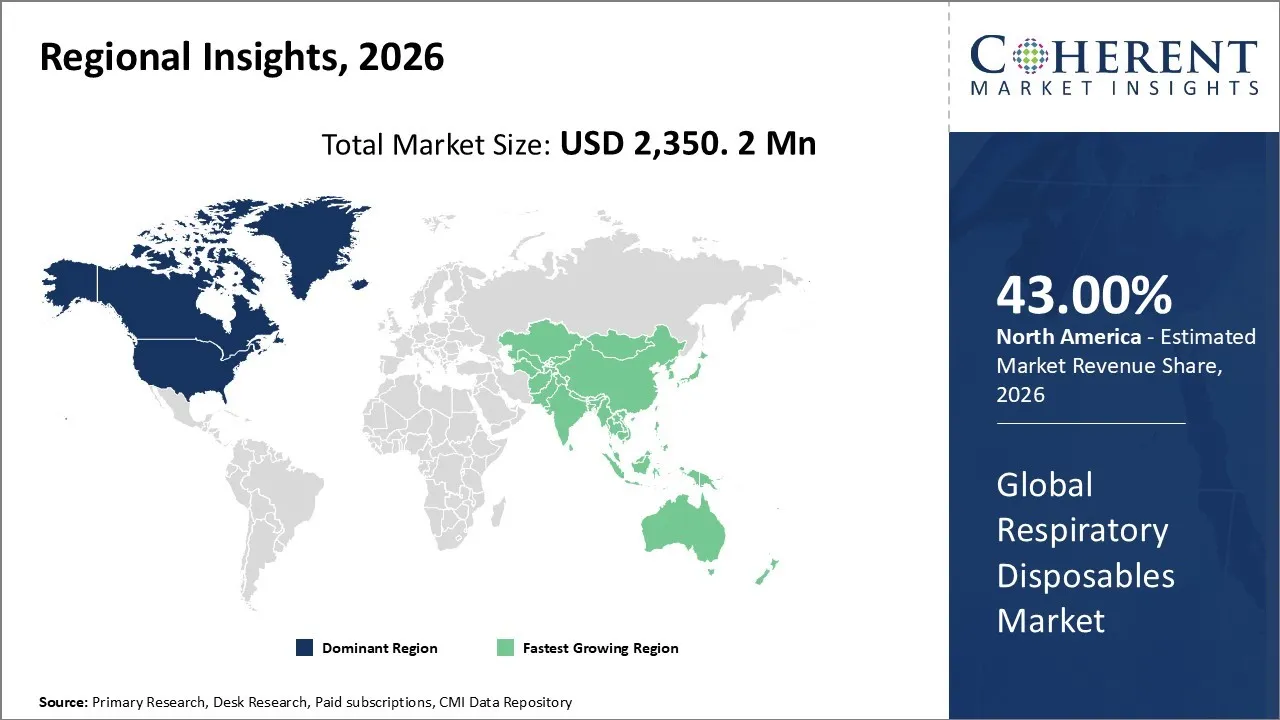
The global respiratory disposables market size is expected to reach approximately USD 2,350.2 Mn in 2026 and USD 4,350.4 Mn by 2033, growing at a CAGR of 9.8% throughout the forecast period (2026-2033).
Respiratory disposables are one time-use medical products utilized in ventilation support, respiratory care, airway management and oxygen therapy. These products are non-reusable and sterile and discarded to prevent hospital-acquired infections, cross-contamination, and ensure patient safety. The respiratory disposables market is gaining strategic importance due to rising burden of respiratory diseases such as Acute Respiratory Distress Syndrome (ARDS), Respiratory infections, COPD (Chronic Obstructive Pulmonary Disease), Asthma and Sleep apnea among others. The healthcare operators are increasingly adopting the use of single-use respiratory products because of strong focus on infection prevention & hospital safety. Moreover, due to expansion of critical care infrastructure and ICU, growth of home respiratory care and regulatory push for single-use medical products is driving the growth of the respiratory disposables market.
|
Current Event |
Description and its Impact |
|
Integration of Technology in Medical Devices |
|
|
Growing concerns about infection control in Healthcare Infrastructure |
|
|
Increasing Adaptability to Home based Respiratory Care |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Month & Year |
Product |
Category |
Company |
Approval Type / Regulatory Action |
|
Apr 28, 2025 |
Shiley Adult Flexible Tracheostomy Tube XLT with Disposable Inner Cannula |
Disposable tracheostomy tube & inner cannula |
Covidien LLC (Medtronic) |
FDA 510(k) clearance — Traditional 510(k) submission (confirmed device cleared and summary available) |
|
2025 |
Optiflow+ Nasal Cannula (Small, Medium, Large) |
Disposable nasal oxygen cannula interfaces |
Fisher & Paykel Healthcare Ltd. |
FDA 510(k) clearance for multiple cannula sizes (K251611) |
|
2025 Nov |
Generic QVAR equivalent (inhaler) |
FDA Approval |
Amneal Pharmaceuticals |
U.S. approval for a generic corticosteroid inhaler formulation (related to respiratory therapy) |
|
Dec 19, 2025 |
Optiflow Switch+ Filtered Nasal Interface with CO₂ sampling |
Nasal interface for oxygen delivery with CO₂ sampling tube |
Fisher & Paykel Healthcare Ltd. |
FDA 510(k) clearance — Filtered nasal interface combined with sampling (document available) |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on product, resuscitator is expected to lead the market, accounting for a revenue share of 47.5% in 2026. Resuscitators are used widely in hospitals, ambulatory surgical centers, emergency medical services, home care, and field scenarios. Hence, the resuscitators are used extensively and has broad application scope compared with more specialized disposables. This broad usage helps capture a larger share of overall respiratory disposables. After the pandemic infection control priorities and stringent hygiene standards have boosted preference for single‑use/ disposable resuscitators to avoid cross‑contamination risk and simplify protocols increasing segment share within the market. Furthermore, ease of use and versatility of resuscitator and regulatory & training support drives the segment growth.
By end user, hospitals segment is slated to account for a prominent market share of 50% in 2026. Due to highest volume of critical respiratory cases that require oxygen therapy, non-invasive ventilation, or mechanical ventilation the patient admission in hospitals increased.
Higher dependence on ventilation and respiratory support equipment and Strong infection-control requirements drive single-use consumption in hospitals, which is a major factor supporting dominance of the hospital segment in the market.
Based on patient group, adult segment led the global respiratory disposables market with share of 55% in 2026. As the adults shows higher incidence of conditions such as asthma, chronic obstructive pulmonary disease, and pneumonia, the demand for the respiratory consumables rises gradually. Growing aging population, lifestyle risk factors, and higher hospitalization rate further support the dominance of the segment in the market.
To learn more about this report, Request Free Sample
North America is expected to dominate the global respiratory disposables market, accounting for a share of 43% in 2026. Due to higher prevalence of the respiratory infection in the North America region there is increase in the for the respiratory disposables. For instance, in March 2026, according to the data published by the Asthma and Allergy Foundation of America, is has been estimated that asthma is still the most common chronic illness in the United States, affecting approximately 28 million people (about 1 in 12) as of early 2026 and costing the country USD50 billion annually.
Strict infection-control laws favor single-use products and strong reimbursement and home-care support drives the growth of the respiratory disposables market in North America region. Moreover, as North America region has presence of leading manufacturers of innovative respiratory disposables and insurance companies in North America, including government-funded programs like Medicare, cover an extensive product portfolio of respiratory disposables under their policies, which stimulates the growth of the market in the region.
The countries in Asia like India, China and Southeast Asia, are facing high levels of air pollution, which directly contributes to the increasing incidence of respiratory diseases such as chronic obstructive pulmonary disease (COPD), asthma, and lung infections. Due to higher burden of respiratory diseases in the Asia Pacific region the respiratory disposables market grows at significant rate in the region. Moreover, factors like expanding healthcare infrastructure, government initiatives & healthcare reforms and increased healthcare expenditure and access, drive the growth of the Asia Pacific respiratory disposables market.
The U.S. country has higher prevalence of chronic respiratory conditions, including pneumonia, asthma, COPD, and sleep apnea. The regulatory bodies in U.S. such as Food and Drug Administration and Centers for Disease Control and Preventions play a significant role in ensuring the safety and effectiveness of respiratory disposables. For instance, in March 2026, the Glenmark Specialty SA, pharmaceutical company reveals that the company has received the United States Food & Drug Administration (U.S. FDA) final approval for the Fluticasone Propionate Inhalation Aerosol USP, 44 mcg per actuation.
In the U.S. country the respiratory care accounts for a large portion of the substantial healthcare expenditures. The Centers for Medicare & Medicaid Services (CMS) estimated that healthcare spending would surpass USD6 trillion by 2028, which will lead to a rise in investment in medical technologies, particularly respiratory disposables. Rising awareness among the U.S. population for the prevention of the chronic respiratory infections, there is a greater push for early diagnosis and continuous monitoring. As a result, respiratory disposables, particularly those utilized in home care settings, are seeing growing adoption, especially in telehealth environments where respiratory monitoring is increasingly integrated into at-home care.
China country has the large number of ageing population which has observed to increase the demand for the respiratory disposables for preventing the spread of viral infection. China country has strong domestic manufacturing base and broad insurance coverage improves access and repeat consumption. China country leads the Asia Pacific respiratory disposable market due to advanced manufacturing facilities and largest demand base with a very large healthcare delivery and manufacturing system.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,350.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 4,350.4 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Fisher & Paykel Healthcare, Teleflex Incorporated, Drägerwerk AG & Co. KGaA, ResMed, Getinge AB, Smiths Medical / Smiths Group, Air Liquide, Invacare Corporation, Hamilton Medical, Ambu A/S, Intersurgical Ltd., Medline Industries, Koninklijke Philips N.V., Becton, Dickinson and Company (BD) |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Growing incidence of chronic respiratory diseases such as asthma and chronic obstructive pulmonary disease has risen the demand for the respiratory disposables. For instance, in March 2026, according to the data published by the Elsevier Ltd., it has been estimated that in India asthma is the major chronic health issue, with an estimated 35 million people suffering from the condition. India has a disproportionately heavy burden, accounting for 42% of asthma deaths worldwide and 13% of all asthma cases worldwide.
Due technology advancement and shift towards in-home respiratory care the treatment of chronic pulmonary obstructive diseases can be provided outside the hospitals. According to NICE's 2024 assessment, several COPD remote-monitoring devices are utilized to facilitate earlier discharge and prevent needless admissions. These devices help to monitor and check symptoms, blood oxygen levels, and decline at home. As a result, the usage of home respiratory supplies, such as nasal cannulas, masks, and oxygen-delivery devices, is increasing.
The key players operating in the market are adopting the inorganic growth strategies like partnership and collaboration with OEMs & service providers which has estimated to create the significant opportunity for the growth of the respiratory disposables market in the near future. For instance, in December 2025, Teleflex Incorporated, a company that supplies medical technology, signed the agreement to sell its Acute Care, Interventional Urology, and OEM businesses to two buyers: Intersurgical Ltd. for Acute Care and Interventional Urology, and Montagu and Kohlberg for OEM, for a total of USD 2.03 billion in cash, subject to certain closing adjustments.
Furthermore, in January 2026, ENT UK, entered the strategic alliance with Ambu, global medical technology company, to expand the adoption of its single-use airway management and respiratory products, including breathing accessories and single-use airway management. This strategic partnership aims to provide valuable opportunities to support the development of community-focused ENT services. To this end, Ambu and ENT UK will team-up on a dedicated discussion group at the ENT UK Innovation Forum in October 2026. The group's focus will be on exploring the design, implementation, and expansion of community ENT models.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients