The Vaginal Fungi Infections Treatment Market is anticipated to grow at a CAGR of 5.4% with USD 1,347.5 Mn in 2026 and is expected to reach USD 1,935.9 Mn in 2033. The Vaginal Fungi Infections Treatment Market is expanding steadily on the back of a high and persistent global disease burden, particularly vulvovaginal candidiasis, which affects an estimated 75% of women at least once in their lifetime, predominantly caused by Candida albicans in nearly 80–90% of cases, underscoring recurrent therapy demand, as reported in a 2026 study by the Springer Nature. In 2026, the market size is supported by strong outpatient demand across gynecology clinics, retail and online pharmacies, and adoption of both azole and topical antifungal therapies. North America remains a leading regional contributor, reflecting high infection prevalence, robust healthcare infrastructure and widespread clinician awareness. Market growth is propelled by rising awareness campaigns, improved diagnostics, and expanding treatment options, although antifungal resistance and OTC access challenges persist.
Source: International Journal of Medical and Pharmaceutical Research; CDC
Fluconazole is projected to account for the largest share of cognitive systems spending in 2026, representing approximately 50.0% of the total volume. The Fluconazole drug‑type segment is dominant in the vaginal fungi infection treatment market due to its established clinical efficacy, ease of administration, cost‑effectiveness, and guideline support. Oral Fluconazole (single 150 mg dose) is widely recommended as a first‑line treatment for uncomplicated vulvovaginal candidiasis, achieving >90% clinical cure rates and faster symptom relief compared with many topical therapies, according to 2026 treatment recommendations by the Dr. Oracle. A 2026 comparative evaluation using the WHO’s rational drug selection approach identified Fluconazole as the top P‑drug based on efficacy, safety, cost, and convenience versus other azoles, reinforcing its preferred status in routine care. Additionally, Fluconazole is FDA‑approved for vulvovaginal candidiasis, supporting widespread adoption in clinical practice.
Based on route of administration, oral dominate the market, accounting for a significant 55.0% share in 2026, due to high bioavailability, convenience, and proven clinical performance. A 2026 review in MDPI noted that oral fluconazole remains the only systemic azole routinely licensed for VVC, with a single 150 mg dose often achieving rapid clinical response and extensive patient acceptance. Oral formulations like fluconazole and newer agents such as ibrexafungerp approved by the FDA provide effective systemic drug levels that are not affected by local vaginal conditions, simplifying adherence compared with multi‑day topical regimens. Clinical data also show oral antifungals deliver consistently high cure rates and are preferred by patients for ease of use and reduced dosing complexity, reinforcing their leading role in treatment practice.
Oral antifungals such as fluconazole, recommended as standard induction therapy in vulvovaginal candidiasis regimens, often consist of single or short‑course doses that improve compliance and clinical outcomes versus prolonged intravaginal applications. Clinical studies show novel oral agents like ibrexafungerp deliver clinical cure rates over 65% in acute and recurrent cases and reduce recurrence when dosed monthly, demonstrating robust systemic effectiveness and tolerability. Oral formulations also bypass variable mucosal absorption limitations seen with inserts or creams, offering predictable pharmacokinetics and broader activity factors repeatedly highlighted in 2026 peer‑reviewed therapeutic reviews and clinical trial results published on National Library of Medicine.
To learn more about this report, Request Free Sample
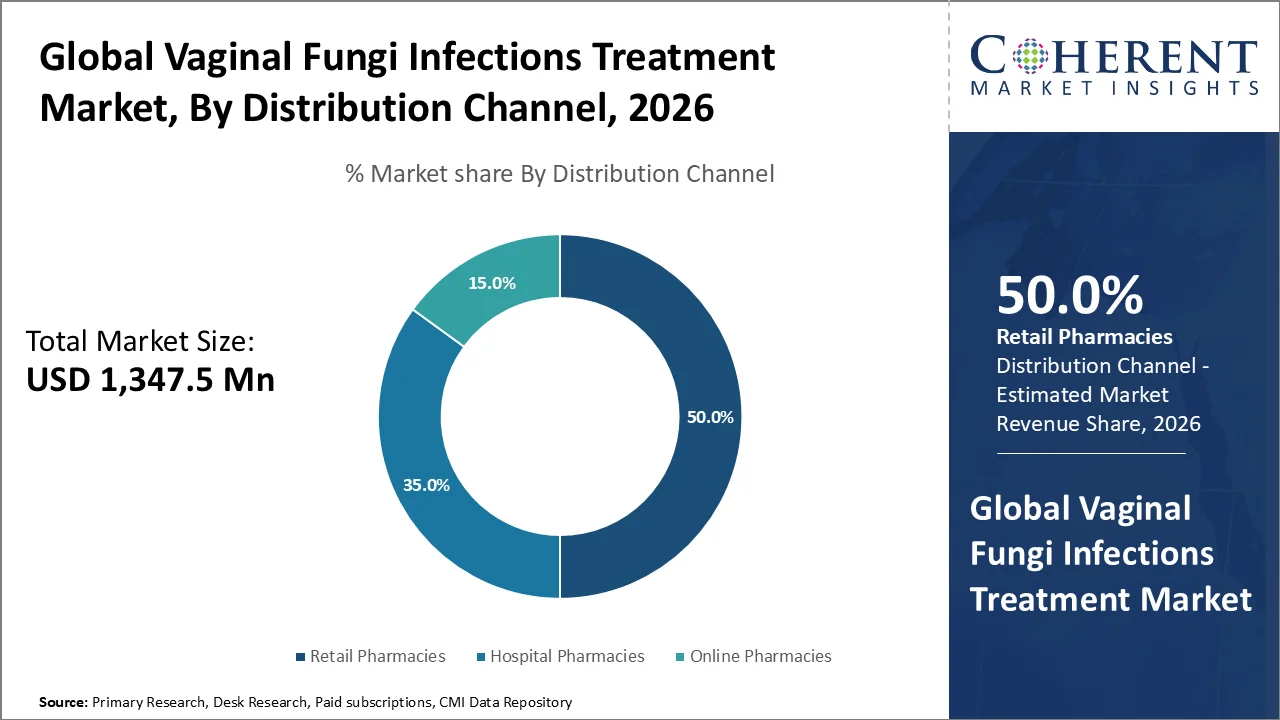
Retail Pharmacies account for the largest share of 50.0% in 2026 as the retail pharmacy outlets offer easy access to both prescription and OTC antifungal medications for outpatient use, enhanced patient education, and pharmacist counselling that encourages proper self‑care and adherence. In the vaginal fungi infection treatment market, the retail pharmacies segment dominates because it provides broad access to both OTC and prescription antifungal therapies, allowing women to conveniently obtain treatments like clotrimazole and miconazole creams or oral fluconazole at local drugstores without a hospital visit. For instance, in 2025, according to the WHO guidance notes topical and oral antifungals are standard first‑line treatments for vulvovaginal candidiasis, and CDC data emphasize that many topical treatments are available OTC, which boosts pharmacy sales and self‑care adoption (2024). Moreover, CDC data show that up to 75% of women experience at least one episode of vulvovaginal candidiasis in their lifetime (lifetime prevalence) and many seek immediate OTC relief (2021 guidance cited by CDC). The availability of topical creams and oral antifungals at retail pharmacies enhances convenience, supports pharmacist counselling, and boosts adherence to treatment, reinforcing pharmacy preference among consumers.
Rising prevalence of vaginal fungi infection has driven the vaginal fungi infections treatment market growth over the forecast period. The increasing incidence of vaginal fungi infections is a key growth driver for the Vaginal Fungi Infection Treatment Market because as more women experience infections like vulvovaginal candidiasis, the demand for effective treatment increases both for initial therapy and for recurrent cases. A 2026 systematic analysis by the National Center for Biotechnology Information (NCBI) estimated that about 32.6% of women globally are affected by vulvovaginal candidiasis, with Candida albicans being the predominant pathogen, reinforcing the ongoing clinical need for antifungal treatments. This high prevalence and recurrence rate naturally expand outpatient visits, physician prescriptions, over‑the‑counter therapy usage, and patient reliance on pharmaceutical options, driving market growth in pharmaceuticals and related healthcare services.
Increasing product approval and launches for treatment of vaginal fungi infection has driven the vaginal fungi infections treatment market growth over the forecast period. Product approvals and launches are driving vaginal fungi infection treatment market growth by expanding therapeutic options, addressing unmet needs, and stimulating clinician and patient uptake. For instance, in March 2026, according to data published by Gedea Biotech, a Swedish women's health company, the launch of pHyph, a promising new antifungal therapy for the treatment of vaginal fungi infections, marks a significant milestone in the vaginal fungi infection treatment market. The drug, which has received regulatory approval in Sweden, is designed to address vulvovaginal candidiasis, one of the most common forms of vaginal fungal infection caused primarily by Candida albicans. pHyph stands out for its dual-benefit effects, which target both the infection and inflammation associated with these conditions, offering a new solution beyond the traditional azole-based treatments like fluconazole. This launch is expected to broaden treatment options for both healthcare providers and patients, addressing an unmet need in the market for more effective, localized therapies with fewer side effects. Gedea Biotech's strategic focus on expanding the scope of antifungal therapies is poised to further enhance market growth, with additional clinical studies set to evaluate pHyph’s effectiveness in various populations. As of March 2026, clinical evaluations for this new treatment are well underway.
Innovation in vaginal fungi infection therapies is expanding treatment options and driving market growth by addressing resistance and recurrence. According to the Vulvovaginal Candidiasis – Pipeline Insight, 2026 report, more than 7 novel antifungal candidates are in development, including next‑generation systemic and localized drugs targeting resistant Candida strains, broadening clinical choice beyond traditional azoles. Recent research published in 2026 has also highlighted advanced treatment modalities such as self‑healing hydrogels for personalized drug delivery, offering improved symptom relief and retention in the vaginal mucosa compared with conventional forms. These breakthroughs are under active investigation to enhance efficacy and patient adherence, reflecting a paradigm shift in therapeutic design and delivery technologies. The focus on innovative formulations and pipeline drugs strongly supports future market expansion by offering differentiated solutions for recurring and resistant infections, setting new standards in women’s health care.
For instance, in January 2026, SCYNEXIS, Inc., biotechnology company announced that the U.S. Food and Drug Administration (FDA) granted Qualified Infectious Disease Product (QIDP) and Fast Track designations to its second‑generation antifungal therapy SCY‑247 for difficult‑to‑treat and recurrent fungal infections, including potential applications in vulvovaginal candidiasis (VVC). This follows the successful approval and ongoing relaunch efforts of BREXAFEMME (ibrexafungerp tablets) to treat VVC and reduce recurrent VVC, highlighting innovation beyond traditional azoles and strengthening treatment options in 2026. The designations aim to accelerate development timelines and address unmet needs amid rising antifungal resistance, signaling robust momentum in new drug development for vaginal fungal conditions in the 2026.
|
Current Event |
Description and its Impact |
|
Increasing Regulatory Approval |
|
|
Launch of Over-the-Counter Antifungal Treatments for Vaginal Fungal Infections |
|
|
Rising Government Initiatives & Government Funding Activity for Antifungal Resistance Research |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
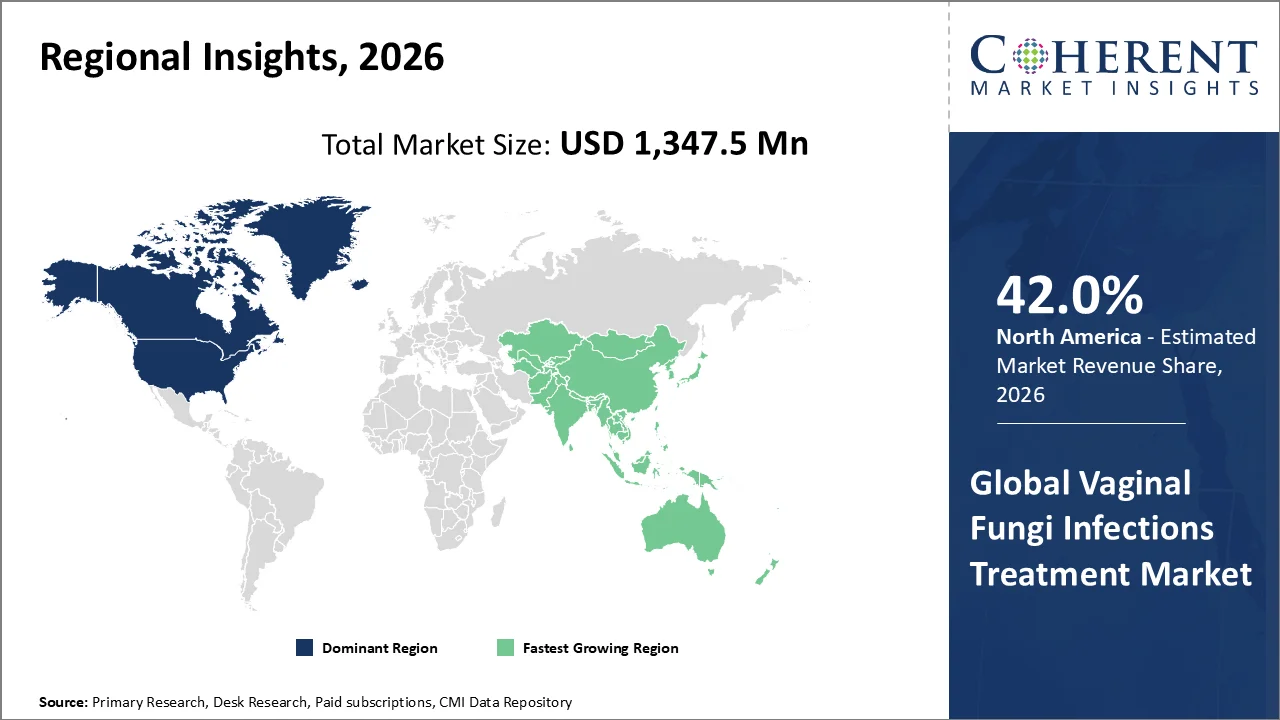
North America account 42.0% market share in 2026, supported by high prevalence of vulvovaginal candidiasis (VVC), For instance, in 2025 according to the data published by the CDC an estimated 75 % of women will have at least one episode of VVC in their lifetime and 40 %–45 % experiencing two or more episodes, underscores significant treatment demand. Moreover, according to the same source, the United States also carries a substantial clinical and economic burden from fungal diseases overall, with CDC estimates indicating approximately 13 million outpatient visits annually related to fungal infections and roughly US$19 billion in associated costs, reflecting strong clinical engagement with antifungal therapies. Advanced healthcare infrastructure, widespread awareness of reproductive health, and proactive regulatory environments (e.g., US FDA approvals of new antifungal agents like non‑azole Ibrexafungerp for vaginal yeast infections) facilitate both early diagnosis and adoption of effective treatments. Furthermore, high healthcare expenditure and robust R&D in the region support rapid integration of novel formulations and evidence‑based clinical guidelines, further reinforcing North America’s leadership in this therapeutic segment.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033. The Asia‑Pacific region is the fastest‑growing market for vaginal fungal infection treatments largely due to high disease burden and expanding healthcare access. Epidemiological studies in Vietnam (published June 2025) found that 22.6 % of women presenting at a dermatology‑venereology hospital had vaginitis, with Candida species causing 66.1 % of cases, highlighting significant regional prevalence of fungal infections among reproductive‑age women.
In India, a cross‑sectional study conducted in 2024–2025 reported 19% prevalence of vaginal candidiasis among women with symptomatic leucorrhoea, with higher rates in younger and pregnant women, compounded by rising non‑albicans species and drug resistance concerns.
Additionally, global fungal infection reports by the World Health Organization in 2025 describe an overall increase in Candida infections with limited diagnostic tools and treatment access in low‑ and middle‑income countries, further driving treatment demand in Asia‑Pacific.
This substantial disease prevalence, combined with expanding healthcare infrastructure, growing awareness, and increased availability of affordable antifungal therapies in nations such as China and India, is accelerating regional market demand faster than in other global regions.
The United States remains the dominant country in the North America vaginal fungi infection treatment market due to its ongoing regulatory activity and expanding treatment landscape in 2026, supported by strong clinical demand and innovation. Epidemiological research published in 2026 by G. Quindós et al. (MDPI) highlights advancements in novel formulations such as sertaconazole‑loaded mucoadhesive liposomes aimed at enhanced drug delivery and efficacy for vulvovaginal candidiasis, reflecting active pharmaceutical progress. Increasing research and development of new drug for vaginal fungal infection treatment in the U.S. has driven the market growth in the country. For instance, U.S. FDA‑recognized developments include ongoing clinical studies such as the VVC2025 trial initiated in March 2026 to evaluate Gedea Biotech’s product pHyph for symptom relief in vulvovaginal candidiasis, indicating future innovation pathways. ([gedeabiotech.com][2]) Established FDA approvals such as ibrexafungerp (Brexafemme) and oteseconazole (Vivjoa) approved in 2021 and 2022 respectively continue to drive market leadership by offering oral non‑azole options and recurrent yeast infection prevention, further strengthening U.S. treatment adoption and clinician preference compared with other North American markets.
Source: MDPI
India’s position as the dominant country in the Asia‑Pacific vaginal fungal infection treatment market is driven by high disease burden and rising treatment accessibility. According to WHO data published in April 2025, vulvovaginal candidiasis (vaginal yeast infection) affects millions of women globally, with higher regional burdens in populous countries like India. Indian studies report that vaginal candidiasis accounts for 15%–20% of symptomatic vaginal discharge cases, reflecting substantial prevalence that fuels treatment demand. Moreover, affordable over‑the‑counter antifungal options like clotrimazole‑based creams and tablets such as Candid‑V3 and Canesten (2026) are widely available in Indian pharmacies, enhancing self‑care and market uptake. These factors, combined with increasing women’s health awareness and strong retail distribution, position India as a leading driver of regional market growth.
Some of the major key players in Global Vaginal Fungi Infections Treatment Market are Pfizer Inc., Bausch Health Companies Inc, ANI Pharmaceuticals, Inc., Hikma Pharmaceuticals Plc, Lupin Limited, Mycovia Pharmaceuticals, Inc., Glenmark Pharmaceuticals Limited, Unique Pharmaceuticals, PEPTONIC medical AB, Aurobindo Pharma Limited, Dr. Reddy's Laboratories, and SCYNEXIS, Inc.
Source: Gedea Biotech
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,347.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.4% | 2033 Value Projection: | USD 1,935.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Bausch Health Companies Inc, ANI Pharmaceuticals, Inc., Hikma Pharmaceuticals Plc, Lupin Limited, Mycovia Pharmaceuticals, Inc., Glenmark Pharmaceuticals Limited, Unique Pharmaceuticals, PEPTONIC medical AB, Aurobindo Pharma Limited, Dr. Reddy's Laboratories, and SCYNEXIS, Inc. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients