Pulmonary Drugs market is estimated to be valued at USD 56.6 Bn in 2026 and is expected to reach USD 96.5 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033.
The global market for pulmonary drugs is expected to grow a lot by 2026. This is because conditions such as chronic respiratory problems like asthma, COPD, allergic rhinitis, and pulmonary arterial hypertension are becoming more prevalent because of issues like pollution in urban centers, the aging population, and lifestyle factors. It is true that medication delivery systems have improved, making them more potent and increasing compliance in patients. There also have been targeted and personalized approaches from pharmaceutical companies.
|
Current Events |
and its impact |
|
China's Air Pollution Crisis and Regulatory Response |
|
|
Breakthrough Gene Therapy and Precision Medicine Developments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on drug class, the inhaled corticosteroids segment is estimated to contribute the highest market share of 29% in 2026, owing to its proven efficacy in managing respiratory diseases like asthma and COPD. Inhaled corticosteroids reduce inflammation in the air passages of the lungs and prevent the worsening of the disease.
For instance, in May 2025, Nucala (mepolizumab) was approved by the US FDA as an add-on maintenance treatment for adults with COPD characterized by an eosinophilic phenotype-a key biologic approval that complements inhaled corticosteroids in hard-to-control disease and underlines the rising role of targeted therapies in pulmonary care.
By application, the asthma & COPD segment is estimated to contribute the highest market share of 42% in 2026. The prevalence of these respiratory diseases across the globe, coupled with increased pollution and lifestyle factors, contributes to the overall need to control these emerging diseases. Healthcare providers still focus significantly on treating and diagnosing the diseases, thereby fueling the overall adoption of medicines across pulmonary disorders.
For instance, January 2025, AstraZeneca announced plans to launch Breztri Aerosphere, a triple combination inhaler indicated for maintenance treatment to relieve symptoms and prevent exacerbations in COPD, in India, expanding the geographic availability of advanced COPD therapy options within emerging markets.
Based on distribution channel, the retail pharmacies segment is estimated to contribute the highest market share of 48% in 2026. Retail pharmacies ensure easy access to prescription and non-prescription pulmonary drugs, and patients can conveniently refill their drugs. They also offer counseling services for better utilization of inhalers.
For instance, March 2025, CVS announced plans to open over a dozen smaller, pharmacy‑only stores focused on full‑service pharmaceutical care, including prescription refills and respiratory drug access such as inhalers, to improve community access and localized healthcare services.
To learn more about this report, Request Free Sample
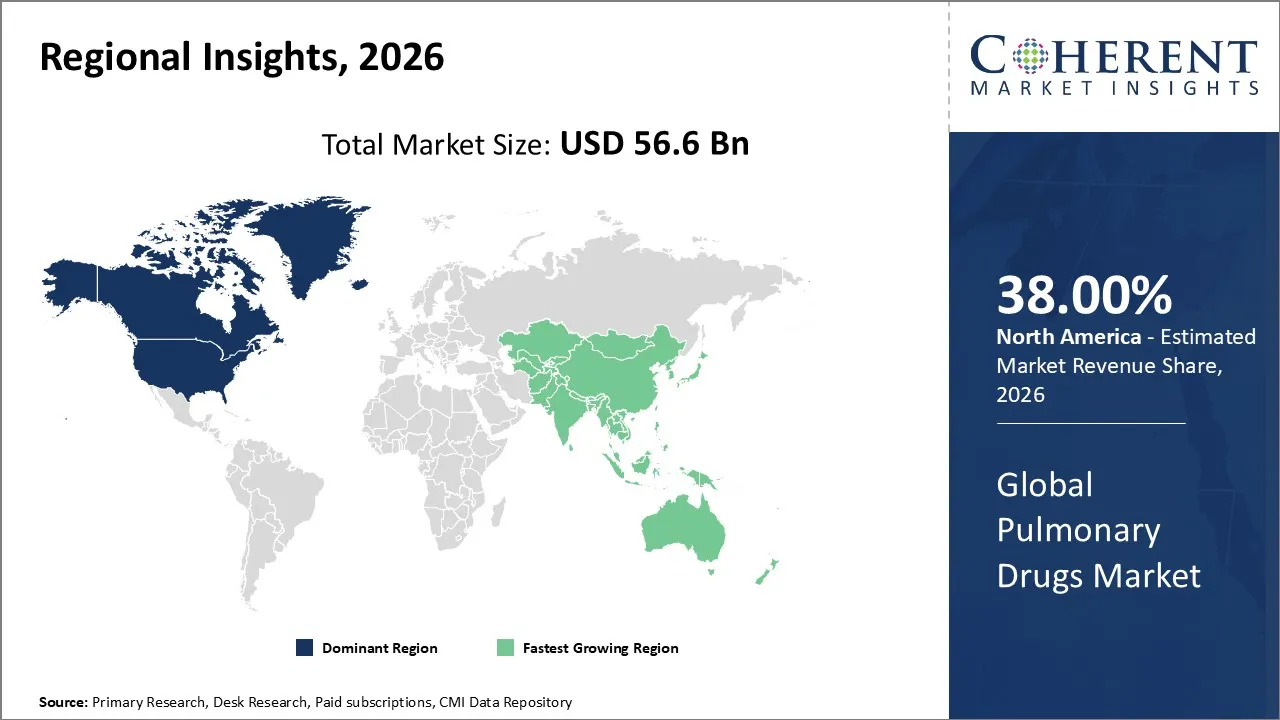
North America is expected to contribute the highest share to the pulmonary drugs market in 2026, accounting for approximately 38% of the market share. The pulmonary market in the North American region can be driven by various factors, including developed infrastructure, the high prevalence of respiratory illnesses like asthma and COPD, the widespread use of innovative medicines like inhalers and biologics, and pharmaceutical sector R&D spends.
For instance, in July 2025, Merck & Co. announced a roughly $10 billion acquisition of Verona Pharma, a biotech focused on respiratory drugs, gaining access to Ohtuvayre, an FDA‑approved inhaled treatment for COPD, which has shown early commercial uptake in the U.S. and is expected to strengthen Merck’s portfolio and distribution reach in pulmonary therapies across North America.
Asia Pacific is likely to witness high growth in terms of pulmonary drugs due to an escalating rate of pulmonary diseases resulting from urban pollution, health awareness among people, increased disposable income, and accessibility of health care services. Domestic pharmaceutical companies, along with global players, are establishing business relationships, thus making innovative pulmonary drugs accessible, hence boosting growth.
For instance, in April 2024, GSK expanded the Asia‑Pacific reach of Trelegy Ellipta (a triple‑combination inhaled therapy for asthma and COPD) with regulatory approval in Singapore, allowing broader use of this maintenance treatment across respiratory indications in the region.
The U.S. pulmonary drugs market is growing at a rapid pace due to a high prevalence of chronic respiratory illnesses like asthma, chronic obstructive pulmonary diseases, and pulmonary arterial hypertension, along with a strong healthcare infrastructure. For instance, in May 2025, GSK’s Nucala (mepolizumab) received
China’s heightened focus on health, wellness, and air quality initiatives is accelerating growth in the pulmonary drugs market. Higher pollution levels and the prevalence of diseases like asthma and COPD are boosting the demand for traditional inhalers and biologic drugs. For instance, in September 2024, Dupixent (dupilumab) was approved in China for adults with uncontrolled COPD and elevated eosinophils, marking a milestone in biologic therapy availability in Asia Pacific.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 56.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 96.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi SA, Meda Pharmaceuticals, Circassia Pharmaceuticals Plc., AstraZenca Plc., GlaxoSmithKline Plc., Mallinckrodt Pharmaceuticals Plc., Cheisi farmaceutici S.p.A., Zambon Company S.p.A., Alaxia SAS, and Merck Sharp & Dohme Limited. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The growing incidence rate of asthma, chronic obstructive pulmonary disease (COPD), allergic rhinitis, and pulmonary arterial hypertension is fueling the pulmonary drugs market. The demographic features of a growing aging population, pollution, and lifestyle factors that predispose individuals to disease are contributing factors. As more individuals with one or more of these chronic diseases strive for better disease management, a strongly growing market for pulmonary drugs is witnessed.
The market has strong growth prospects in terms of the further evolution of next-generation drug delivery systems and biologic agents. For example, further innovation is expected to take place regarding smart inhalers, combination inhalers, and biologic agents. In addition, access to these technologies will increase in the emerging markets. Moreover, access to new agents for the treatment of underserved diseases will increase.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients