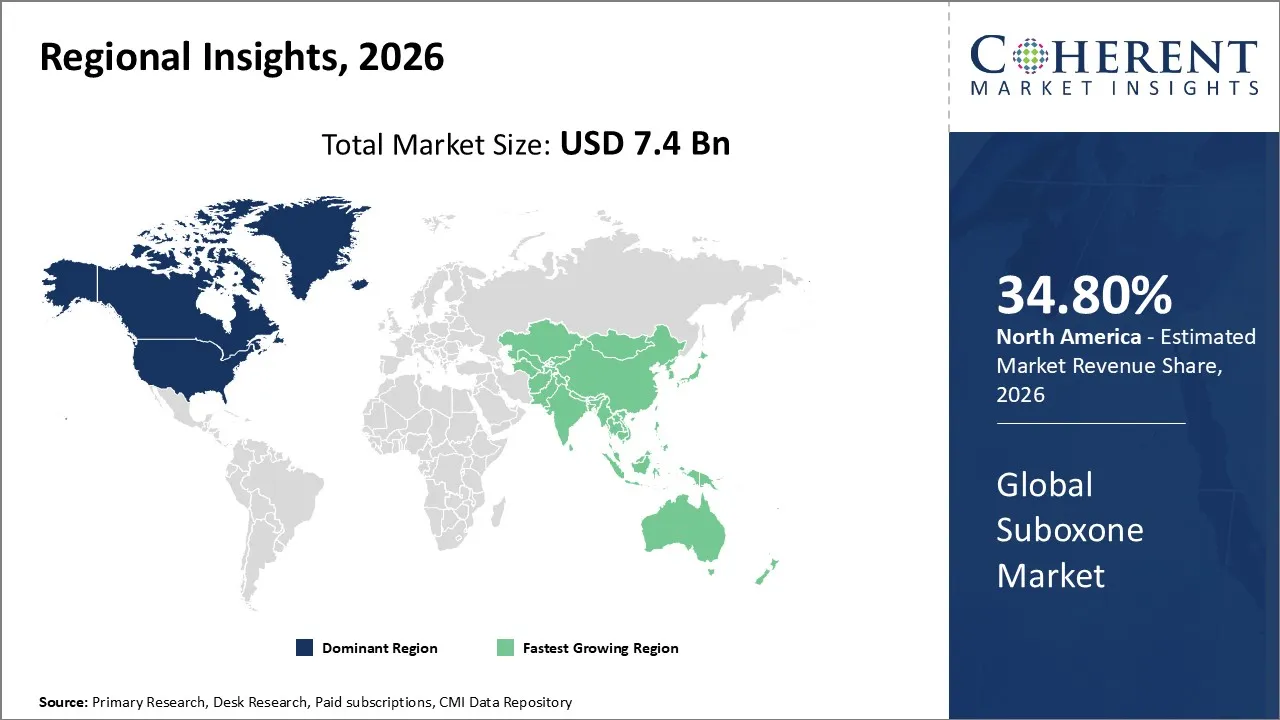
The Suboxone Market is estimated to be valued at USD 7.4 Bn in 2026 and is expected to reach USD 9.6 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.1% from 2026 to 2033.
Healthcare systems are driving the Suboxone market by increasing the adoption of medication-assisted therapy and addressing the rising need for effective treatments for opioid use disorder. Suboxone, which combines buprenorphine and naloxone, reduces withdrawal symptoms and helps prevent opioid misuse, leading addiction treatment programs to use it widely. Governments and healthcare organizations are raising awareness of substance use disorders and expanding access to treatment centers, which strengthens market demand. Furthermore, pharmaceutical companies are introducing generic formulations and expanding distribution through hospital and retail pharmacies, supporting the global growth of the market.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Changes in Key Markets |
|
|
Opioid Epidemic Trends and Public Health Initiatives |
|
|
Technological Innovations in Treatment and Monitoring |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Generics hold the largest market share of 53.3% in 2026. Patent expirations in the Suboxone market have allowed multiple manufacturers to enter, driving down treatment costs and fueling the growth of generics. Healthcare providers and payers increasingly choose generics for their affordability and accessibility. The rising prevalence of opioid use disorder and the expansion of medication-assisted treatment programs increase demand, while government initiatives and insurance coverage actively promote generic adoption. Moreover, manufacturers leverage established production capabilities and broad distribution networks to make generic Suboxone widely available, enhancing its use in hospitals, clinics, and retail pharmacies.
Tablets expected to hold largest market share of 52.2% in 2026. Healthcare providers and treatment centers drive the demand for Suboxone tablets due to their cost-effectiveness, wide availability, and simpler administration compared to film formulations. Multiple generic tablet options increase accessibility and affordability, encouraging hospitals and outpatient clinics to adopt them. As medication-assisted treatment programs expand and awareness of opioid use disorder grows, clinicians actively utilize tablets, making them a practical and reliable option for managing opioid dependence over the long term. For instance, India-based Dr. Reddy’s has launched generic omeprazole delayed-release tablets for heartburn and resumed its generic Suboxone (buprenorphine/naloxone) in the U.S. after the injunction was lifted.
Hospital Pharmacies acquired the prominent market share of 36.7% in 2026. Hospital pharmacies drive the Suboxone market by providing timely access to buprenorphine/naloxone for inpatient and emergency care. Hospitals actively initiate medication-assisted treatment for patients with opioid use disorder, increasing demand through their pharmacies. By integrating addiction services into hospital care and leveraging supportive government policies and opioid crisis funding, they strengthen their role in treatment. Additionally, hospital pharmacies supply initial doses and coordinate with outpatient programs, ensuring continuity of care and positioning themselves at the center of Suboxone distribution and patient management.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 34.8% in 2026. Healthcare providers are driving growth in the North America Suboxone market by increasingly adopting medication-assisted treatment for opioid use disorder, supported by rising awareness and government initiatives. Hospitals, clinics, and outpatient programs actively integrate buprenorphine/naloxone therapy, while telemedicine expands patient access. Manufacturers and pharmacies enhance affordability and reach by offering generic formulations. Furthermore, policymakers and funding programs addressing the opioid crisis influence prescribing practices, actively boosting demand for Suboxone throughout the region.
Healthcare systems across the Asia Pacific region are driving the Suboxone market by strengthening addiction treatment services and increasing awareness of opioid use disorder. Hospitals, clinics, and rehabilitation centers actively adopt buprenorphine/naloxone therapy, while government initiatives and public health programs expand access. Manufacturers and pharmacies promote adoption by supplying affordable generic formulations to both urban and rural areas. Additionally, investments in healthcare infrastructure, training of addiction specialists, and integration of medication-assisted treatment into outpatient and community programs are actively boosting Suboxone use throughout the region.
Healthcare providers are driving the United States Suboxone market by increasing awareness of opioid use disorder and integrating medication-assisted treatment into hospitals, clinics, and outpatient programs. They actively prescribe buprenorphine/naloxone to manage withdrawal symptoms and prevent relapse, while telemedicine expands patient access in remote areas. Manufacturers and pharmacies enhance adoption by offering affordable generic formulations. Furthermore, federal and state initiatives, along with opioid crisis funding, shape treatment protocols and actively reinforce Suboxone’s role in addressing opioid dependence nationwide. For instance, Mylan N.V. announced the U.S. launch of its Buprenorphine and Naloxone Sublingual Film in 8/2 mg and 12/3 mg strengths, a generic version of Indivior's Suboxone Sublingual Film.
Healthcare authorities in China are driving the Suboxone market by expanding efforts to address opioid dependence and strengthen addiction treatment services. Hospitals, clinics, and rehabilitation centers actively adopt buprenorphine/naloxone therapy, supported by government initiatives and pilot programs targeting substance use disorders. Manufacturers and pharmacies improve accessibility by supplying affordable generic formulations, overcoming historical barriers of limited awareness and high costs. Additionally, investments in training addiction specialists, establishing outpatient programs, and integrating medication-assisted treatment into community healthcare are actively boosting Suboxone use nationwide.
Healthcare providers increasingly integrate Suboxone into treatment protocols for opioid use disorder. Hospitals, outpatient clinics, and rehabilitation centers are actively initiating MAT programs to manage withdrawal symptoms and prevent relapse. Telemedicine is expanding access, allowing remote prescribing and follow-up care. Patient awareness campaigns and educational initiatives by healthcare authorities encourage acceptance of MAT, driving broader utilization. These efforts position Suboxone as a cornerstone therapy in both acute care and long-term addiction management.
The entry of multiple generic buprenorphine/naloxone products is reshaping the Suboxone market. Generics improve affordability and accessibility for patients, especially in public health programs and community clinics. Pharmacies and hospitals increasingly stock generics, creating price competition and widening patient reach. The availability of lower-cost alternatives enables healthcare systems to expand treatment programs and reduce barriers to care, strengthening Suboxone adoption across urban and rural regions.
The increasing availability of generic buprenorphine/naloxone creates an opportunity to make treatment more affordable and accessible. Healthcare providers, hospitals, and pharmacies can adopt generics to reduce costs, improve patient adherence, and expand coverage in public health programs. Manufacturers can leverage competitive pricing, distribution networks, and partnerships with government-funded addiction programs to strengthen market penetration, particularly in regions with cost-sensitive patient populations.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7.4 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.1% | 2033 Value Projection: | USD 9.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Indivior PLC, Dr. Reddy’s Laboratories Ltd., Amneal Pharmaceuticals LLC., Mylan N.V., Novartis AG, Teva Pharmaceutical Industries Ltd., Mallinckrodt, Lannett Co Inc., Rhodes Pharmaceuticals L.P., Alvogen, Sun Pharmaceutical Industries Ltd., Alkem Labs, and Hikma Pharmaceuticals PLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients