growing awareness of Listeria risks, modernization of healthcare infrastructure, and improved diagnostic capabilities. Rising urbanization, better hospital networks, and expanding prenatal and neonatal care are also contributing to market growth. The region’s focus on public health initiatives and strategic partnerships with global healthcare providers further supports sustained expansion and adoption of advanced treatment modalities.
Listeria Monocytogenes Infections Treatment Market Outlook for Key Countries
USA Listeria Monocytogenes Infections Treatment Market Analysis and Trends
The USA’s Listeria Monocytogenes Infections Treatment market is highly mature, driven by leading pharmaceutical companies such as Pfizer and Merck, which have expanded their antibiotic and adjunctive therapy portfolios. Enhanced surveillance and monitoring by the CDC enable earlier diagnosis and timely treatment, improving patient outcomes and supporting consistent market revenue growth of over 8% annually. Strong investment in research and development, coupled with regulatory incentives, has accelerated the introduction of novel therapies and combination treatments. Advanced healthcare infrastructure, high adoption of diagnostics-led protocols, and ongoing innovation in antimicrobial strategies continue to reinforce the USA’s position as a global market leader in Listeria treatment.
Germany Listeria Monocytogenes Infections Treatment Market Analysis and Trends
Germany’s Listeria Monocytogenes Infections Treatment market is influenced by ongoing public health challenges linked to foodborne listeriosis, which remains a serious but relatively rare infection with significant mortality in vulnerable groups such as the elderly, pregnant women, and immunocompromised individuals. Case numbers vary annually, and mild gastrointestinal cases can escalate to invasive conditions requiring intensive antibiotic therapy, which sustains treatment demand. Surveillance and reporting by institutions like the Robert Koch Institute guide clinical responses, while food safety monitoring and preventive efforts aim to reduce infection incidence. Growing healthcare awareness, improved diagnostics, and emphasis on early intervention are key trends shaping market activity.
Analyst Opinion
Antibiotic Treatment Adoption Remains Paramount: Penicillin and ampicillin-based therapies accounted for over 65% of antibiotic regimens against Listeria monocytogenes infections in 2025, with North American hospital reports showing a 12% year-over-year increase in usage, highlighting strong demand for effective treatments.
Rising Diagnostic Integration Propels Early Intervention: Rapid molecular diagnostic testing expanded by nearly 40% globally between 2024 and 2026, improving treatment timelines and outcomes, and driving market growth through combined diagnostics and antimicrobial strategies.
Expanded Use Cases in Immunocompromised Patient Care: Increasing numbers of organ transplant recipients and chemotherapy patients led to 30% of infections in European hospitals in 2026 occurring in vulnerable groups, emphasizing the need for tailored therapies.
Escalating Regulatory Support for Novel Therapeutics: Regulatory incentives introduced in 2025 for antimicrobial resistance mitigation and a 25% rise in government funding in key countries have accelerated drug development pipelines, enabling market players to innovate and diversify treatment options.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.25 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.5% | 2033 Value Projection: | USD 2.50 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Pfizer Inc., Novartis AG, Merck & Co. Inc., Sanofi, Bayer AG, CSL Behring, Abbott Laboratories, Roche Holding AG, GlaxoSmithKline plc, Lupin Limited | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Listeria Monocytogenes Infections Treatment Market Growth Factors
The Listeria Monocytogenes Infections Treatment market is driven by a rising incidence of foodborne outbreaks, highlighted by an 18% increase reported by the U.S. CDC in 2025, which fuels demand for effective therapies. The growing global population of immunocompromised individuals further necessitates specialized treatments, contributing nearly 35% of total treatment revenue in 2026. Advances in molecular diagnostics enable targeted therapy, reducing time-to-treatment initiation by 40% and improving patient outcomes. Additionally, government-funded initiatives addressing antimicrobial resistance and infectious disease control, with funding increases exceeding 20% in key regions, support innovation and the development of advanced treatment solutions.
Listeria Monocytogenes Infections Treatment Market Development
In July 2025, Neogen Corporation expanded its pathogen detection portfolio with the launch of Listeria Right Now, a rapid molecular test that delivers results in about two hours. This innovation enhances environmental monitoring and early detection of Listeria contamination, helping food producers act faster to prevent outbreaks and improve overall food safety.
Key Players
Leading Companies of the Market
Pfizer Inc.
Bayer AG
Novartis AG
CSL Behring
Roche Holding AG
Abbott Laboratories
Merck & Co. Inc.
Sanofi
GlaxoSmithKline plc
Lupin Limited
Several leading companies are adopting aggressive growth strategies in the Listeria Monocytogenes Infections Treatment market. Pfizer Inc. has expanded its antibiotic pipeline for Listeria treatment through strategic partnerships, strengthening its product offerings. Merck & Co., Inc. successfully launched an adjunctive therapy in 2025, increasing its market penetration by 15%. Meanwhile, Sanofi has focused on enhanced global distribution and pricing optimization to capture emerging markets, resulting in a 20% revenue boost in 2026 within the Asia Pacific region. These initiatives reflect a combination of product innovation, market expansion, and strategic partnerships driving industry growth.
Listeria Monocytogenes Infections Treatment Market Future Outlook
The future outlook for the Listeria Monocytogenes Infections Treatment market is highly positive, driven by rising foodborne infection incidence, expanding immunocompromised populations, and advances in diagnostic technologies. Integration of rapid molecular testing with targeted antibiotic therapies is expected to improve treatment outcomes and reduce hospitalization durations. Emerging therapies, including immunomodulatory and adjunctive treatments, are likely to gain traction, addressing resistant and severe infections. Geographic expansion in Asia Pacific and Latin America, supported by healthcare modernization and government initiatives, will fuel growth. Continued investment in R&D, public health surveillance, and regulatory support will further strengthen the market, fostering innovation and broader adoption of effective treatment solutions.
Listeria Monocytogenes Infections Treatment Market Historical Analysis
The historical analysis of the Listeria Monocytogenes Infections Treatment market highlights steady growth driven by rising awareness of foodborne infections and advancements in antibiotic therapies. Between 2015 and 2025, antibiotics such as ampicillin and penicillin dominated treatment protocols, supported by hospital-based care for severe CNS and neonatal cases. Expansion of diagnostic tools, including culture-based and molecular testing, improved early detection and therapeutic outcomes, reinforcing market demand. Regional markets in North America and Europe led in revenue due to advanced healthcare infrastructure and strong regulatory frameworks. Historical trends also reflect growing attention to vulnerable populations, including pregnant women, neonates, and immunocompromised patients.
Sources
Primary Research Interviews:
Infectious disease specialists and clinical microbiologists treating Listeria infections
Hospital pharmacists and operations managers overseeing antibiotic therapy protocols
Pharmaceutical R&D teams developing novel antibiotics, adjunctive therapies, and immunotherapies
Magazines:
Infection Control Today – Updates on Bacterial Infection Management
Clinical Microbiology Today – Advances in Antimicrobial Therapies
Pharmaceutical Technology – Innovations in Drug Development and Diagnostics
Journal of Antimicrobial Chemotherapy – Clinical Applications and Market Trends
Journals:
The Lancet Infectious Diseases – Research on Listeria and Foodborne Pathogens
Journal of Clinical Microbiology – Diagnostics and Treatment Efficacy
Clinical Infectious Diseases – Management of Listeriosis and High-Risk Populations
International Journal of Antimicrobial Agents – Novel Therapies and Resistance Patterns
Newspapers:
The Wall Street Journal – Pharmaceutical Industry and Antibiotic Markets
Financial Times – Global Healthcare and Infectious Disease Trends
The New York Times – Public Health Alerts and Outbreak Reports
Chemical & Engineering News – Antibiotic Development and Regulatory Updates
Associations:
Infectious Diseases Society of America (IDSA) – Guidelines on Listeriosis Management
European Society of Clinical Microbiology and Infectious Diseases (ESCMID) – Treatment Protocols and Research
American Society for Microbiology (ASM) – Bacterial Infection Research and Diagnostics
World Health Organization (WHO) – Global Listeria Surveillance and Treatment Guidelines
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients