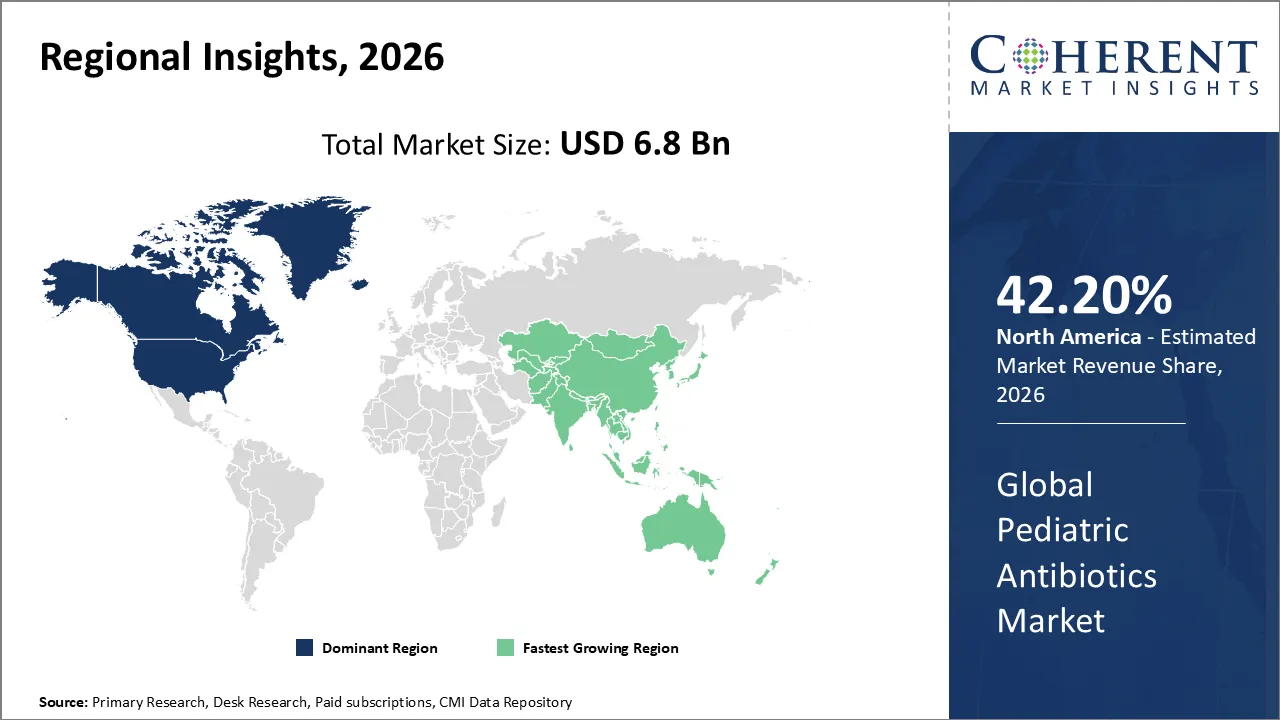
The Pediatric Antibiotics Market is estimated to be valued at USD 6.8 Bn in 2026 and is expected to reach USD 9.2 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 3.1% from 2026 to 2033.
Healthcare providers drive the pediatric antibiotics market by treating a high prevalence of infectious diseases among children, especially respiratory, ear, and skin infections that require timely intervention. Expanding healthcare access, increasing hospital admissions, and the availability of child-friendly formulations such as suspensions and dispersible tablets support pediatric antibiotics market demand. Pharmaceutical companies advance growth through drug development, while hospital and retail pharmacies strengthen distribution. At the same time, regulators and clinicians address antimicrobial resistance by enforcing stricter prescribing guidelines and promoting more cautious, targeted antibiotic use.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Changes in Key Regions |
|
|
Technological and Scientific Advancements |
|
|
Competitive and Industry Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospital Pharmacies acquired the prominent market share of 39.3% in 2026. Hospital pharmacies drive the pediatric antibiotics market by supplying essential medications promptly to children with acute and complex infections. They offer specialized formulations, such as liquid suspensions and age-specific doses, to ensure accurate and safe administration. Pharmacists collaborate closely with pediatricians and infectious disease specialists to optimize therapy and monitor treatment. Increasing pediatric hospitalizations, especially in intensive care units, boost reliance on hospital pharmacies, while their active role in managing multidrug-resistant infections and following regulatory standards strengthens ongoing antibiotic demand.
Acute Sinusitis hold the largest market share of 42.2% in 2026. Clinicians drive the pediatric antibiotics market by treating children who present with persistent nasal congestion, facial pressure, and fever after upper respiratory infections. School exposure and underdeveloped immunity increase children’s susceptibility to acute sinusitis. Physicians often prescribe antibiotics due to difficulty distinguishing viral from bacterial cases. Parents seeking quick relief further influence prescribing behavior. Expanding healthcare access and the availability of effective first-line antibiotics sustain demand, while seasonal infection surges continue to support consistent treatment volumes.
Intravenous expected to hold largest market share of 37.8% in 2026. Healthcare providers drive the pediatric antibiotics market by prioritizing intravenous delivery for rapid and reliable treatment of severe infections such as sepsis and complicated pneumonia. Hospitals and intensive care units use IV administration to ensure precise dosing and immediate therapeutic action. Increasing admissions of critically ill and immunocompromised children boost demand. Clinicians choose IV therapy when oral options fail or prove unsuitable. The growing presence of resistant pathogens also pushes providers to use potent injectable antibiotics for effective disease management. For instance, in March 2026, Long Grove Pharmaceuticals launched the generic Dalvance, dalbavancin for injection, a single-dose antibiotic administered intravenously for pediatric patients to treat acute Gram-positive bacterial skin infections.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 42.20% in 2026. Pharmaceutical companies and healthcare providers drive the North America pediatric antibiotics market by developing advanced formulations and targeted therapies for children. Widespread healthcare access, established hospital networks, and strong insurance coverage enable early diagnosis and prompt treatment of infections. Clinicians address rising antimicrobial resistance through strategic prescribing, while government initiatives, awareness campaigns, and robust regulations ensure safe and effective use. These combined efforts create a market defined by high-quality pediatric care and sustained demand for antibiotics across the region. For instance, in March 2025, GSK plc announced that the FDA approved Blujepa (gepotidacin) to treat adults (≥40 kg) and pediatric patients (≥12 years, ≥40 kg) with uncomplicated urinary tract infections caused by susceptible E. coli, K. pneumoniae, C. freundii complex, S. saprophyticus, and E. faecalis.
Healthcare providers and pharmaceutical companies drive the Asia Pacific pediatric antibiotics market by expanding access and raising awareness of childhood infections. Hospitals and clinics increasingly use pediatric-specific formulations, while rising hospital admissions boost demand for oral and intravenous antibiotics. Companies develop affordable, child-friendly medications to address diverse regional needs, and clinicians prioritize early treatment and infection management in high-prevalence areas. Government programs and public health initiatives actively promote safe antibiotic use, collectively shaping a market driven by accessibility, growing demand, and evolving pediatric care practices.
Healthcare providers drive the United States pediatric antibiotics market by prioritizing early diagnosis and effective management of childhood infections. Hospitals and clinics use pediatric-specific formulations, such as liquid and dispersible options, to ensure accurate and safe dosing. Pharmaceutical companies innovate with broad-spectrum and child-friendly antibiotics, while clinicians apply strategic prescribing to address antimicrobial resistance. Strong insurance coverage, established hospital networks, and government health programs enhance access and treatment. These combined efforts shape a market defined by safety, innovation, and steady pediatric antibiotic demand. For instance, Krystal Biotech announced that the FDA approved VYJUVEK (beremagene geperpavec‑svdt) to treat patients aged six months and older with dystrophic epidermolysis bullosa (DEB).
Healthcare providers and pharmaceutical companies drive the China pediatric antibiotics market by treating a high burden of childhood infections and managing increasing hospital visits. Hospitals and clinics use pediatric-specific formulations, such as liquid suspensions and age-appropriate dosages, to ensure safe and accurate administration. Growing parental awareness and proactive infection management by clinicians increase antibiotic use. Pharmaceutical manufacturers develop affordable, accessible child-friendly medications, while government initiatives and public health programs enforce responsible prescribing, collectively strengthening market growth and enhancing pediatric healthcare outcomes.
Pharmaceutical companies increasingly focus on developing child-friendly formulations such as flavored suspensions, dispersible tablets, and age-adjusted dosages. These products improve adherence, reduce administration difficulties, and cater to infants, toddlers, and school-aged children. Hospitals and pharmacies adopt these formulations widely to ensure safe dosing, while caregivers appreciate easier administration. This trend highlights a growing emphasis on convenience, accuracy, and patient-centric drug design in pediatric antibiotic therapy, shaping product development strategies globally.
Clinicians are increasingly choosing antibiotics based on pathogen identification and local resistance patterns. Broad-spectrum and targeted therapies are preferred for severe infections or resistant strains. This trend is supported by advancements in diagnostic tools and antimicrobial stewardship programs. Pediatric care emphasizes balancing efficacy with minimizing unnecessary exposure. The adoption of precision prescribing and guideline-driven therapy is driving the development of antibiotics that are both effective and safe for children.
There is a strong opportunity for pharmaceutical companies to develop palatable, easy-to-administer pediatric antibiotics, including flavored suspensions, chewables, and dispersible tablets. Caregivers seek solutions that simplify dosing and improve adherence, especially for younger children. Expanding product lines to cover a wider age range and offering formulations that reduce gastrointestinal discomfort can enhance market adoption. Companies that focus on patient-centric, convenient, and safe formulations can capture significant pediatric demand and strengthen brand loyalty.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.1% | 2033 Value Projection: | USD 9.2 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Astellas Pharma, Inc, Pfizer Inc., Novartis AG, Bayer AG, Bristol-Myers Squibb Company, Abbott., DAIICHI SANKYO COMPANY, LIMITED, Dr. Reddy’s Laboratories Ltd, Teva Pharmaceuticals USA, Inc., Aurobindo Pharma USA, GlaxoSmithKline Plc., Hikma Pharmaceuticals PLC, Cipla Inc., Glenmark Pharmaceuticals, and Torrent Pharmaceuticals Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients