The Global Buprenorphine Market is estimated to be valued at USD 2,780.6 Mn in 2026 and is expected to reach USD 4,316.9 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.1% from 2026 to 2033. This steady growth reflects increasing adoption of buprenorphine in opioid dependency treatments and expanding healthcare infrastructure worldwide, driving market expansion over the forecast period.
|
Current Events |
Description and its Impact |
|
Expansion of telehealth-based opioid treatment services |
|
|
Growing shift toward long-acting buprenorphine formulations |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The Sublocade (buprenorphine extended‐release) segment is expected to hold the largest market share of 38.1% in 2026, due to its advanced pharmacological profile and substantial benefits in treating opioid use disorder (OUD). Because of the slow release of buprenorphine over more than a month, the extended-release formulation of Sublocade offers a persistent release of plasma drug. This reduces the uneven plasma drug levels that are typical of the daily dosing.
This slower-acting process aids in stabilizing therapeutic levels and this greatly minimizes the withdrawal symptoms and cravings, which are major problems of OUD patients. In addition, the monthly injection increases compliance with treatment regimens, which is one of the key conditions of the success of long-term opioid dependency management.
For instance, in November 2018, Indivior announced Health Canada’s approval of Sublocade for patients with moderate to severe opioid use disorder. This was a major international expansion milestone for the branded long-acting buprenorphine product and supported its position beyond the U.S. buprenorphine market.
(Source- Indivior)
The branded segment is expected to hold 68.9% of the market share in 2026, owing to patent protections, perceived quality, and enhanced physician confidence. Healthcare providers and patients tend to consider branded buprenorphine products as a more high-quality source since they are supported by a multitude of clinical trials, high quality control and full safety data. This is the perception that causes more physician confidence in prescribing branded medications, particularly in combating the intricate cases of opioid dependence, in which treatment efficacy and patient safety are the most important considerations.
Branded formulations with patent protection also enable the pharmaceutical firms to invest a lot in research and development, which makes the introduction of new drug delivery systems, like extended-release injectable formulations or a combination with naloxone. The cycle of innovations supports the clinical appeal of branded products, as pharmaceutical businesses can insist on high-pricing and demand loyalty on their part among the treatment centers.
For instance, in September 2023, Brixadi, the U.S. brand of Camurus’ long-acting buprenorphine product marketed by Braeburn, continued its rollout momentum after its U.S. launch, with Camurus later reporting that it had surpassed prior launches in the segment and gained meaningful share in long-acting buprenorphine. That is another clear sign that branded formulations are leading the higher-value side of the market.
(Sources- Camurus)
The injectable segment is expected to hold the largest market share of 41.1% in 2026, due to enhanced compliance, reduced diversion risks, and improving clinical outcomes. The injectable method provides a controlled-release system that enables continuous release of medication within a long period of time, usually one month, independent of the self-administering of the drug on a daily basis. This time-release profile is very effective with regard to patient compliance, because chances of missed doses which is a major relapse factor and treatment failure are reduced.
The use of injectables also mitigates the issues of diversion and abuse risk of sublingual or buccal administration which is more prone to abuse or accidental exposure. As the medication is injected under the supervision of healthcare workers, the injectable formulations will provide the drug with the appropriate dosage and minimize the risk of selling the medication on the black market or misuse. These reasons have rendered injectables to be a choice in structured treatment programs between clinicians and patients.
To learn more about this report, Request Free Sample
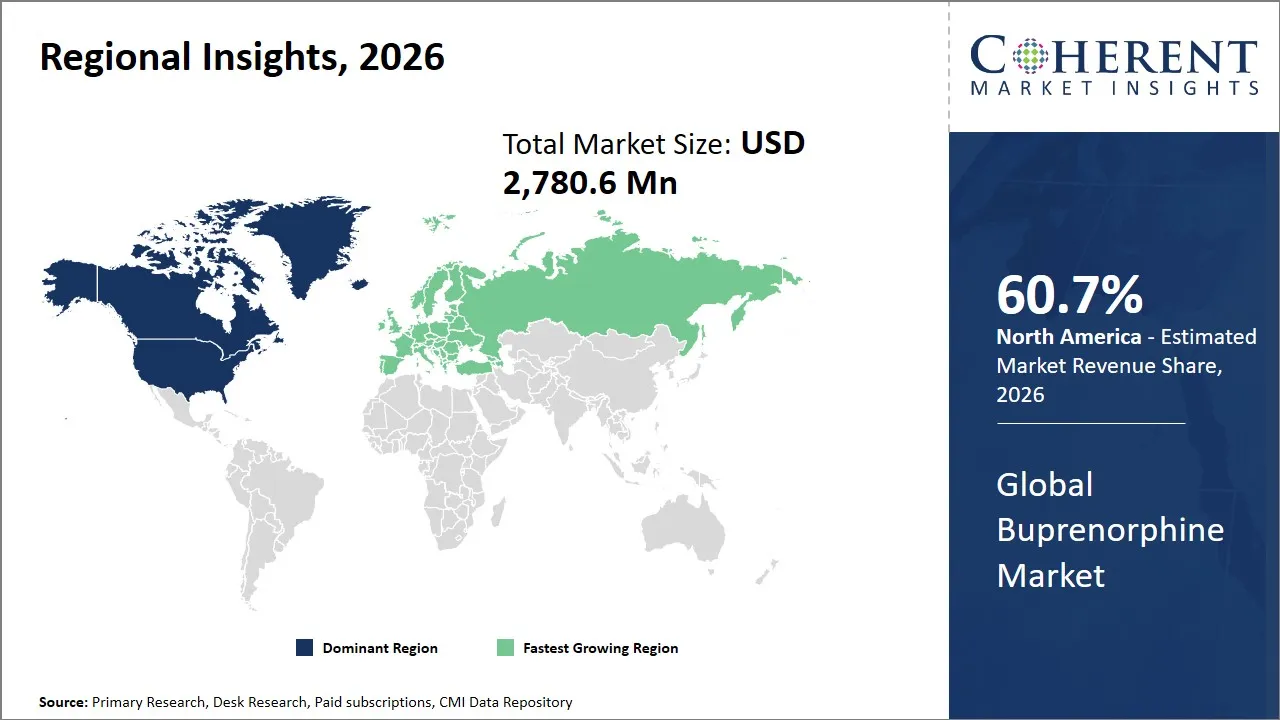
The North America region is projected to lead the market with a 60.7% share in 2026, driven by a well-established healthcare infrastructure, strong government initiatives to combat opioid addiction, and a mature pharmaceutical industry ecosystem. The American government has enacted far-reaching policies to advance opioid use disorder treatment, such as increased access to medication-assisted treatment (MAT) programs, which involve the usage of buprenorphine.
Strong reimbursement systems and favorable regulatory channels also complement penetration in the market. Major industry players like the Indivior PLC and the AbbVie Inc. are very important as they offer new formulations and increased access to treatment. Sustained demand is supported by the existence of a large number of addiction treatment facilities and the extremely high rate of opioid dependence. Also, buprenorphine products can be imported and exported through trade policies, which augment supply.
For instance, in February, 2025, the U.S. FDA approved label changes for Indivior’s Sublocade, allowing a rapid initiation protocol that reduced time to treatment from one week to one hour after a single transmucosal dose. This is a strong regional instance because it reflects how the North American market is moving toward faster initiation, better adherence, and wider use of extended-release buprenorphine therapies.
(Source- Indivior)
The Europe region is expected to exhibit the fastest growth in the buprenorphine market, contributing 28.9% share in 2026, owing to the growing awareness of opioid dependence treatment, expanding access to medication assisted therapy, and the presence of structured public healthcare systems across many countries. The use of buprenorphine is still significant because it is effective, has a low safety rate and can also be used in both maintenance therapy and as a long-lasting recovery support. Community care, increasing access to treatment outpatient programs and the increased inclusion of addiction treatment into mainstream healthcare services are also contributing to demand.
For instance, in May 2025, Camurus launched Buvidal weekly and monthly in Portugal, expanding access to prolonged-release buprenorphine in the country. Earlier in January 2025, Buvidal was launched in Switzerland, while Camurus also reported patient uptake in Luxembourg following prior pricing and reimbursement approvals, marking a broader 2025 European expansion for the brand.
(Sources- Camurus)
The buprenorphine market in the U.S. is the epicenter of buprenorphine use that is widely supported by the government through the Substance Abuse and Mental Health Services Administration (SAMHSA) that issues waivers to permit more healthcare providers to prescribe buprenorphine. The pharmaceutical market in the U.S. is very competitive and Indivior PLC is top at the extended-release forms, with AbbVie growing the generic drug after acquiring the generic business of Allergan.
These organizations pay much attention to R&D to provide better delivery systems, which are more likely to increase patient compliance. Patients are helped with a wide network of addiction care providers and high insurance reimbursements.
The buprenorphine market in India is also in a dynamic state as more people are becoming aware of the incidences of opioid use disorders and the efforts of the government to enhance the services of managing addiction. The regulatory bottlenecks are slowly being overcome and more extensively Medication-Assisted Treatment (MAT) including buprenorphine is used. In addition to that, the international generics manufacturers are collaborating with Indian companies to increase the manufacturing capacity. The integration of buprenorphine into the public health efforts is conducive to the expansion of the market in the long-run in case of increased patient awareness.
Germany is the market leader in Europe with a highly regulated pharmaceutical landscape as well as well-established healthcare reimbursement schemes that support buprenorphine. Progressive policies favor the country in the sense that they make opioid substitution therapies a normative treatment with health insurance cover. Other major industry actors like Mundipharma GmbH and Laboratorios LICONSA maintain consistent supply by providing high quality branded products and generic products. Having research institutions also facilitates drug formulation and treatment procedures. Germany as a trading bloc in the European Union also boosts the availability of products in the adjacent markets.
The market of buprenorphine in China has been on its way to growing due to the rising considerations of mental health and addiction therapy in the national health agendas by the government. New regulatory frameworks enable more affordable access to buprenorphine products in urban areas and pilot programs to implement treatment in the rural areas.
Chinese drug producers such as CSPC Pharmaceutical Group and Jiangsu Hengrui Medicine are also investing in their production and distribution capacity. Cooperation with foreign companies will contribute to the implementation of new recipes and compatibility with the international requirements. Public health campaigns led by the government enhance patient acceptance making China a major growth market in Asia Pacific.
To learn more about this report, Request Free Sample
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players lead the buprenorphine market by investing in improved formulations and delivery systems that enhance adherence and treatment outcomes. They also use partnerships and wider geographic expansion to strengthen market reach, especially in regions where opioid dependence treatment access is still developing. |
Indivior strengthened its position in February 2025 when the U.S. FDA approved label changes for Sublocade, allowing faster treatment initiation and supporting broader use of long- acting buprenorphine therapy. |
|
Mid-Level Players |
Mid-level players compete by offering more affordable buprenorphine products while using partnerships to improve technology access, manufacturing strength, and regional distribution. They usually focus on selected markets or patient segments instead of competing head-on with the largest companies. |
In January 2025, ZIM Laboratories and Neuraxpharm received European marketing authorization for buprenorphine sublingual film, showing how mid-sized companies can use collaboration and formulation innovation to expand market presence cost-effectively. |
|
Small-Scale Players |
Small-scale players stay relevant by focusing on niche innovation, such as new delivery systems and patient-friendly formulations, instead of competing on scale. They also rely on targeted collaborations with research bodies or regional partners to build visibility and enter specific markets faster. |
In July 2025, Akyso Therapeutics announced NIH funding to develop a buprenorphine implant, showing how a smaller company can use innovation-led, partnership-backed development to target unmet needs in opioid use disorder treatment. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,780.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.1% | 2033 Value Projection: | USD 4,316.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
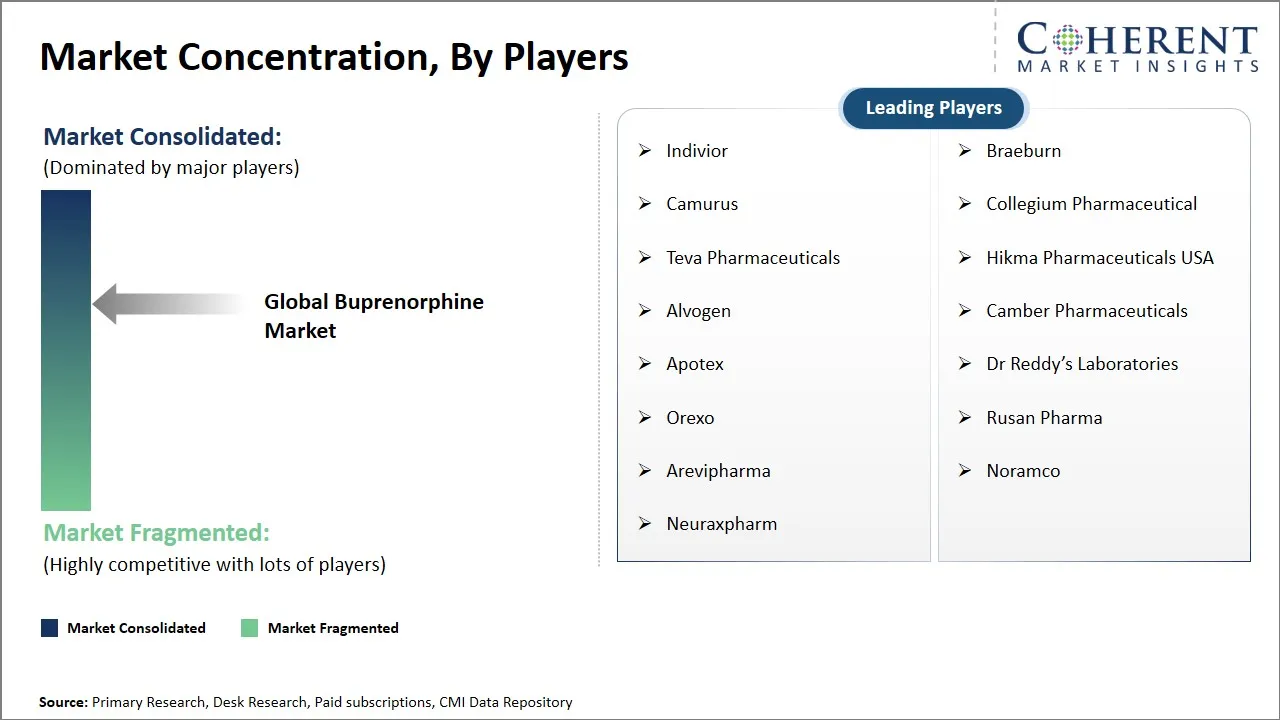
Indivior, Braeburn, Camurus, Collegium Pharmaceutical, Teva Pharmaceuticals, Hikma Pharmaceuticals USA, Alvogen, Camber Pharmaceuticals, Apotex, Dr Reddy’s Laboratories, Orexo, Rusan Pharma, Arevipharma, Noramco, and Neuraxpharm |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
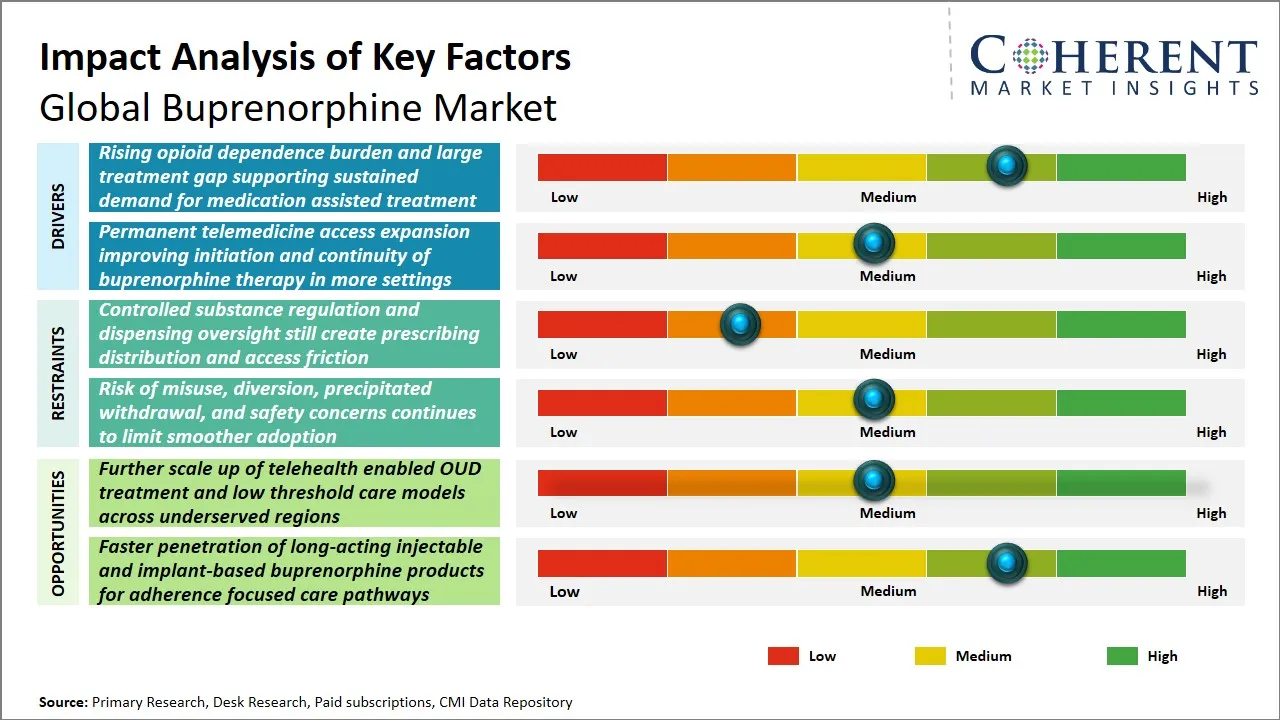
The rising rates of opioid dependence across the world have been placing a significant strain on healthcare systems presenting an urgent need to develop effective therapeutic interventions to curb the crisis like buprenorphine. Although there is an increasing awareness, this treatment gap continues with the lack of access, stigmatization, and lack of access to specialized care forcing many people to be untreated or poorly treated.
The pharmacological characteristics of buprenorphine, including the reduction of the withdrawal symptoms and cravings with the less risk of misuse than other opioids, precondition its status as a key component of medication-assisted treatment (MAT) programs. The focus on increasing access to MAT remains on the rise as healthcare providers are trying to mediate the multi-faceted issues related to opioid use disorder, such as high relapse rates and comorbidity.
For instance, in the U.S., opioid use disorder remains high while treatment access is still limited, supporting the demand for buprenorphine. Centers for Disease Control and Prevention (CDC) reported that in 2022, that 3.7% of U.S. adults needed Opioid Use Disorder (OUD) treatment, and 25.1% of them received medications for OUD; similarly, NIH reported that in 2021 only about 1 in 5 adults with OUD received medications to treat it.
(Sources- Centers for Disease Control and Prevention (CDC))
The acquisition of the additional scale of the telehealth-enabled treatment of opioid use disorder (OUD) and the introduction of low-threshold care models is a major market expansion prospect in the international buprenorphine sector, especially in underserved and rural areas. The current opioid crisis has highlighted the necessity of accessible and patient-centered treatment options, and telehealth platforms have become a highly important approach to addressing geographical and social disparities in care.
As a first-line treatment of OUD, buprenorphine is also suitable as a remote management option because of its safety and lack of regulations in most jurisdictions. Telemedicine integration expands access because healthcare providers are able to perform an assessment and prescribe medicine along with checking medication adherence without having to physically visit the patient, alleviating certain concerns of stigma, transportation challenges, and inadequate healthcare infrastructure.
For instance, on December 31, 2025, the Drug Enforcement Administration (DEA) announced a fourth temporary extension of telemedicine flexibilities through December 31, 2026, allowing remote prescribing of buprenorphine without a prior in-person visit under specified conditions. Drug Enforcement Administration (DEA) said this helps ensure continuity of care, particularly for patients in rural and underserved areas, making it a strong access-expansion development for buprenorphine treatment.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients