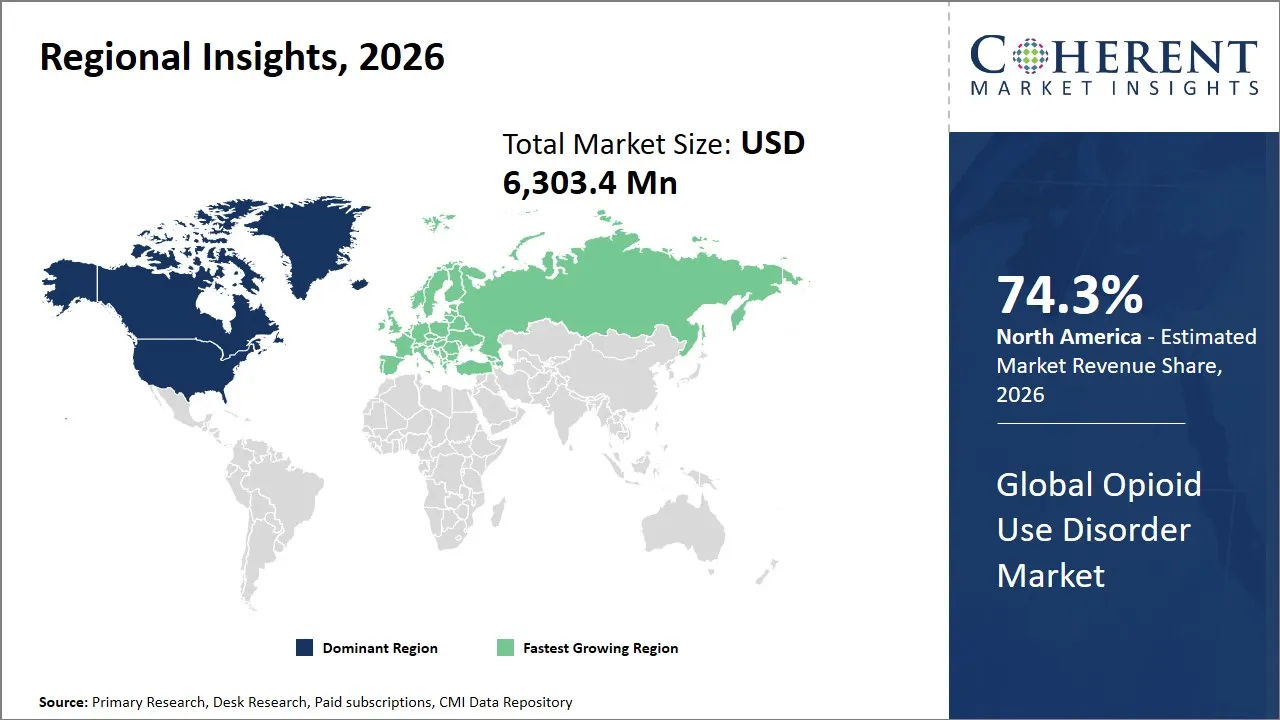
The global opioid use disorder market is estimated to be valued at USD 6,303.4 Mn in 2026 and is expected to reach USD 9,860.2 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.4% from 2026 to 2033. This significant growth reflects increasing awareness, advancements in treatment options, and rising prevalence of opioid use disorders worldwide, driving demand for innovative therapies and support services in this critical healthcare sector.
|
Current Events |
Description and its Impact |
|
Permanent U.S. telemedicine rule for buprenorphine prescribing |
|
|
Ongoing expansion of office-based buprenorphine prescribing after waiver removal |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The buprenorphine segment is expected to hold the largest market share of 37.0% in 2026, attributed primarily to its well-established safety profile and superior efficacy in managing opioid dependency. The pharmacological action of buprenorphine, a partial opioid agonist, is the reason why it is often preferred by healthcare providers because it lowers the risk of misuse and overdose, unlike full opioid agonists. This safety benefit is inherent and is a major driving factor behind its use as the first-line treatment of opioid use disorder (OUD).
The fact that the drug is able to greatly reduce the withdrawal symptoms and decrease the cravings indicates its usefulness in the process of long-term recovery. The flexibility in formulation of Buprenorphine, such as: oral, parenteral and others, also increases the access of patients to it and allows to facilitate individual treatment strategies. Also, its use has been enhanced by the regulatory support as well as prescription policies in different areas that have attracted its use compared to other opioid antagonists such as Naloxone and Naltrexone, which could have restricted clinical use.
For instance, in May 2024, the U.S. Food and Drug Administration approved Brixadi, a long-acting buprenorphine injection available in weekly and monthly formulations to treat moderate to severe opioid use disorder. The therapy is designed to provide sustained medication delivery and improve treatment adherence within comprehensive recovery programs.
(Source- U.S. Food and Drug Administration)
The parenteral segment is expected to hold 47.3% of the market share in 2026, due to its ability to provide rapid and consistent drug absorption while maintaining patient safety. Treatment of opioid use disorders must be conscientious in dosage and fast action, to adequately manage withdrawal syndrome and decrease relapses, and parenteral routes: including subcutaneous and intramuscular injections, provide these significant clinical attributes.
Parenteral administration of extended-release injections has acquired significant momentum since it allows a steady state of the drug concentrations to be established at a prolonged duration of time, minimizing dose potentials and increasing patient ease. It is especially important when it comes to the treatment of OUD, whereby it may be difficult to secure treatment adherence since the disorder is a chronic and relapsing condition. Continuity of treatment is improved because parenteral therapies bypass the problem of noncompliance in daily dosing that is observed with oral or sublingual methods of administration.
The injections segment is expected to hold the largest market share of 47.3% in 2026, because of its precision in dosing and capacity to improve patient compliance. Being a route of administration, injections are an effective and efficient way of administering therapeutic agents, which in many cases affect faster onset of action than oral or sublingual preparations. It has been important in OUD treatment, where the quick reduction of withdrawal symptoms could have a major effect on patient retention and engagement in care.
The use of injections, particularly long-acting formulations allows the creation of fewer dosing regimes, which allows the circumvention of the barriers to adherence to daily medications. Injectable dosage forms facilitate the treatment process by alleviating the burden and burden associated with treatment, thereby enhancing consistency in the treatment process by patients thereby minimizing the risk of relapse. Additionally, the injections administered in a supervised environment provide to the benefit of monitoring and support opportunities, which is necessary due to the multifaceted psychosocial aspects of opioid addiction.
To learn more about this report, Request Free Sample
The North America region is projected to lead the market with a 74.3% share in 2026, attributed to several key factors. The presence of a well-developed healthcare infrastructure and a large rate of opioid use disorder in the region contributes to the unwavering need to find efficient treatment options. Strict state policies like the U.S. Substance Abuse and Mental Health Services Administration (SAMHSA) programs and additional funding of addiction treatment assist the market expansion. There is also strong industry representation by having such large pharmaceutical companies like Alkermes, Indivior, and Braeburn Pharmaceuticals that actively develop and promote novel treatments that affirm leadership in North America.
For instance, in February 2025, Health Canada expanded support for community-based substance use programs under the Substance Use and Addictions Program, including projects linked to treatment access, virtual care, recovery support, and overdose response, highlighting continued market growth potential for opioid use disorder treatment in North America.
(Source- Health Canada)
The Europe region is expected to exhibit the fastest growth in the opioid use disorder market contributing 12.7% share in 2026, owing to the established opioid agonist treatment system, backed by public health programs and ongoing demand for long term therapy. The European Union Drugs Agency reported that about 511,000 clients received opioid agonist treatment in 2023 across 26 EU member states, with methadone accounting for 55% of treatment use and buprenorphine 35%, showing a well-established treatment base that supports market demand. Growth is also supported by rising policy attention on overdose prevention, broader integration of psychosocial support, and updated global treatment guidelines that continue to favor medication-assisted treatment for most opioid-dependent patients.
For instance, in June 2024, the European Monitoring Centre for Drugs and Drug Addiction, now the European Union Drugs Agency EUDA reported that more than half of patients receiving opioid agonist treatment in Europe were treated with methadone while a large share received buprenorphine-based medicines across EU countries.
(Source- EUDA)
The U.S. is one of the leading markets for opioid use disorder because of the high rate of opioid abuse and the existence of a complex system of laws aimed at fighting the crisis. Other programs like Medication-Assisted Treatment (MAT) are also commonly embraced, with companies like Indivior (using its Suboxone medication) and Alkermes (using Vivitrol).
Good relationships between the government and pharmaceutical companies will encourage the research and innovation of new treatments. In the U.S opioid use disorder market, there is an already developed insurance coverage of opioid use disorder drugs, which help in achieving treatment. Also, the continuous government programs constantly revise policies to achieve good care provision and reduce abuse.
The Canada opioid use disorder market is defined by the progressive government policies with focus on reduction of harm and treatment accessibility. The Canadian government promotes the general access to opioid use disorder medications under the form of governmental healthcare coverage. Two major participants, like Purdue Pharma and Braeburn Pharmaceuticals, are actively involved in the provision of both prescription and extended-release formulations which are appropriate to the needs of the patients.
The comprehensive healthcare systems in Canada and a focused strategy in the field of mental health services are associated with better outcomes in patients and encourage a gradual implementation of opioid use disorder treatments.
The market for opioid use disorder in China is developing very fast, as the population becomes more aware of the issues of substance abuse and the governmental efforts to increase access to mental health care. Companies such as Johnson & Johnson and local pharmaceutical companies invest in the development of treatment facilities and the implementation of new therapies. The growing medical spending and adoption of national interventions to fight drug abuse are some of the reasons why China has better rates of diagnosis and access to treatment. The government is also concerned with the delivery of healthcare in the rural areas, which increases accessibility to hitherto under-served segments, as well as market penetration.
India is also becoming an important developmental market and opportunities are facilitated by the growth in healthcare facilities and growing awareness of the population on the problem of addiction to opioids. The Ministry of Health and Family Welfare initiatives are in the process of incorporating mental health services in primary care. Pharmaceutical firms like Cipla and Sun Pharmaceutical are playing their role by coming up with accessible and affordable treatment of opioid use disorder with local market orientation. In spite of the difficulties in providing healthcare services in rural regions and stigma, the focus on substance use disorders on the part of the government is establishing a more productive setting to expand the market.
|
Metric |
Global Status |
|
Global number of Medication Assisted Treatment programs |
There is no single standardized global count of MAT programs because countries track opioid treatment capacity differently and often report medication availability or treatment coverage instead of exact program numbers. What is clear is that MAT availability remains fragmented and uneven across regions. WHO continues to recommend opioid agonist maintenance treatment using methadone and buprenorphine as a core evidence-based approach for opioid dependence treatment. |
|
Treatment gap patients needing treatment vs receiving therapy |
The treatment gap remains severe at the global level. UNODC reported that only 1 in 12 people with drug use disorders received any form of treatment in 2023, showing that the majority of people needing care are still outside the treatment system. |
|
Opioid specific burden supporting MAT demand |
WHO estimates that about 125,000 people died of opioid overdose in 2019, and opioid overdoses that do not lead to death are several times more common. This continuing mortality burden supports the need for wider MAT access globally. |
|
Regional evidence of uneven access |
Access is also highly inconsistent across health systems. In WHO’s Eastern Mediterranean Region progress update published in September 2025, only eight countries reported having at least one opioid agonist agent available for maintenance treatment, underlining how limited MAT infrastructure still is in several regions. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players in the global opioid use disorder market maintain leadership by investing heavily in research and development to improve treatment effectiveness and expand medication options. They also strengthen their position through partnerships with pharmaceutical companies and healthcare providers while expanding distribution networks into emerging markets where treatment demand is rising. |
Indivior demonstrates this strategy through continuous innovation in its buprenorphine portfolio. In February 2025, the U.S. FDA approved a label update for Sublocade, allowing a rapid treatment initiation pathway, which reflects how leading companies use product development to improve patient access and reinforce their market presence. |
|
Mid-Level Players |
Mid-level players in the opioid use disorder market compete by offering cost-effective treatment options that balance affordability and clinical effectiveness. They often focus on price-sensitive markets and expand their reach through partnerships with technology providers, contract manufacturers, and regional distributors to improve production capacity and distribution efficiency. |
Amneal Pharmaceuticals represents a mid-tier strategy by launching over-the-counter naloxone hydrochloride nasal spray in the U.S. in 2024, providing a lower-cost overdose reversal option and expanding access to opioid overdose treatment. |
|
Small-Scale Players |
Smaller scale players in the global opioid use disorder market usually compete by targeting niche gaps that larger companies do not fully address. They tend to focus on specialized formulations, digital support tools, or more tailored treatment approaches, and they often rely on partnerships with local distributors, academic groups, or technology developers to build market access without needing the scale of major pharma companies. |
Orexo is a strong example. The company positions itself around OUD focused treatment solutions and has combined its core medicine Zubsolv with digital support offerings such as MODIA for patients undergoing clinical treatment plans for opioid use disorder. That shows how a smaller player can stay relevant by pairing a niche OUD focus with innovation and targeted partnerships rather than competing head on across the full market. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6,303.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.4% | 2033 Value Projection: | USD 9,860.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Indivior, Braeburn, Camurus, Alkermes, Emergent BioSolutions, Orexo, Hikma Pharmaceuticals, Dr Reddy’s Laboratories, Alvogen, Sun Pharmaceutical Industries, Amneal Pharmaceuticals, Viatris, Teva Pharmaceuticals, Sandoz, and Kaléo |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
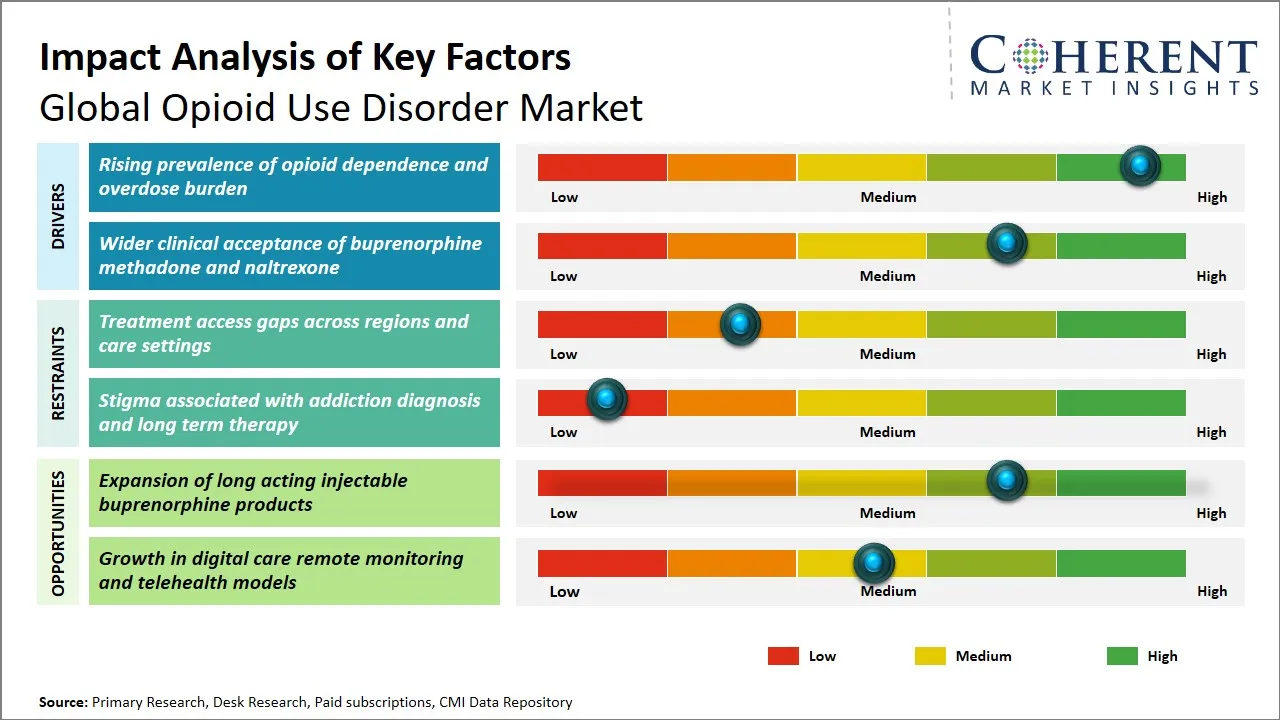
The rising cases of opioid dependence in the world today has emerged as a pressing issue that is fueling the need of having viable means to remedy the market in opioid use disorder. Prescription opioids have become widely abused and numerous illegal opioids like heroin and synthetic forms including fentanyl have been on the rise leading to an increasing number of people being affected as a result of opioid addiction. This increase has been met with an agonizing upsurge in cases of opioid-related overdoses, and this compounds a lot of pressure on any healthcare system to deliver timely and holistic interventions. The complicated nature of opioid use disorder management, as well as the persistence of long-term care to prevent relapse and treatment of comorbid mental health conditions, predetermines the necessity of sophisticated pharmacological interventions and support services.
For instance, in November 2024, the Centers for Disease Control and Prevention (CDC) reported over 100,000 drug overdose deaths in the U.S. during the 12-month period ending June 2024, with opioids responsible for the majority of cases, underscoring the growing global burden of opioid dependence and overdose.
(Source- CDC)
The market of opioid use disorder globally is a great opportunity due to the growth of long-acting injectable buprenorphine. The partial agonist, opioid buprenorphine has played a significant role in medication-assisted treatment (MAT) because it is effective in alleviating withdrawal symptoms and cravings. Nevertheless, the conventional sublingual dosage is often vulnerable to the non-adherence factors in patients, such as the need to take the medication daily and the possibility of misuse or diversion.
Long-acting injectable formulations help counter these weaknesses by offering extended drug release during longer periods usually weeks or months, thereby improving compliance and limiting chances of abuse. This system of administration does not only enhance the efficacy of treatment but also helps to reduce the workload of healthcare providers by the fact that they do not need to visit patients as often to administer the treatment. New regulatory approvals and the increasing acceptance within the clinical practice have facilitated the increased uptake of these products.
For instance, in May 2023, the U.S. FDA approved Brixadi extended-release buprenorphine injection, available in weekly and monthly doses, highlighting the expanding availability of long-acting injectable buprenorphine products for opioid use disorder treatment.
(Source- U.S. FDA)
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients