The global skin fibrosis treatment market size is estimated to reach USD 796 Mn in 2026 and is projected to grow at a compound annual growth rate (CAGR) of 6.9% during the forecast period (2026-2033), surpassing USD 1,267 Mn by 2033. This is attributable to rising prevalence of skin disorders and fibrosis-related complications, along with growing demand for anti-fibrotic therapies for skin.
The global skin fibrosis treatment market is projected to grow steadily during the forecast period. This is mostly due to increasing prevalence of skin fibrosis disorders, rising awareness about early diagnosis and treatment, and growing adoption of advanced therapeutic options. In addition, the expansion of healthcare infrastructure, shift towards non-invasive skin fibrosis therapies, and rising geriatric population are supporting market expansion.
Skin fibrosis is a critical clinical condition that can lead to serious disability and increased morbidity. It occurs in both skin-restricted and systemic disorders, including localized scleroderma, systemic sclerosis (SSc), and chronic graft-versus-host disease. Fibrosis often develops following epidermal or dermal damage caused by trauma, chemical exposure, or immune-mediated processes. It is frequently associated with chronic inflammation and is characterized by an increase in fibrous connective tissue in the dermis or subcutis.
Increasing awareness of rare conditions such as localized scleroderma, eosinophilic fasciitis, scleromyxedema, and scleredema is driving demand for skin fibrosis treatments. For example, systemic sclerosis, a major form of skin fibrosis, has a global prevalence of approximately 18.9 per 100,000 persons and an incidence of about 8.6 per 100,000 person-years, highlighting its clinical significance. These disorders are typically managed with corticosteroids, antifibrotic agents, immunotherapy drugs, and other immunosuppressants.
|
Current Event |
Description and its Impact |
|
Advances in Gene Therapy and CRISPR Technology |
|
|
Artificial Intelligence Integration in Drug Discovery |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As per CMI’s latest skin fibrosis market outlook, the corticosteroids segment is set to account for a revenue share of 36% in 2026. Corticosteroids remain the most commonly used drug type for the treatment of skin fibrosis because of their strong anti-inflammatory properties and ability to reduce fibroblast activation. They are particularly effective in controlling disease progression and minimizing skin thickening in both localized and systemic forms of fibrosis.
Topical or intralesional corticosteroids, such as prednisone or triamcinolone, are often considered first-line therapy for localized fibrotic conditions like hypertrophic scars, keloids, morphea, and lichen sclerosus. The use of corticosteroids depends on how severe and widespread the condition is. Topical or intralesional treatments are preferred for localized lesions, while systemic use is reserved for more severe or widespread disease.
Based on distribution channel, hospital pharmacies are expected to dominate the global skin fibrosis treatment market, accounting for a share of 40% in 2026. This is mostly due to their direct access to patients with severe or chronic conditions, higher prescription rates from dermatologists and rheumatologists, and the ability to dispense specialized or costly antifibrotic medications that are often not available through retail or online pharmacies.
In addition, hospitals provide integrated care and monitoring for patients undergoing complex treatment regimens, further reinforcing their leading role in the distribution of skin fibrosis therapies. On the other hand, online pharmacies segment is anticipated to grow at a higher CAGR during the assessment period, owing to increasing adoption of e-pharmacy platforms, convenience of home delivery, and growing patient preference for discreet and accessible medication procurement.
To learn more about this report, Request Free Sample
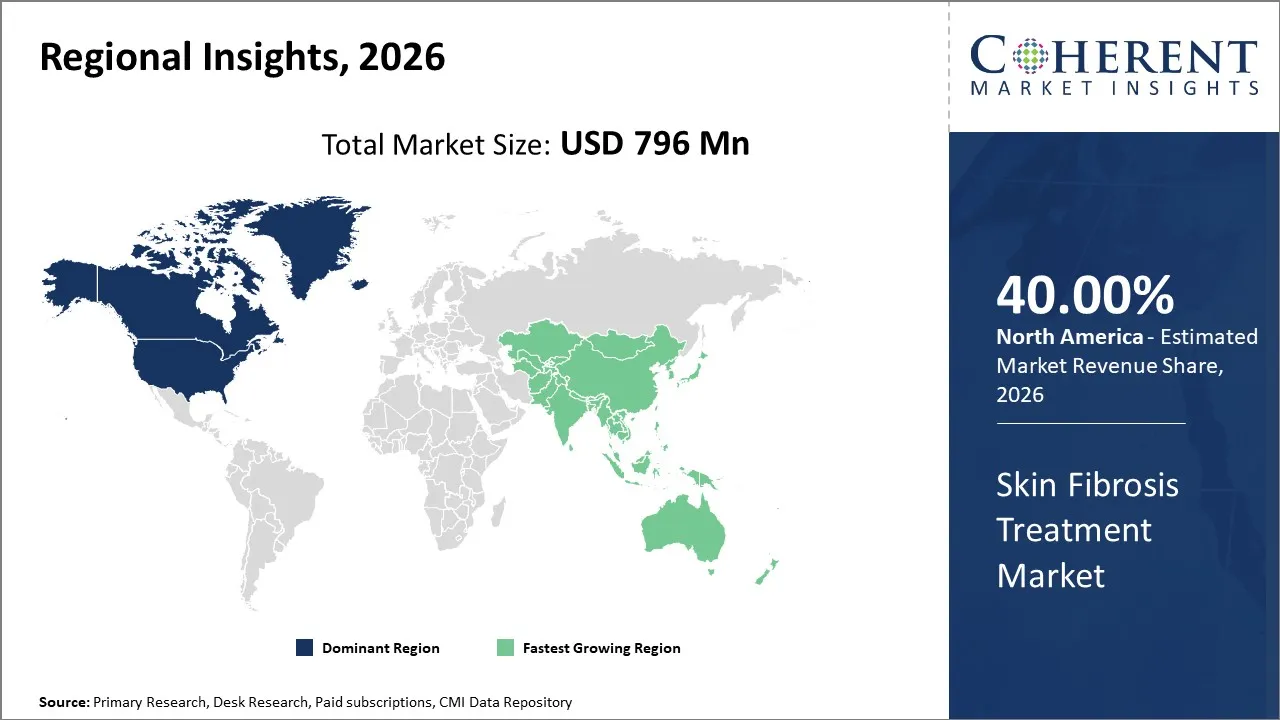
North America is projected to lead the global skin fibrosis treatment market, accounting for approximately 40% of the market share in 2026. This is mostly due to high patient awareness, widespread adoption of advanced treatment options, robust healthcare infrastructure, and strong presence of leading pharmaceutical companies.
Rising prevalence of systemic sclerosis, chronic graft-versus-host disease, and other fibrotic skin conditions is driving demand for effective therapies. The region also gains from strong investment in research and development, which has led to new treatments like antifibrotic drugs, targeted biologics, and combination therapies that improve patient outcomes and make treatments more accessible.
Growing interest in regenerative medicine is also increasing the skin fibrosis treatment market value in North America. Therapies such as stem cell treatments, platelet‑rich plasma (PRP), and other biologic approaches for tissue repair and remodeling are attracting attention as alternatives or additions to conventional therapies.
Asia Pacific is set to become a highly profitable market for manufacturers of skin fibrosis drugs during the forecast period. This is mainly because of increasing disease awareness, expanding healthcare infrastructure, and improving access to specialist care. The rising prevalence of skin-restricted and systemic fibrotic disorders in countries like Japan, China, and India is encouraging both public and private sectors to invest in early detection and management programs.
In addition, collaborations between local hospitals and global pharmaceutical companies are facilitating clinical trials, training programs, and introduction of advanced antifibrotic therapies. As a result, Asia Pacific, led by China and India, is anticipated to hold 22% of the global skin fibrosis industry share in 2026.
The United States skin fibrosis treatment industry is expected to grow steadily due to increasing patient awareness, robust healthcare spending, and the rapid adoption of advanced therapeutics. A major driver in this market is the high focus on research and development of novel antifibrotic drugs and biologics.
Continuous R&D investment has led to the approval of innovative therapies that target the underlying mechanisms of fibrosis, improving efficacy and safety, and thereby driving widespread clinical adoption and market expansion. Emerging trends include precision medicine approaches that utilize biomarkers and genetic profiles for patient stratification and tailored therapeutic regimens.
The skin fibrosis treatment industry is poised to exhibit strong growth during the assessment period. This is mostly due to increasing recognition of systemic sclerosis, growing patient awareness, and expansion of healthcare infrastructure. Among these, the most significant driver is the rising investment in specialized dermatology and rheumatology centers, which improves early diagnosis and access to advanced therapies. These centers facilitate timely intervention with antifibrotic medications and biologics, enhancing treatment outcomes and supporting wider adoption of innovative therapies across the country.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 796 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.9% | 2033 Value Projection: | USD 1,267 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pharmaxis, Jubilant Cadista, Horizon Pharma USA, Pfizer, Sanofi, F. Hoffmann-La Roche, Boehringer Ingelheim, Bristol-Myers Squibb Company |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising Popularity of Combination Treatments: Emerging multimodal strategies that combine anti-fibrotic agents with immunomodulators or advanced drug delivery systems have shown promise in improving patient outcomes. Ongoing research in these combination approaches is expected to drive innovation and create lucrative growth opportunities for the skin fibrosis market during the assessment period.
Advancements in R&D and Clinical Trials: Increasing research into novel fibrosis pathways, regenerative therapies, and immunomodulators is expanding the therapeutic pipeline. Although many therapies are in preclinical or early clinical stages, these developments are expected to fuel market growth in the coming years.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients