Plant-Based Vaccines Market is estimated to be valued at USD 332.8 Mn in 2026 and is expected to reach USD 6,097.2 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 51.5% from 2026 to 2033.
Analysts’ Views on the Global Plant-based Vaccines Market:
The increasing research and development of plant-based vaccines is expected to drive the growth of the market over the forecast period. For instance, according to data published in September 2021 by Genetic Engineering & Biotechnology News University of California-Riverside, researchers are studying whether it can turn edible plants like lettuce into mRNA vaccine factories.
Figure 1. Global Plant-based Vaccines Market Share (%), By Application, 2026
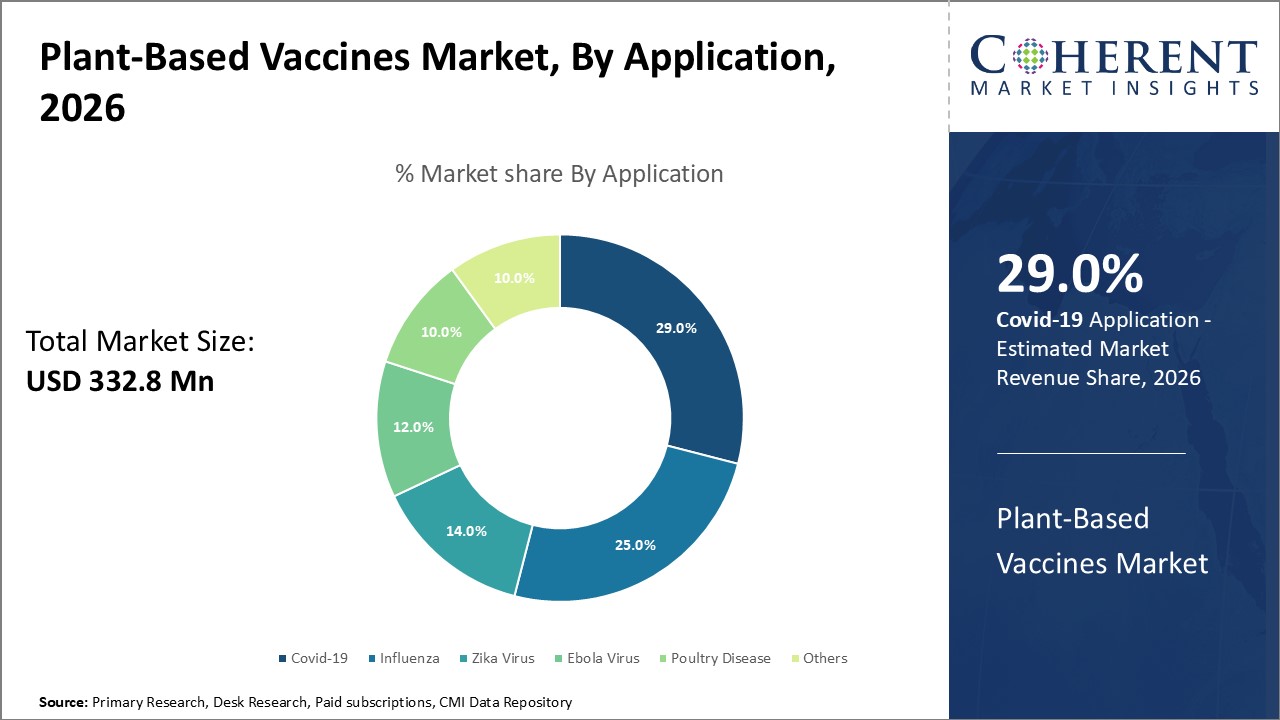
To learn more about this report, Request Free Sample
Global Plant-based Vaccines Market - Drivers
Figure 2. Global Plant-based Vaccines Market Share (%), By Region, 2026
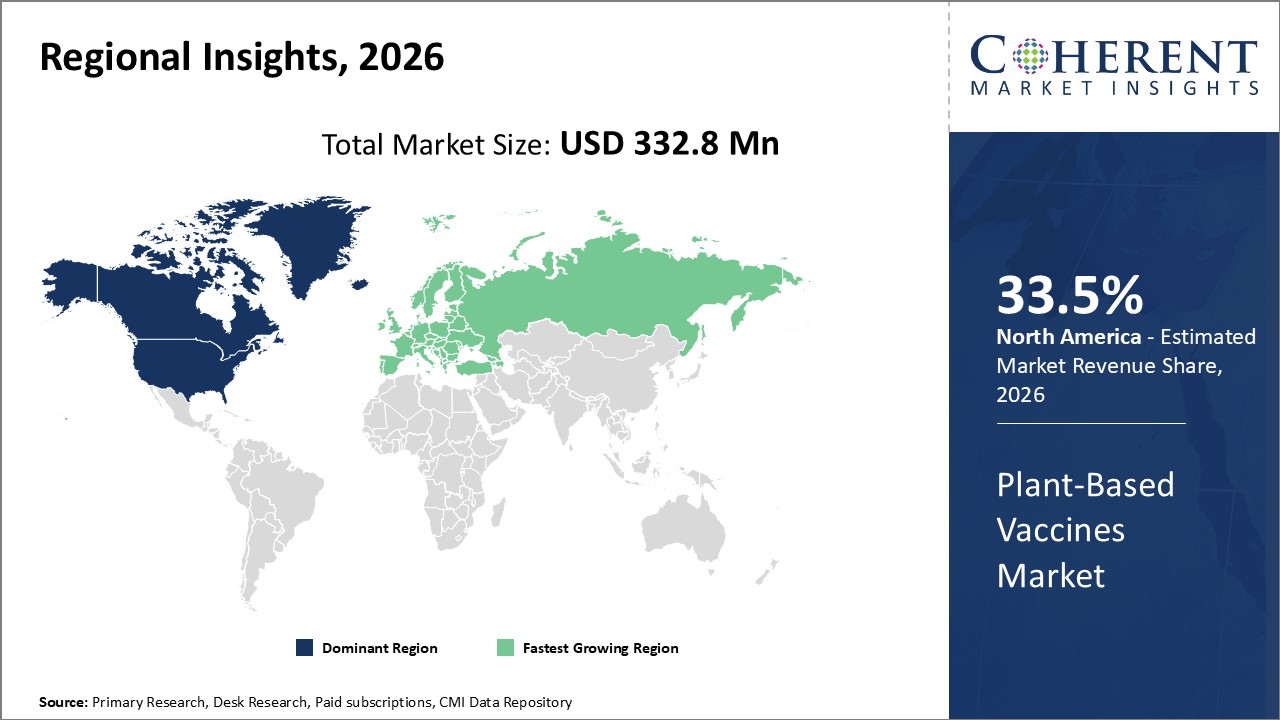
To learn more about this report, Request Free Sample
Global Plant-based Vaccines Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global plant-based vaccines market over the forecast period. The global plant-based vaccines market is expected to witness significant growth in the coming years, driven by the increasing technology advancement of plant based vaccines.
Plant-based Vaccines Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 332.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 51.5% | 2033 Value Projection: | USD 6,097.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medicago, Inc., IBIO, Inc., Icon Genetics GmbH, Lumen Bioscience, Inc., British American Tobacco Plc., Creative Biolabs, Inc., Leaf Expression Systems Ltd., ZYUS Life Sciences Inc., PlantForm Corporation, and Infectious Disease Research Institute (IDRI). |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Plant-based Vaccines Market Segmentation:
The global plant-based vaccines market report is segmented into vaccine type, source, application and region.
Global Plant-based Vaccines Market - Cross Sectional Analysis:
Increasing prevalence of the Ebola Virus in Africa is expected to drive the growth of the Africa market over the forecast period. For instance, according to data published in December 2021 by the National Library of Medicine, in 2020, 3470 Ebola virus disease cases had been reported, including 3317 confirmed and 153 probable cases, with 2287 deaths in the Democratic Republic of the Congo, Africa.
Global Plant-based Vaccines Market: Key Developments
Global Plant-based Vaccines Market: Key Trends
Global Plant-based Vaccines Market: Restraint
Global Plant-based Vaccines Market - Key Players
The major players operating in the global plant-based vaccines market include Medicago, Inc., IBIO, Inc., Icon Genetics GmbH, Lumen Bioscience, Inc., British American Tobacco Plc., Creative Biolabs, Inc., Leaf Expression Systems Ltd., ZYUS Life Sciences Inc., PlantForm Corporation, and Infectious Disease Research Institute (IDRI).
Definition: Plant-derived vaccines are produced by recombinant technology, in which the gene encoding the desired antigen protein is integrated into the plant genome. Agrobacterium tumefaciens is commonly used for gene transfer and transformation.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients