The global neurofibromatosis treatment drugs market size is projected to hit around USD 20,785 Mn by 2026 and is expected to exhibit a CAGR of 13.4% during the forecast period (2026–2033), surpassing USD 49,085 Mn by 2033. Some other factors that are further expected to drive growth include the introduction of novel targeted treatments, such as MEK inhibitors, driven by orphan drug approval and research programs from the FDA, and increased investments in rare disease drugs.
Neurofibromatosis 1 (NF1) is estimated to occupy the largest market share among drugs for neurofibromatosis treatments by 2026, comprising around 72% of the overall market size. The reason behind this market dominance can be attributed to the high incidence rate of NF1 in the global population. According to National Institutes of Health (NIH)-supported clinical literature, NF1 affects approximately 1 in 3,000–4,000 individuals globally, making it the largest patient pool among all neurofibromatosis subtypes.
High prevalence rate will increase the demand for treatment, specifically due to complications like plexiform neurofibromas, which need ongoing treatment and pharmacotherapy. NF1 represents the largest revenue-generating segment within the neurofibromatosis treatment market.
In February 2025, the U.S. Food and Drug Administration approved Mirdametinib, developed by SpringWorks Therapeutics, for the treatment of adult and pediatric patients (aged 2 years and older) with neurofibromatosis type 1 (NF1) who have symptomatic plexiform neurofibromas that are not amenable to complete surgical resection.
To learn more about this report, Request Free Sample
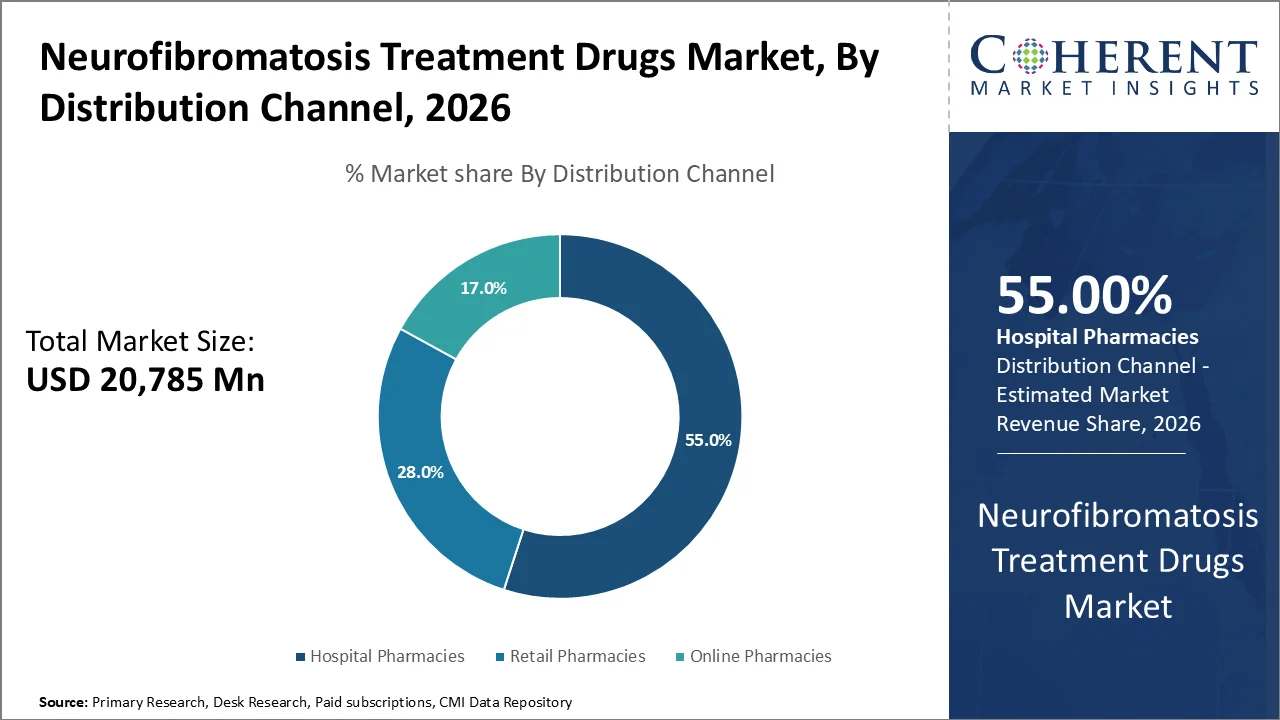
Hospital pharmacies are projected to account for the largest share of the neurofibromatosis treatment drugs market in 2026, representing approximately 55% of total distribution. This is because medications for neurofibromatosis are considered specialty and orphan drugs that need to be administered in the hospital setting.
As per the clinical care studies supported by NIH, NF1 and similar tumor disorders are generally treated through specialist clinics available at hospitals owing to the need for specialized treatment, genetics testing, and safety monitoring. Such treatments usually involve dosage control, adverse effect monitoring, and physician prescription, which is why hospital pharmacies are the best means of drug delivery.
In November 2025, the FDA expanded approval of selumetinib (Koselugo) to include adult NF1 patients with symptomatic, inoperable plexiform neurofibromas.
The approval of targeted treatments like MEK inhibitors (such as selumetinib) has marked a significant change in the management of NF1. The US FDA granted the approval to selumetinib (Koselugo), the first drug that targets plexiform neurofibromas in children with NF1 and are inoperable. Research showed that 66% of patients experienced tumor shrinkage by at least 20%.
In May 2024, the U.S. Food and Drug Administration (FDA) expanded the approval of selumetinib (Koselugo) to include adult patients with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas (PN).
|
Current Event |
Description and its Impact |
|
2025 FDA Expansion of Selumetinib (Koselugo) to Adult NF1 Patients |
|
|
2025 NIH Expansion of Rare Diseases Clinical Research Network (RDCRN) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
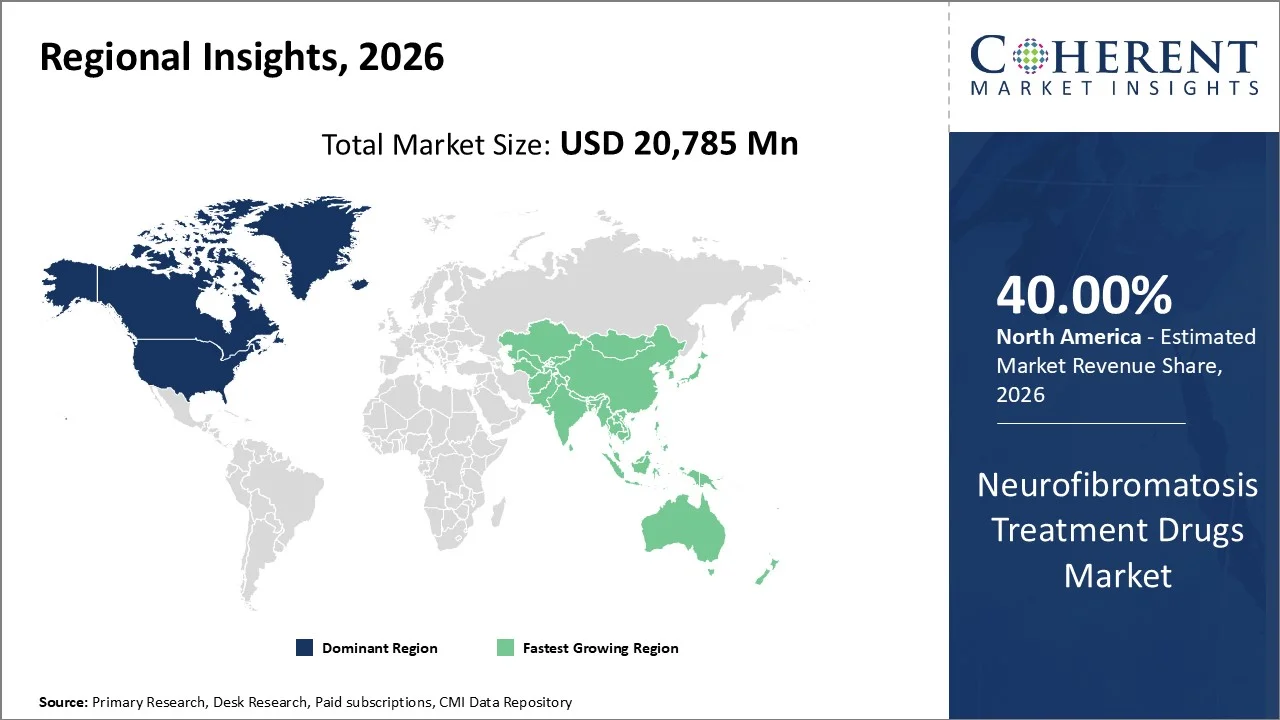
The Neurofibromatosis Treatment Drugs Market in North America is projected to contribute to about 40% of the market share in 2026. This projection will be fueled by early genetic screening, disease awareness, and well-established healthcare systems. The region enjoys the advantage of having readily available molecular diagnosis that facilitates early detection of NF1 and NF2 cases, leading to treatment adherence. From the clinical epidemiological studies conducted by the NIH and NCBI, it is observed that the United States of America records the highest number of diagnosed Neurofibromatosis cases globally.
In November 2025, AstraZeneca announced an update regarding Koselugo (selumetinib), highlighting continued strong market performance and expanded adoption in neurofibromatosis type 1 (NF1) treatment.
The Asia-Pacific region has emerged as one of the fastest growing markets for Neuropathy drugs because of advanced diagnosis, increased understanding about genetic diseases, and improved healthcare infrastructures in developed economies like China, India, and Japan. On the other hand, it accounts for a relatively lower share than that of North America owing to underdiagnosis and lack of genetic testing facilities in certain developing nations within the region. According to studies conducted by the National Institutes of Health (NIH), NCBI (National Center for Biotechnology Information), NF remains underdiagnosed in parts of the region in the Asia-Pacific region because of lack of new-born screening programs.
The U.S. industry for Neurofibromatosis therapy drugs is witnessing considerable growth owing to increased awareness about the condition, availability of better diagnostic procedures, and availability of state-of-the-art facilities for researching rare disorders through NIH funding. The United States houses the largest number of individuals diagnosed with NF1 and NF2 globally owing to the large availability of molecular diagnostics equipment.
In November 2025, the U.S. FDA expanded Koselugo approval to include adult NF1 patients with symptomatic, inoperable plexiform neurofibromas, marking a major milestone in extending treatment beyond pediatric use and strengthening long-term disease management strategies.
Japan's Neurofibromatosis treatment drugs market is likely to show rapid growth during the coming years owing to its sophisticated healthcare facilities along with a focus on treating rare disorders. The aging population of Japan and innovations in techniques of genetic analysis have enabled increased diagnosis of NF1, thereby leading to growth in treatments through pharmaceutical means.
Japan has emerged as an early adopter for precision oncology therapies, including MEK inhibitor-based treatments for NF1. The availability of therapies such as Koselugo in pediatric NF1 patients has improved treatment outcomes for plexiform neurofibromas and strengthened clinical adoption in specialized hospitals.
Some of the major key players in Neurofibromatosis Treatment Drugs are AstraZeneca, SpringWorks Therapeutics, Novartis, Pfizer, Takeda Pharmaceutical, Roche, Healx, NFlection Therapeutics, Fosun Pharmaceutical, and Recursion Pharmaceuticals.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 20,785 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.4% | 2033 Value Projection: | USD 49,085 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca, SpringWorks Therapeutics, Novartis, Pfizer, Takeda Pharmaceutical, Roche, Healx, NFlection Therapeutics, Fosun Pharmaceutical, and Recursion Pharmaceuticals. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients