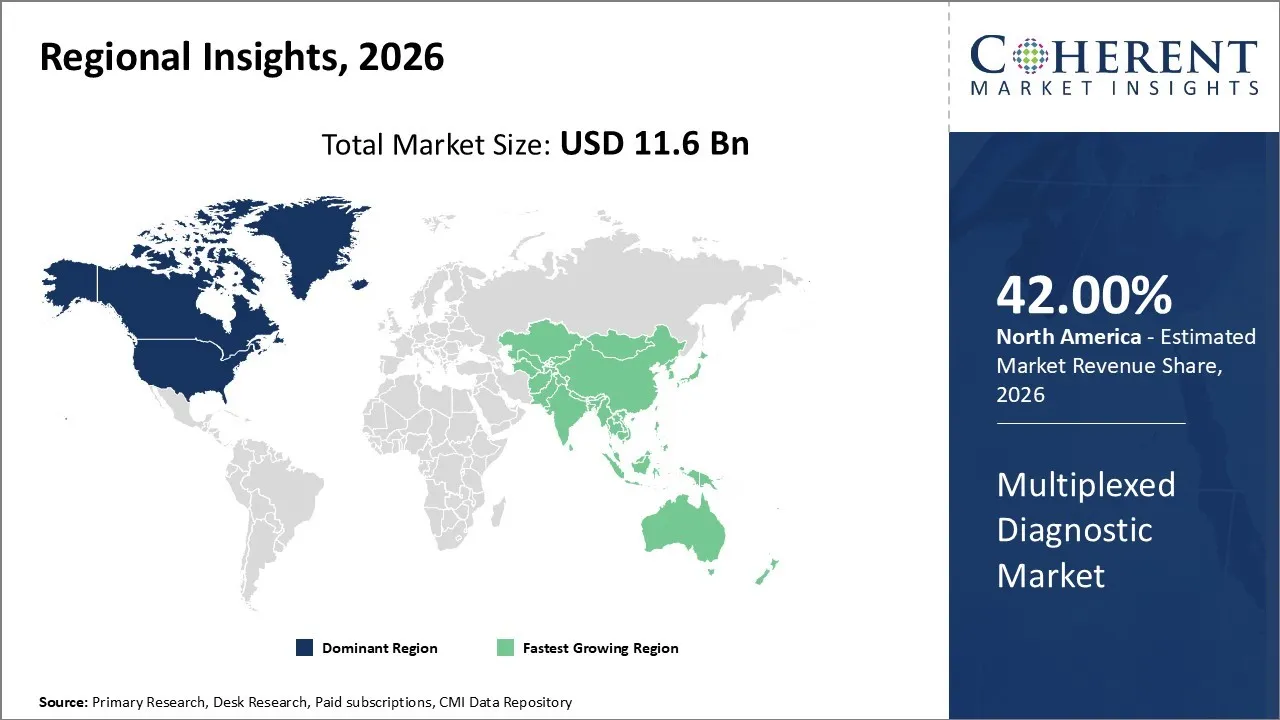
The multiplexed diagnostic market was valued at USD 11.6 Bn in 2026 and is forecast to reach a value of USD 16.08 Bn by 2033 at a CAGR of 5.1% between 2026 and 2033.
Multiplexed diagnostics are used for analysis of biological samples, which are capable of completing a screening process with a single run. Multiplexed diagnostic assays are involved in the detection and diagnosis of different diseases and infections, which include cancer, autoimmune diseases, allergies, and cardiovascular diseases. Assays performed in multiplex diagnostics include high density multiplexed assays, medium density multiplexed assays, low density multiplexed assays, and next-generation sequencing assays. Early diagnosis can minimize the risk factors associated with chronic diseases/infection and helps in proper treatment management.
|
Current Event |
Description and its Impact |
|
Launch of Advanced Multiplex Assays for Infectious Diseases |
|
|
Regulatory Approvals along with Market Expansion in APAC |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the reagents & consumables segment is expected to lead the market with 65% share in 2026, owing to their indispensable role in the functioning as well as efficiency of multiplexed diagnostic assays. These reagents and consumables include various biochemical substances such as antibodies, primers, probes, dyes, enzymes, buffers, etc., which are crucial for enabling simultaneous detection of multiple biomarkers in a single test run. Their ubiquitous usage across diverse applications such as infectious disease diagnostics, genetic testing, oncology, as well as personalized medicine underscores their dominance.
Also, as multiplexed tests are used more for studying complex diseases, there is higher demand for special reagents and consumables that make tests more accurate and able to check many targets at once. For instance, advanced fluorophores and magnetic beads are used in bead-based multiplex immunoassays to differentiate numerous analytes within a single sample.
In terms of application, the disease diagnostics segment is expected to hold 55% share of the market in 2026. Primarily attributable to its high adoption across various healthcare settings due to the escalating need for rapid, accurate, as well as multiplexed detection of multiple pathogens or biomarkers associated with a broad spectrum of diseases. Multiplexed diagnostic tests are very useful for infectious diseases like tuberculosis, HIV, and hepatitis, because they can check for many germs at the same time and help doctors make better decisions.
For instance, the World Health Organization supports using multiplexed tests for tuberculosis in areas with few resources. These tests find the disease faster and check for more problems using only a small sample, which helps people who have limited access to healthcare.
In terms of end user, the hospital segment is projected to account for 45% share of the market in 2026. Owing to their significant role as primary healthcare providers and their critical need for rapid and accurate diagnostic tools to manage complex patient cases. Hospitals, especially large tertiary and quaternary care centers, require multiplexed diagnostic platforms to simultaneously test for multiple pathogens or biomarkers, which significantly enhances diagnostic efficiency and supports critical decision-making in patient management.
To learn more about this report, Request Free Sample
North America is expected to dominate the multiplexed diagnostic market with 42.0% share in 2026, owing to several key factors that underscore its dominance. Primarily, North America, and especially the United States, is home to a highly developed healthcare infrastructure equipped with advanced diagnostic laboratories, cutting-edge research institutions, as well as a significant presence of multinational diagnostic companies.
For instance, in November 2025, Co-Diagnostics, Inc. (Co-Dx), a company that makes molecular diagnostic tests, announced it has started clinical testing for its Co-Dx PCR Flu A/B, RSV Test Kit. The tests will help the company apply to the U.S. FDA for approval of this multiplex point-of-care test and the Co-Dx PCR Pro instrument.
Asia Pacific is expected to exhibit the fastest growth, owing to rapidly increasing investments in healthcare infrastructure, rising prevalence of chronic diseases, and growing government initiatives to enhance diagnostic capabilities at the population level. Countries like China and India are aggressively scaling up their laboratory networks to meet the increasing demand for timely and accurate diagnosis of complex diseases such as infectious diseases, cancer, and autoimmune disorders, which multiplexed diagnostics are well-suited to address.
For instance, in October 2025, Mirxes, a top molecular cancer detection company in Asia Pacific, said its main product, GASTROClear, got NMPA approval in China. It is now the first non-invasive blood test for gastric cancer screening, marking a major step in the molecular diagnostics market in China.
The U.S. contributes the highest share in the multiplexed diagnostic market within the region, primarily due to its advanced healthcare infrastructure, significant R&D investments, as well as a high adoption of cutting-edge diagnostic technologies. In the U.S., healthcare focuses on precise and personalized testing. This increases demand for multiplexed diagnostics, which can check many markers at once and make disease diagnosis and monitoring faster and more accurate.
Additionally, the U.S. Food and Drug Administration’s (FDA) highly streamlined regulatory approvals for multiplex diagnostic assays foster quicker market entry as well as expansion for innovative products. The presence of numerous leading diagnostic companies headquartered in the U.S., coupled with high healthcare expenditure per capita and vast reimbursement infrastructure, further consolidates the U.S. as the leading subsegment in North America’s multiplexed diagnostics landscape.
China contributes the highest share in the multiplexed diagnostic market due to its extensive investments in healthcare infrastructure, rapid technological adoption, as well as government-driven initiatives focused on advancing precision medicine. The country’s healthcare system has witnessed significant modernization, particularly with the integration of multiplexed diagnostics into clinical workflows.
For instance, China’s National Health Commission is encouraging the use of advanced tests to find diseases like cancer and infections early and to give patients the right treatment.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 11.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.1% | 2033 Value Projection: | USD 16.08 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Luminex Corporation, Thermo Fisher, Illumine Inc., Bio-Rad Laboratories, Inc., Qiagen N.V., Abbott Laboratories, Siemens Healthineers, Agilent technologies, BioMerieux SA, and F.Hoffmann-La Roche Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The high prevalence of infectious diseases globally is a significant driver propelling the expansion of the multiplexed diagnostic market. Infectious diseases such as tuberculosis, HIV, hepatitis, etc., present complex diagnostic challenges due to their variable etiology along with the need for rapid, accurate differentiation between pathogens. Multiplexed diagnostic technologies, which allow for the simultaneous detection of multiple pathogens or biomarkers in a single test, offer considerable advantages over traditional single-target assays.
The increasing demand for rapid and accurate diagnostics is a pivotal factor driving the expansion of the multiplexed diagnostic market. Multiplexed diagnostics allow simultaneous detection of multiple biomarkers, pathogens, or genetic targets in a single test, significantly reducing the time and resources needed compared to traditional single-analyte tests. This capability is particularly crucial in clinical settings where timely diagnosis directly impacts patient outcomes, such as infectious disease outbreaks or complex chronic conditions requiring multi-faceted evaluation.
Integration with point-of-care testing (POCT) devices stands out as a transformative opportunity within the multiplexed diagnostic market, primarily because it addresses critical demands for rapid, precise, and comprehensive diagnostic information at the site of patient care. Multiplexed diagnostics, which allow simultaneous detection of multiple analytes from a single sample, synergize exceptionally well with POCT platforms to provide timely clinical insights that can significantly influence treatment decisions.
The multiplexed diagnostic market is experiencing significant momentum, driven primarily by a high demand for rapid, accurate, as well as cost-effective diagnostic solutions. The surge in prevalence of chronic and infectious diseases has underscored the need for simultaneous testing of multiple biomarkers, enhancing diagnostic efficiency while reducing time as well as resource expenditure.
Advances in molecular diagnostics as well as the integration of novel technologies such as microfluidics, biosensors, etc., are further propelling market growth. However, high costs associated with multiplex platforms as well as stringent regulatory frameworks remain major restraints that could potentially slow widespread adoption.
North America continues to dominate this market, attributable to robust healthcare infrastructure, major R&D investments, and early adoption of innovative technologies. Conversely, the Asia Pacific region is emerging as the fastest-growing market, fueled by increasing healthcare awareness, expanding diagnostics infrastructure, and growing government initiatives to improve healthcare accessibility. Additionally, rising investments in personalized medicine and point-of-care testing present substantial growth opportunities for market participants.
The multiplexed diagnostics segment provides an invaluable tool for improving patient outcomes by enabling comprehensive disease profiling and supporting precision medicine approaches. Nonetheless, addressing challenges such as assay complexity and the need for skilled personnel will be critical for broader market penetration.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients