Global Triptorelin Market – Global Industry Insights
Gonadotropin-releasing hormone (GnRH) agonists have greater safety and efficacy profile than estrogens and anti-androgens among various therapies of achieving medical castration. Furthermore, slow-release formulations of GnRH agonists offer patients flexibility, improves quality of life, and eventually reduce cost.
Most of the times, prostate cancer progresses with increasing testosterone hormone. Moreover, the production of testosterone can be stopped by surgically removing the testicles or thorough medication therapy. Triptorelin is GnRH agonists which is designed to stop the tentacles from making testosterone. This eventually reduces testosterone levels and minimizes progression of prostate cancer.
Triptorelin is a decapeptide analogue of Luteinising Hormone-Releasing Hormone (LHRH, also called gonadotropin-releasing hormone [GnRH]). Triptorelin is extensively used in palliative treatment of hormone dependent advanced prostate cancer.
Moreover, according to National Institute for Health and Care Excellence (NICE), in May 2013, the licensed indications for luteinising hormone-releasing hormone (LHRH) agonist: triptorelin were extended to include two new indications for prostate cancer: first in neoadjuvant treatment before radiotherapy in high-risk localized or locally advanced disease and second in adjuvant treatment to radical prostatectomy in locally advanced disease at high risk of progression. Triptorelin is administrated intramuscularly or subcutaneously.
Furthermore, triptorelin is indicated for the treatment of pediatric patients two years of age and older with Central Precocious Puberty (CPP). In July 2017, U.S. Food and Drug Administration (FDA), approved Arbor Pharmaceuticals’: Triptodur, in pediatric patients with central precocious puberty. The approval follows a phase 3 clinical trial (Efficacy, Safety, and Pharmacokinetics [PK] of Triptorelin 6-month Formulation in Patients with Central Precocious Puberty), which examines prepubertal luteinizing hormone levels using triporelin therapy. Study demonstrated a return to pre-pubertal luteinizing hormone (LH) levels in 93% of enrolled patients, with pre-pubertal luteinizing hormone (LH) suppression maintained at 12 months by 98% of patients.
The global triptorelin market size was valued at US$ 426.4 Mn in 2017, and is expected to witness a CAGR of 5.2% over the forecast period (2018 – 2026).
Global Triptorelin Market Share, By Product Type: 2018 & 2026
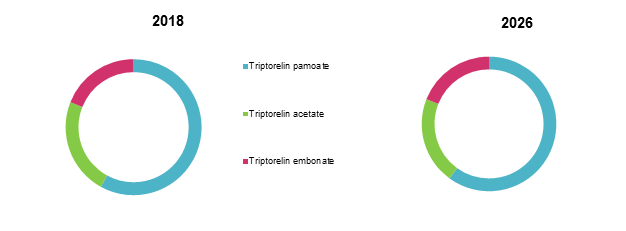
To learn more about this report, Request Free Sample
Source: Coherent Market Insights Analysis (2018)
Extended Application of Triptorelin in Various Treatment Modalities is expected to Drive Market Growth during the Forecast Period
The GnRH agonist, triptorelin is a first-line hormonal therapy, which offers efficacy and safety in clinical trials of patients with non-metastatic or metastatic prostate cancer. Furthermore, triptorelin has been increasingly studied for varied treatment options, present in clinical development phases. For instance, GnRH analogue, triptorelin is being studied to analyze their efficacy and safety for HIV-1 reservoir reduction in Antiretroviral Therapy (ART) treated HIV-1 infected patients. According to ClinicalTrials.gov, in May 2018, this study was conducted by Immune System Regulation AB, and currently in phase II clinical stage.
Triptorelin is further being studied to analyze effects of adjuvant hormonal therapy treatments of Tamoxifen, Letrozole and Letrozole + Zoledronate on bone loss in breast cancer patients. According to ClinicalTrials.gov, in July 2017, this study was sponsored by National Cancer Institute (NCI), Naples; and currently present in phase III clinical stage. Moreover, triptorelin market shows lucrative revenue growth in the near future, with the successful completion of this study and potential launch of new products during the forecast period.
Moreover, higher efficacy and effectiveness of triptorelin in the suppression of gonadotropin in Central Precocious Puberty (CPP) among young boys and girls is expected to drive the triptorelin market growth. For instance, according to data published in the Scientific World Journal, in May 2012, triptorelin 11.25 mg per 90 days efficiently suppressed the pituitary-gonadal level in children with CPP from first administration.
Furthermore, increasing research and development activities focused on application of androgen deprivation therapy with triptorelin to lower progression of salivary gland cancer expressing androgen receptors. For instance, in January 2016, Debiopharm International SA collaborated with European Organization for Research and Treatment of Cancer (EORTC) to investigate Triptorelin for treatment of salivary gland cancer. Under the collaboration, Debiopharm International SA will supply triptorelin pamoate 3.75 mg 1-month formulation to patients suffering from salivary gland cancer participating in a clinical study sponsored by EORTC. During the clinical study, researchers will evaluate the efficacy and safety of androgen chemotherapy versusandrogen deprivation therapy (ADT) in patients with recurrent and/or metastatic androgen receptors-expressing salivary gland cancers.
Global Triptorelin Market Share, By Application: 2018 & 2026
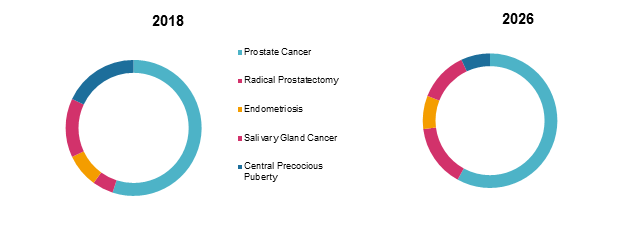
To learn more about this report, Request Free Sample
Source: Coherent Market Insights Analysis (2018)
Increasing Prevalence of Prostate Cancer among Global Population is expected to Aid in Market Growth
Incensing prevalence of prostate cancer worldwide and wide applications of triptorelin in prostate cancer treatment to limit the cancer progression by reducing the testosterone synthesized by body is expected to drive growth of triptorelin market during the forecast period.
For instance, according to the World Cancer Research Fund (WCRF) International, 2012, over 1.1 million cases of prostate cancer were recorded, accounting for around 8% of all new cancer cases worldwide. Furthermore, around 68% of prostate cancer cases were registered in the developed economies. Highest incidence of prostate cancer was reported in Oceania and North America, while lowest incidence was reported in Asia and Africa.
Moreover, according to the Cancer Research U.K., in 2015, around 47,151 new cases of prostate cancer were registered in the U.K. Furthermore, prostate cancer accounted for 13% of the total cancer cases in the U.K., in 2015. Prostate cancer is the second-most common cause of cancer deaths among males in the U.K. in 2014, accounting for 13% of all cancer deaths among males. In addition, according to the same source, in 2014, around 11,287 prostate cancer deaths were recorded among males in the U.K. The crude mortality rate shows that there were 36 prostate cancer deaths for every 100,000 males in the country.
Key players operating in triptorelin market include Ipsen, Bachem, Ferring Pharmaceuticals Pvt Ltd., Chengdu Tiantaishan pharmaceutical Co., Ltd., Tecnofarma, Dr. Reddy’s Laboratories Ltd., Teva Pharmaceutical Industries Ltd. (Actavis Specialty Pharmaceuticals Co.), Debiopharm Group, and Arbor Pharmaceuticals, LLC.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients