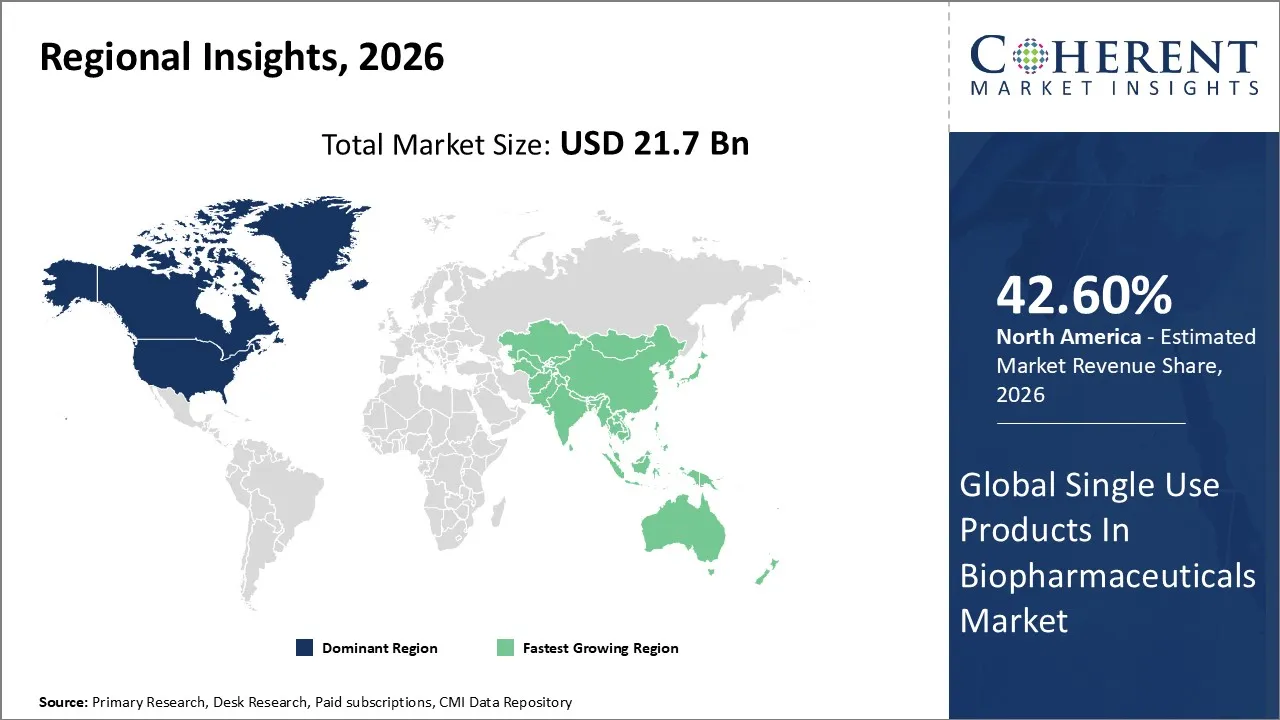
The Single Use Products in Biopharmaceuticals Market is estimated to be valued at USD 21.7 Bn in 2026 and is expected to reach USD 34.4 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 11.7% from 2026 to 2033.
The Single Use Products in Biopharmaceuticals Market is evolving rapidly as biopharmaceutical companies increasingly demand flexible, efficient, and contamination‑free manufacturing solutions. Companies are adopting single‑use technologies such as disposable bioreactors, tubing, filters, and sensors for biologics, vaccines, cell and gene therapies, and personalized medicines. These technologies reduce cleaning, sterilization, and setup time, boosting operational efficiency and enabling small‑batch, multiproduct workflows. Ongoing R&D, supportive regulations, and the push for faster time‑to‑market continue to drive market expansion.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Advancements and Innovation |
|
|
Regional Industry Growth Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Pharmaceutical Companies acquired the largest market share of 53.1% in 2026. Pharmaceutical companies are driving the adoption of single-use products in biopharmaceuticals to boost production efficiency and maintain high-quality, contamination-free operations. They implement disposable systems such as bioreactors, filters, tubing, and sensors to speed up setup, minimize cleaning and validation, and enable flexible small-batch or multiproduct manufacturing. Companies use single-use technologies to accelerate drug development, adapt to emerging biologics and personalized therapies, and meet regulatory standards. Ongoing innovations and the integration of advanced monitoring tools continue to encourage their use in agile, modern biomanufacturing. For instance, in April 2025, Parker Bioscience Filtration has launched the SciLog NFF+ PF, an automated Normal Flow Filtration system that streamlines Pre-Use Post-Sterilisation Integrity Testing in biopharmaceutical production
Work Equipment hold the largest market share of 52.3% in 2026. Manufacturers are driving the adoption of work equipment in the Single Use Products in Biopharmaceuticals Market to achieve streamlined, contamination-free production. They deploy single-use bioreactors, mixers, filtration units, and disposable sensors to increase operational flexibility and minimize downtime by removing the need for extensive cleaning and sterilization. Companies rely on these systems for small-batch and multi-product production, enhanced sterility, and easy integration into modular facilities. Ongoing advances in materials and monitoring technologies further boost efficiency and reliability, positioning single-use work equipment as a key choice in modern biomanufacturing. For instance, ABEC has introduced a single-use bioreactor system with a 6,000 L working volume, tripling the industry’s typical maximum. While single-use bioreactors are now common, most vendors cap at 2,000 L, forcing manufacturers to operate multiple units simultaneously or switch to stainless-steel tanks for larger production needs.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 42.6% in 2026. North America drives strong growth in the Single Use Products in Biopharmaceuticals Market through several emerging trends. Companies across the region actively adopt disposable bioprocessing systems, supported by advanced biopharmaceutical infrastructure and substantial R&D investments. Manufacturers integrate single-use technologies into both upstream and downstream operations for biologics and vaccine production. Regulatory bodies encourage innovation, while firms expand automation and flexible manufacturing platforms. Growing contract manufacturing activities and increasing demand for efficient, contamination-free workflows further strengthen the market across the U.S., Canada, and Mexico. For instance, in July 2025, Fresenius Kabi is expanding its U.S. biosimilars portfolio by introducing Conexxence (denosumab-bnht) and Bomyntra® (denosumab-bnht). Bomyntra, available as a 120 mg/1.7 mL vial and single-use prefilled syringe, is FDA approved for all indications of the reference product Xgeva® (denosumab).
Asia Pacific is advancing the Single Use Products in Biopharmaceuticals Market through dynamic regional trends. Biopharmaceutical manufacturers in China, India, Japan, and Southeast Asia are accelerating the adoption of disposable bioprocessing systems to produce biologics, vaccines, and advanced therapies. Companies are expanding local manufacturing capacity to meet growing healthcare demand while prioritizing modular and scalable solutions. They are also integrating automation and digital technologies to enhance productivity. Increasing outsourcing activities and stronger focus on sustainability further drive the adoption of single-use technologies across the region. For instance, in December 2025, the Government of Telangana inaugurated Telangana 1 Bio, India’s first single-use bioprocess design and scale-up facility in Genome Valley, to strengthen the country’s biologics and next-generation therapeutics capabilities.
Manufacturers in the United States Single Use Products in Biopharmaceuticals Market are actively prioritizing disposable bioprocessing systems to produce biologics, vaccines, and advanced therapies, moving away from traditional equipment toward more flexible and efficient solutions. U.S. companies are advancing their single-use portfolios by developing innovative bioreactors, mixers, fluid assemblies, and other disposable components to address evolving manufacturing requirements. Strong biopharmaceutical demand, greater adoption of single-use consumables in upstream processes, and expanded use of modular and automated systems continue to drive market progress. For instance, in August 2025, Accord BioPharma, the specialty division of Intas Pharmaceuticals, launched IMULDOSA (ustekinumab-srlf), a biosimilar to STELARA.
Manufacturers are driving growth in India’s Single Use Products in Biopharmaceuticals Market by integrating disposable bioprocessing systems to enhance production efficiency and address rising demand for biologics and vaccines. Companies in key hubs such as Delhi, Mumbai, Bengaluru, and Hyderabad are adopting modular, flexible solutions to support small-batch and advanced therapy manufacturing while minimizing contamination risks and costs. Firms are also increasing the use of single-use sensors and probes for real-time monitoring, and expanding partnerships with CMOs and CROs to accelerate technology adoption nationwide. For instance, Aligning with the ‘Make in India’ initiative, the government-backed Mumbai-based company launched its first integrated manufacturing unit at PharmNXT Biotech LLP in Chakan, Pune, to provide single-use bioprocess solutions and tackle challenges in biologics production.
The market is shifting toward flexible, modular manufacturing platforms that integrate single-use systems across multiple bioprocessing stages. Manufacturers are designing facilities that accommodate rapid changeovers, support multi-product workflows, and reduce dependency on fixed stainless-steel infrastructure. This flexibility helps companies adapt quickly to fluctuating demand and streamline facility utilization while maintaining high quality and sterility standards in biologics and advanced therapy production.
Biopharmaceutical companies are increasingly embedding digital technologies and automation into single-use workflows to enhance process control and data integrity. Real-time monitoring, advanced sensors, and automated control systems improve consistency, reduce manual intervention, and enable predictive maintenance. These trends support higher productivity, facilitate regulatory compliance, and drive broader acceptance of single-use solutions in both upstream and downstream processes.
Contract development and manufacturing organizations (CDMOs) and contract manufacturing organizations (CMOs) present a major opportunity for single‑use product suppliers. Outsourcing continues to increase as pharmaceutical firms seek capacity without heavy capital investment. By providing ready‑to‑install single‑use systems and comprehensive integration support, suppliers can attract partners needing rapid deployment, flexible production lines, and efficient multiproduct manufacturing, strengthening their foothold across global biomanufacturing networks.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.7 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.7% | 2033 Value Projection: | USD 34.4 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Aber instruments, Adolf Kühner AG, Thermo Fisher Scientific, Bio-equip, Pall corp., C-Cit Sensors AG, Corning Life Sciences, Eppendorf, GE Healthcare, Gemu GmbH, Merck, Avantor, Meissner, New Horizon Biotech, PBS-Biotech, Xcellerex, and Single Use Support GmbH. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients