Neuromodulation Devices Market is estimated to be valued at USD 21.59 Bn in 2026 and is expected to reach USD 58.85 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 15.4% from 2026 to 2033.
The neuromodulation devices market is experiencing robust growth in 2026, driven by rising demand for non-invasive treatments for chronic pain, neurological disorders, and movement impairments. Technological advancements in spinal cord and deep brain stimulation are expanding therapeutic options and improving patient outcomes. Increasing awareness, aging populations, and the need for opioid-free pain management are fueling neuromodulation devices market demand across hospitals and specialized clinics. As healthcare systems prioritize personalized and minimally invasive solutions, neuromodulation continues to gain traction as a transformative approach in modern medicine.
|
Current Event |
Description and its Impact |
|
FDA Breakthrough Device Designation Program Acceleration |
|
|
Artificial Intelligence Integration in Healthcare Regulation |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial intelligence (AI) is playing a transformative role in the neuromodulation devices market by enhancing personalization, precision, and clinical outcomes.
AI integration enables neuromodulation systems to adapt stimulation parameters in real time based on patient-specific neural responses, making therapies more effective and tailored. For instance, AI-powered closed-loop systems can monitor brain or nerve activity and automatically adjust electrical impulses to optimize treatment for conditions like chronic pain, Parkinson’s disease, and epilepsy. This dynamic feedback loop reduces trial-and-error programming and improves long-term efficacy. Additionally, AI algorithms assist in analyzing large datasets from neuromodulation devices, helping clinicians identify patterns, predict treatment responses, and refine protocols. Wearable neuromodulation technologies are also leveraging AI to deliver non-invasive, user-friendly solutions for movement disorders and mental health conditions. As regulatory approvals for AI-enhanced devices increase, the Neuromodulation Devices Market is expected to grow rapidly, driven by smarter, more responsive, and patient-centric innovations.
For instance, in July 2025, University of Minnesota spinout Neurotype received FDA clearance for its AI-powered wearable device to treat essential tremor. The non-invasive neuromodulation system uses artificial intelligence to monitor tremor patterns and deliver personalized stimulation. This marks a significant advancement in combining AI with neuromodulation, expanding treatment options for movement disorders and enhancing precision in neurotherapeutics.
In terms of device type, the spinal cord stimulation devices segment is expected lead the market with 50% share in 2026, due to their widespread use in managing chronic pain conditions. These devices deliver electrical impulses to the spinal cord, disrupting pain signals before they reach the brain. Their popularity stems from their effectiveness in treating conditions like failed back surgery syndrome, complex regional pain syndrome, and neuropathic pain. Technological advancements, such as high-frequency stimulation and closed-loop systems, have further enhanced their efficacy and patient satisfaction. Additionally, favorable reimbursement policies and increasing awareness among healthcare providers have contributed to their dominance in this segment.
For instance, in July 2025, NeuroSigma unveiled its novel sensing technology for closed-loop spinal cord stimulation (SCS) systems. This innovation enables real-time monitoring and adaptive stimulation based on spinal cord activity, aiming to enhance chronic pain management. The system represents a major advancement in personalized neuromodulation therapy.
To learn more about this report, Request Free Sample
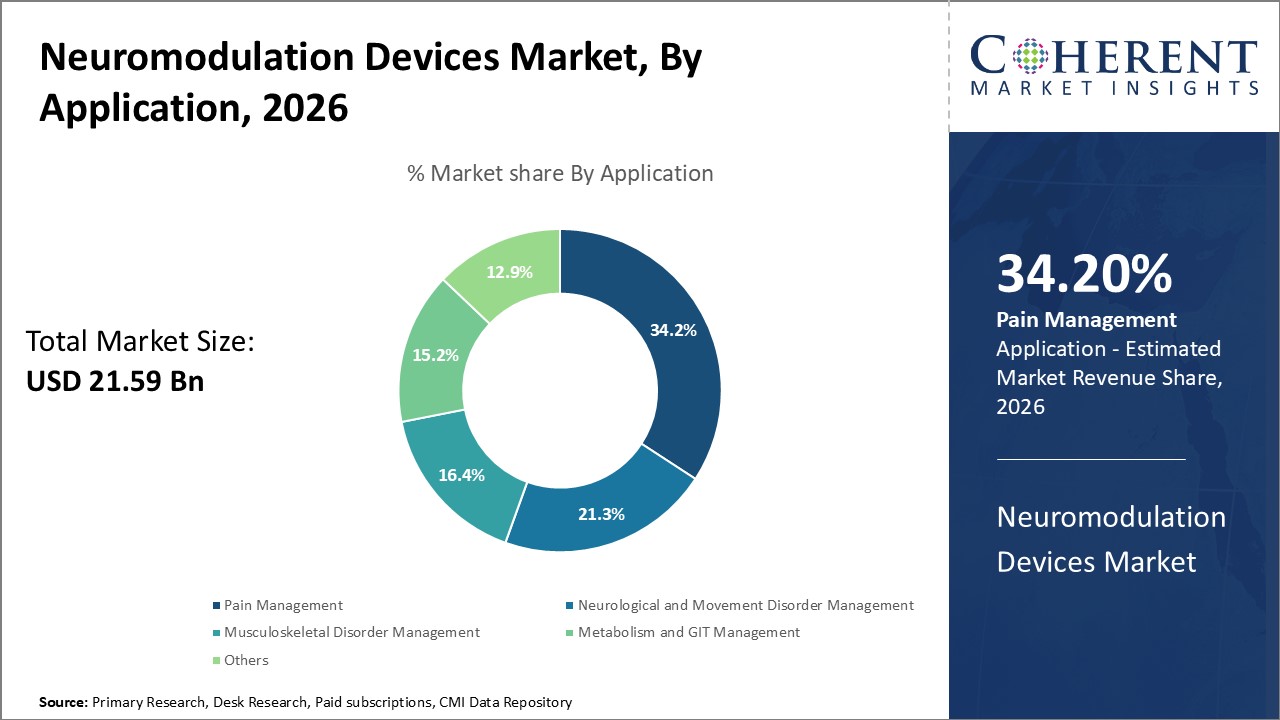
In terms of application, the pain management segment is expected to hold the largest share of the market in 2026. With chronic pain affecting a significant portion of the global population, there is a growing demand for non-opioid, long-term treatment solutions. Neuromodulation offers a minimally invasive alternative that can significantly improve quality of life for patients suffering from persistent pain. The opioid crisis has also accelerated the adoption of these devices, as healthcare systems seek safer and more sustainable approaches to pain relief. As a result, pain management continues to be the primary driver of growth in the neuromodulation market.
For instance, in June 2025, Boston Scientific received FDA approval for its W4 and D4 lead splitters, enhancing the Precision Plus™ spinal cord stimulation (SCS) system. These splitters enable multi-site lead placement, offering physicians greater flexibility in targeting pain areas and improving outcomes for patients undergoing neuromodulation-based chronic pain management.
In terms of end-user, the hospitals segment is projected to account for the highest share of the market in 2026, due to their comprehensive infrastructure and multidisciplinary expertise. These institutions are equipped to handle complex procedures such as spinal cord and deep brain stimulation, which require specialized surgical skills and post-operative care. Hospitals also serve as referral centers for patients with chronic pain and neurological disorders, making them the first point of contact for neuromodulation therapies. Their role in clinical trials, research, and training further reinforces their central position in the market.
For instance, in August 2025, AIIMS Bhubaneswar launched a Centre for Neuromodulation Services under its Psychiatry Department, offering advanced therapies like rTMS, ultra-brief pulse ECT, and TES. These non-invasive treatments target drug-resistant psychiatric disorders, enhancing precision and patient acceptance. The initiative aims to improve mental health care and research across eastern India.
To learn more about this report, Request Free Sample
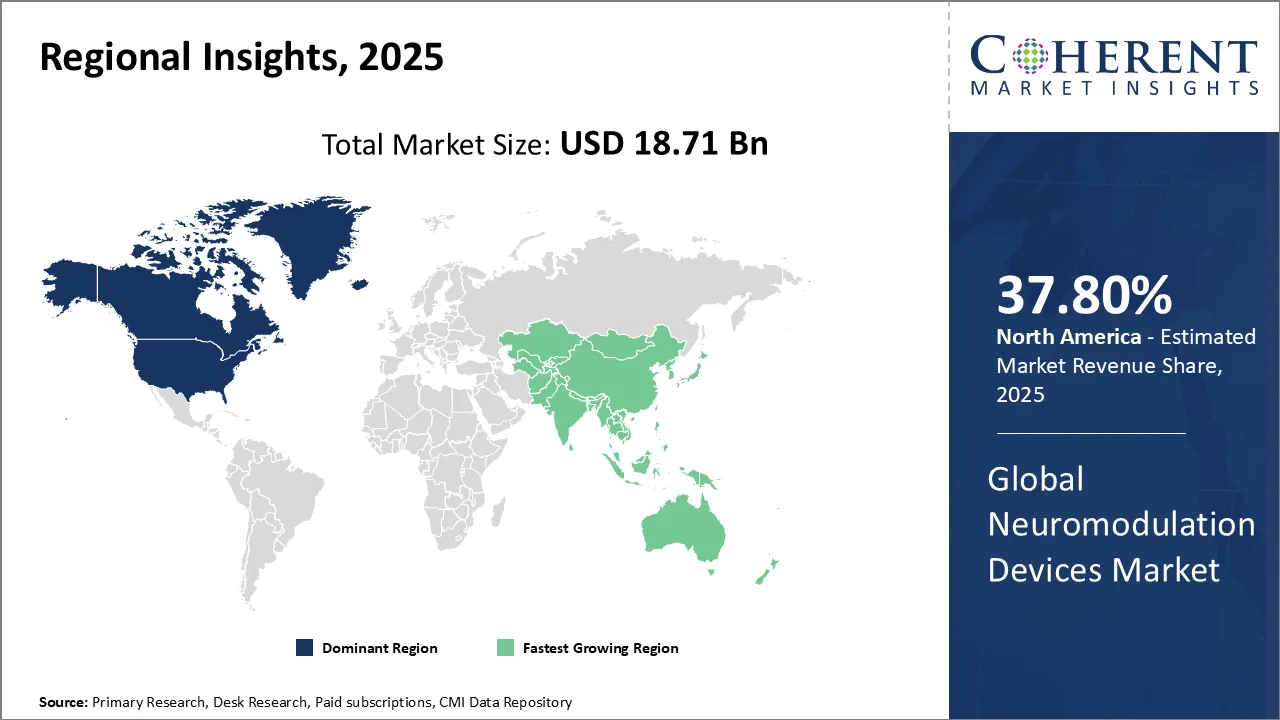
North America, holding a share of 37.8% share, is expected to dominate the neuromodulation devices market in 2026, due to rising neurological disorders, advanced healthcare infrastructure, and strong reimbursement policies. High adoption of innovative therapies like spinal cord and deep brain stimulation, coupled with robust R&D investments and presence of key industry players, further fuels market demand across the region.
For instance, in October 2026, US-based Ampa secured $8.5 million in funding to launch its FDA-cleared neuromodulation device, Ampa One, for treating major depressive disorder. The portable TMS system offers a single-day therapy protocol, aiming to improve accessibility and efficiency in mental health care. The round was led by Nexus NeuroTech Ventures, boosting Ampa’s total funding to $18 million.
Asia Pacific is projected to show the fastest growth in the neuromodulation devices market, due to rising neurological disorders, expanding healthcare infrastructure, and increasing awareness of advanced therapies. Government support, growing medical tourism, and demand for minimally invasive treatments further drive adoption. Countries like China and India are key contributors to the region’s rapid market growth.
For instance, in October 2026, Australis Scientific, based in Australia, won the 2026 MedTech Innovator Asia Pacific Grand Prize for its smart neuromodulation patch targeting overactive bladder. The wearable device uses AI-driven bioelectronic therapy to deliver personalized, non-invasive nerve stimulation. This innovation offers a drug-free alternative for millions affected by bladder dysfunction across the Asia-Pacific region.
The demand for neuromodulation devices in the U.S. is driven by rising cases of neurological and psychiatric disorders, growing preference for non-invasive treatments, and advancements in brain stimulation technologies. Increased awareness, supportive healthcare infrastructure, and expanding applications in pain management, depression, and bladder control are fueling adoption across clinical and outpatient settings.
For instance, in November 2026, Cefaly become the top neuromodulation device prescribed by Veterans Health Administration providers in the U.S. The FDA-cleared wearable targets the trigeminal nerve to treat migraines non-invasively. This milestone highlights growing trust in drug-free migraine therapies and marks a significant step in expanding neuromodulation use within veteran healthcare settings.
China’s neuromodulation devices market is growing due to rising neurological disorders, increased demand for non-invasive treatments, and expanding healthcare infrastructure. Technological advancements and government support for medical innovation are accelerating adoption. Applications in chronic pain, Parkinson’s disease, and epilepsy are driving clinical interest, making neuromodulation a key focus in China’s evolving healthcare landscape.
For instance, in August 2026, China approved its first REN-based wearable neuromodulation device for migraine treatment, offering a drug-free alternative to over 130 million patients. The device uses remote electrical stimulation to modulate pain pathways non-invasively. This regulatory milestone marks a major step in expanding innovative neuromodulation therapies within China’s healthcare system.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.59 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 15.4% | 2033 Value Projection: | USD 58.85 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, BD, LivaNova, Axonics, Inc., ShiraTronics, Abbott, Bioventus, Neuronetics, NEVRO CORP, Integer Holdings Corporation, BIOTRONIK, NeuroPace, Inc., NeuroMetrix, Inc., Helius Medical Technologies, Inc., and Synapse Biomedical Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing distribution agreements among key market players are significantly boosting the neuromodulation devices market size. These strategic collaborations enhance product accessibility, streamline supply chains, and expand geographic reach, especially in emerging healthcare markets. By partnering with regional distributors and healthcare providers, manufacturers can accelerate device adoption for conditions like chronic pain, depression, and epilepsy. Such agreements also facilitate faster regulatory approvals and localized marketing efforts, driving competitive advantage. As demand for non-invasive and personalized therapies grows, these partnerships are crucial in meeting clinical needs and scaling innovation. Ultimately, they play a pivotal role in shaping the future of neuromodulation technologies globally.
The neuromodulation devices market is witnessing robust growth driven by the increasing adoption of organic strategies, particularly acquisitions by key industry players. These strategic moves enable companies to expand their product portfolios, enhance technological capabilities, and penetrate untapped markets. By acquiring innovative startups or complementary businesses, established firms strengthen their competitive edge and accelerate product development cycles. This trend reflects a broader shift toward consolidation and innovation, fostering neuromodulation devices market growth. As neurological disorders rise globally, such acquisitions ensure a diversified and advanced range of solutions, positioning companies to meet evolving patient needs and regulatory standards effectively.
The neuromodulation devices market forecast highlights significant growth potential driven by expanding mental health applications. Rising global cases of depression, anxiety, and PTSD are fueling demand for non-invasive neuromodulation therapies such as transcranial magnetic stimulation (TMS) and vagus nerve stimulation (VNS). These devices offer effective alternatives to traditional pharmaceuticals, minimizing side effects and enhancing patient compliance. As awareness and acceptance of neurotherapeutic interventions increase, healthcare providers are integrating these technologies into mainstream treatment protocols. This trend, coupled with ongoing innovation and favorable reimbursement policies, positions mental health as a key driver in the future expansion of the neuromodulation devices market.
The neuromodulation devices market value is entering a phase where clinical durability and health-economic validation outweigh mere innovation. While closed-loop and adaptive systems have advanced beyond prototypes, reducing overstimulation and improving patient comfort—their adoption will hinge on real-world performance and reduced revision rates. Evidence from large clinical reviews indicates explanation rates nearing 10%, primarily due to loss of efficacy and lead issues, underscoring the urgency for reliability-focused engineering.
Established modalities like spinal cord stimulation (SCS) and sacral neuromodulation dominate, together accounting for an estimated over 40% of total implant activity, supported by robust evidence and procedural familiarity. Yet, payer scrutiny is intensifying, with reimbursement increasingly tied to demonstrable reductions in revision surgeries and total cost of care.
Market incumbents are consolidating technologies, integrating peripheral and sacral stimulation to expand coverage and strengthen value propositions. The competitive edge will belong to manufacturers that generate strong real-world evidence linking device use to improved outcomes and cost savings.
Ultimately, market leadership will be defined not by feature innovation but by sustained clinical efficacy, procedural reliability, and payer alignment. Neuromodulation’s long-term winners will be those who convert technological sophistication into tangible, repeatable therapeutic and economic advantages.
Definition: Neuromodulation is the alteration of neuronal and synaptic properties by neurons. Neuromodulation is a term that broadly describes a field of pain medicine involving implantable and non-implantable technologies that act directly on nerves, electronically or pharmaceutically, to alter pain signals travelling to the brain in order to alleviate physical suffering. It is widely regarded as a solution for chronic pain, but it can be used to treat a variety of different conditions.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients