The ADME Toxicology Testing Market is anticipated to grow at a CAGR of 10.4% with USD 7.8 Bn in 2026 and is expected to reach USD 15.23 Bn in 2033. The ADME Toxicology Testing market is experiencing significant growth, driven by increasing investments in drug R&D, technological advancements, and stricter regulatory requirements. North America leads the market, with Asia-Pacific as the fastest-growing region. The ADME toxicology testing landscape supports regulatory safety evaluation by integrating chemical property information and standardized testing protocols used worldwide. The U.S. EPA’s CompTox Chemicals Dashboard provides publicly accessible chemical toxicity and bioassay data for over 875,000 chemicals, updated as of October 2025, aiding hazard identification and prioritization of further testing. EPA’s high‑throughput screening data inform non‑animal predictive toxicology approaches that are increasingly used in early safety assessment. Additionally, the OECD Guidelines for the Testing of Chemicals offer internationally accepted methods for health effects and chemical safety testing, forming a core reference framework in ADME‑tox evaluations across regulatory systems.
Source: Environmental Protection Agency; OECD
Increasing drug discovery and preclinical testing activity has driven the global ADME toxicology testing market growth over the forecast period. Rising drug discovery and preclinical testing activity is a key driver of growth in the ADME Toxicology Testing market. In 2026, increasing demand for early-stage ADME testing is driven by the need to assess absorption, metabolism, distribution, and toxicity before costly clinical trials. According to the U.S. FDA's draft guidance (March 2026), new approach methodologies (NAMs) in drug development are gaining traction, enabling safer, faster, and cost-effective testing. These regulatory guidelines encourage in vitro and computational models over traditional animal testing. Additionally, in October 2025, the CompTox Chemicals Dashboard (U.S. EPA), provides extensive data assisting in ADME-Tox profiling, enhancing drug safety evaluation. As drug development accelerates and becomes more complex, pharmaceutical and biotech firms are investing in ADME testing to avoid late-stage drug failures and regulatory delays. Regulatory frameworks like the OECD Guidelines (2026) further strengthen the market’s reliance on these innovative testing methods.
Increasing new drug discovery has driven the market growth in the near future. For instance, in April 2026, Intellia Therapeutics, Inc., biotechnology company has reported positive topline results from its global Phase 3 HAELO clinical trial of lonvoguran ziclumeran (lonvo‑z, formerly NTLA‑2002) for hereditary angioedema (HAE), marking a major milestone in 2026 drug discovery and clinical research. The randomized, placebo‑controlled study demonstrated strong efficacy and safety, supporting the company’s decision to initiate a rolling Biologics License Application (BLA) to the U.S. Food and Drug Administration. Intellia anticipates completing the 2026 BLA submission in the second half of the year, targeting a potential first‑half 2027 U.S. launch if approved.
Increasing use of computational toxicology databases has driven the ADME toxicology testing market expansion. These databases, such as the CompTox Chemicals Dashboard (U.S. EPA), provide extensive, high-quality chemical data on toxicity, pharmacokinetics (ADME), and environmental exposure, enabling researchers to predict a compound's safety profile early in the drug development process. The increasing reliance on in silico models allows pharmaceutical companies to identify toxic effects before animal or clinical testing, reducing costs and improving efficiency. Furthermore, regulatory bodies like the U.S. FDA and OECD are encouraging the use of these computational tools to replace traditional animal testing, driving market growth. As these databases evolve, their integration into preclinical testing workflows continues to accelerate the adoption of ADME toxicology assessments, making them essential for modern drug discovery.
For instance, in March 2026, the U.S. Environmental Protection Agency (EPA) expanded its CompTox Chemicals Dashboard to include new predictive ADME‑related endpoints and updated data on over 1.2 million chemical‑biological interactions, enhancing in silico toxicology evaluations used by researchers and industry. This expansion improves early prediction of metabolism and toxicity without animal testing, supporting pharmaceutical and chemical developers in prioritizing safer compounds. The enhanced database was specifically cited in regulatory science discussions and preclinical modeling workshops throughout 2026, illustrating how richer computational data accelerates ADME toxicology testing demand.
Innovation in ADME toxicology testing is shifting preclinical drug evaluation from slow, animal-heavy screening toward faster, predictive, and human-relevant models. Advanced in vitro assays, organ-on-chip platforms, computational toxicology, AI-enabled ADME prediction, and IVIVE models help researchers estimate absorption, distribution, metabolism, excretion, and toxicity risks earlier in discovery. This reduces late-stage failures, supports safer candidate selection, and improves regulatory confidence by generating standardized evidence before clinical entry. A 2026 instance is the U.S. EPA CompTox Chemicals Dashboard Version 2.8, released on March 17, 2026, which updated the ADME > IVIVE data table using HTTK R-package v2.3 and added measured rat volume of distribution and pharmacokinetic half-life data. Similarly, FDA’s CDER page, published on March 8, 2026, confirms openness to streamlined nonclinical programs and validated NAMs, including approaches that reduce animal use in toxicology studies while maintaining safety evaluation quality for modern drug development workflows and regulatory submissions globally and portfolios.
Furthermore, in July, 2025, Ginkgo Bioworks Holdings, Inc., biotech company, revealed the introduction a new ADME profiling service designed to accelerate drug discovery by delivering high‑throughput, machine‑learning‑ready data at competitive prices. The offering which covers Absorption, Distribution, Metabolism, and Excretion assays uses the company’s proprietary RAC laboratory automation platform to provide rapid, scalable, and cost‑effective profiling services entirely from a U.S.‑based operation.
|
Current Event |
Description and its Impact |
|
Launch of New Supportive Regulatory Laws |
|
|
FDA and NIH Announce Large Funding for NAM Development |
|
|
FDA’s NAM Roadmap Support for In Silico and Predictive Models |
|
|
European Medicines Agency (EMA) Regulatory Acceptance of NAMs |
|
|
Adoption of International Test Guidelines for In Vitro Methods (OECD/US/EU) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Cell Culture Tech is projected to account for the largest share of cognitive systems spending in 2026, representing approximately 44.8% of the total volume. Cell culture systems especially advanced 3D cultures and organoids closely mimic human tissue structure, cellular interactions, and biochemical functions. This improves the predictive accuracy of drug absorption, metabolism, and toxicity compared to traditional animal models. Cell culture systems (especially 2D and automated platforms) allow rapid testing of large numbers of compounds simultaneously, which is essential in early-stage drug discovery. Innovations such as 3D cell cultures, organoids, and microphysiological systems provide more realistic simulation of human organs, improving the reliability of ADME and toxicity predictions. For instance, in March 2026, PHCbi, the biomedical division of PHC Holdings Corporation, unveiled its LiCellGrow cell expansion system, a next‑generation platform designed for real‑time monitoring and optimization of 3D cell culture production. The system incorporates in‑line metabolic monitoring technology that continuously tracks glucose and lactate key indicators of cellular health enabling automatic adjustment of culture conditions to ensure optimal growth and quality of 3D cellular constructs used in drug discovery, regenerative medicine, and cell and gene therapy manufacturing.
To learn more about this report, Request Free Sample
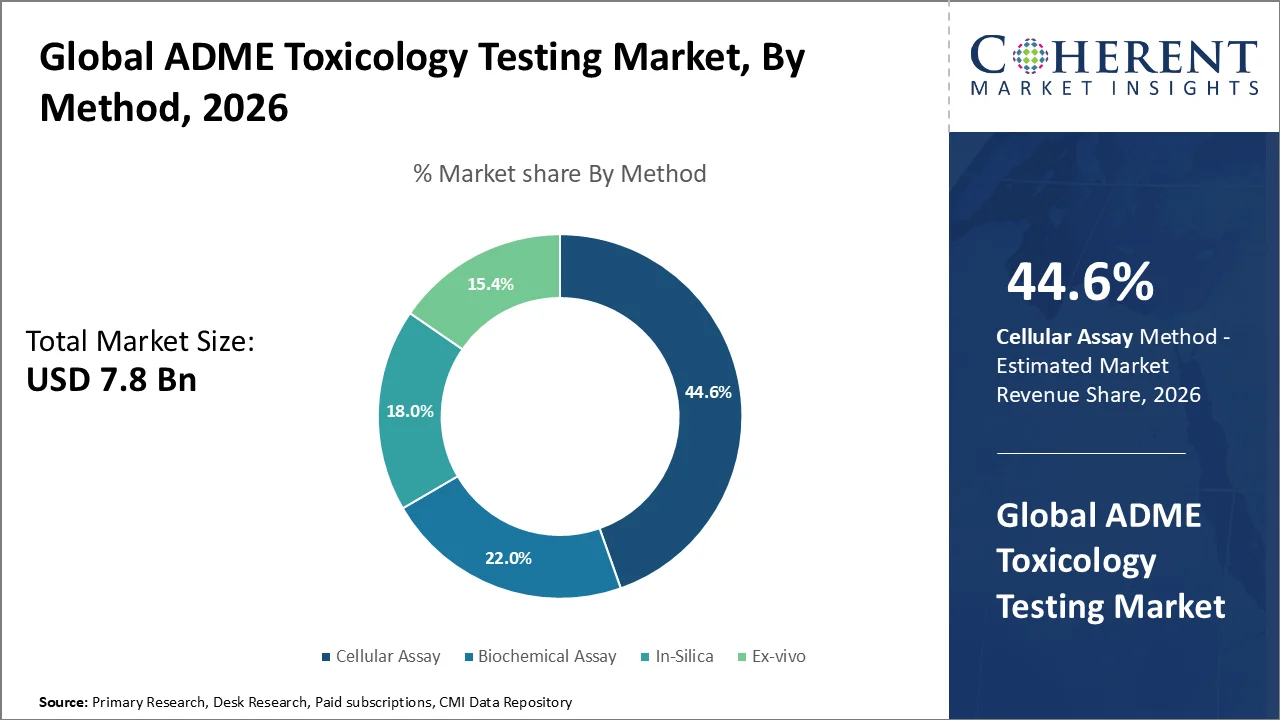
Based on method, cellular assay dominates the market, accounting for a significant 44.6% share in 2026, because it delivers highly predictive, human‑relevant biological data that improves early identification of absorption, metabolism, and toxicity risks. Cell assays including hepatocyte metabolism, cytotoxicity, transporter function, and receptor binding tests allow rapid screening of large compound libraries with reproducible, quantifiable endpoints, reducing reliance on animal models and accelerating lead optimization. In 2026, regulators like the U.S. FDA continued endorsing non‑animal methodologies, as evidenced by draft guidance promoting New Approach Methodologies (NAMs) such as advanced in vitro cell assays to support safety assessments. Additionally, platforms like PHCbi and Cyfuse’s LiCellGrow launched next‑generation real‑time 3D cell culture systems in 2026, improving assay throughput and quality. These trends reinforce cell assays’ dominance by enhancing data reliability, reducing cost, and aligning with regulatory expectations.
Systemic Toxicity account for the largest share of 55.0% in 2026 because it provides comprehensive whole‑body safety evaluations that are crucial for predicting adverse drug effects before clinical trials, aligning with major public health goals to reduce harmful outcomes. The CDC’s 2024 medication safety initiative highlights the importance of preventing adverse drug events and improving surveillance of medication‑related harm to protect patient health. The WHO’s Pharmacovigilance Programme through 2025 underscores global efforts to monitor adverse drug reactions throughout product lifecycles, aiding early risk detection and management worldwide. In the United States, the FDA’s MedWatch program (current as of April 30 2026) collects and disseminates safety alerts and adverse event reports for regulated drugs to support evidence‑based regulatory decisions and safeguard public health. Because systemic toxicity testing directly informs these real‑world safety frameworks and regulatory reporting mechanisms, pharmaceutical developers and regulators rely heavily on it to ensure drug candidates have acceptable safety profiles before advancing to human use.
To learn more about this report, Request Free Sample
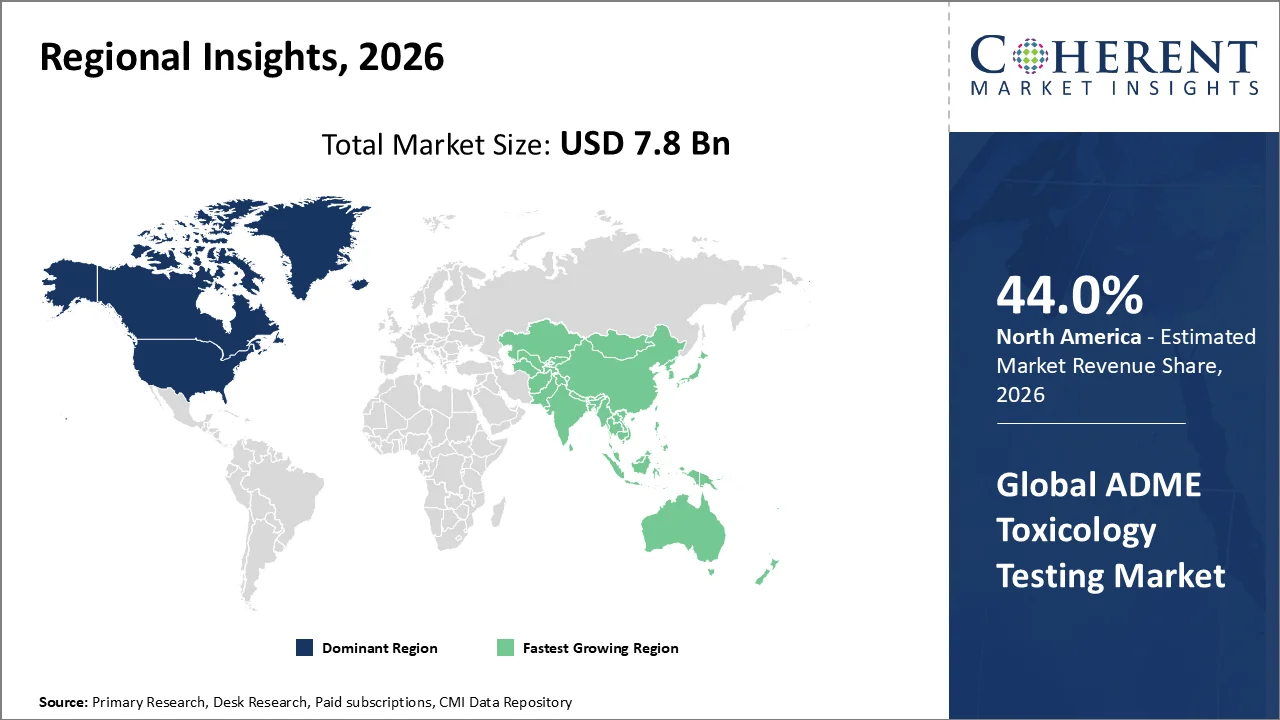
North America account 44.0% market share in 2026, supported by because the region has a strong U.S.-led pharmaceutical and biotechnology R&D base, strict FDA nonclinical safety requirements, and early adoption of advanced in vitro, in silico, organ-on-chip, and AI-enabled toxicology platforms. The FDA stated on March 18, 2026 that drug sponsors must submit nonclinical pharmacology and toxicology data before investigational drugs move into clinical trials, supporting continuous demand for ADME-Tox testing. The region is also supported by active drug development; FDA’s page, current as of May 1, 2026, listed 13 novel drug approvals in 2026, showing strong regulatory and clinical pipeline activity. In addition, NIH announced more than US$150 million on March 18, 2026 for human-based NAMs, strengthening U.S. toxicology innovation. Thermo Fisher also opened a South San Francisco drug discovery center in March 2026, reinforcing North America’s advanced testing infrastructure.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, because China, India, Japan, South Korea, Singapore, Malaysia, and Australia are expanding early-stage drug discovery, clinical trials, biologics research, and CRO-based preclinical testing. The region also has large treatment-naïve patient pools, lower trial operating costs, improving regulatory alignment, and rising use of in vitro, PK/PD, and computational toxicology methods.
The U.S. ADME toxicology testing market dominates North America due to its unparalleled pharmaceutical R&D ecosystem, stringent regulatory frameworks, and early adoption of advanced testing technologies. The U.S. hosts the world’s largest biopharma industry, driving extensive preclinical testing demands. The U.S. Food and Drug Administration (FDA) enforce rigorous ADME and toxicology data requirements before granting Investigational New Drug (IND) and New Drug Application (NDA) approvals, anchoring high market activity. For example, the FDA’s March 18, 2026 draft guidance promoting New Approach Methodologies (NAMs) like in vitro and in silico systems showcases an active regulatory push toward predictive and human‑relevant safety testing.
Additionally, significant federal funding continues to drive innovation: in 2026 the NIH announced over USD 150 million for human‑based safety science initiatives to reduce animal models, enhancing U.S. capability for advanced ADME‑Tox platforms.
Presence of major CROs and technology providers such as Thermo Fisher Scientific, Charles River Laboratories, and Covance/Labcorp further consolidates U.S. leadership through deep infrastructure, high‑throughput platforms, and integrated safety services that attract global clients. Intensive clinical pipelines, regulatory harmonization, and robust investment in predictive toxicology underpin the U.S. as the dominant country within North America’s ADME toxicology testing market.
China has become the biggest player in the Asia Pacific ADME toxicology testing market in 2026. China has emerged as a dominant country in the Asia‑Pacific ADME toxicology testing market due to its rapidly expanding drug development ecosystem, regulatory reforms, and growing Contract Research Organization (CRO) infrastructure. China’s National Medical Products Administration (NMPA) implemented a major regulatory overhaul in January 2026, codifying accelerated pathways like Breakthrough Therapy and Priority Review, which reduced review timelines by ~25 % compared with earlier years, encouraging local innovation and faster preclinical testing demand. A 2026 report noted Chinese pharmaceutical companies accounted for 19 % of all global drug R&D firms, up from 17% in 2025, underscoring China’s expanding role in early discovery and safety profiling. China also witnessed record activity in biotech licensing deals in early 2026, with total deal value from Greater China rising nearly tenfold by 2025 to USD 137.7 billion, attracting multinational investment and integration into global pipelines. These trends drive heightened ADME/Tox testing requirements in China, fueled by strong local development, regulatory acceleration, and global collaboration.
Some of the major key players in ADME Toxicology Testing Market are Thermo Fisher Scientific, Inc.; Danaher; Charles River Laboratories; Promega Corporation; Agilent Technologies, Inc.; Curia Global, Inc.; Dassault Systèmes; Catalent, Inc.; Labcorp; Eurofins Scientific; Miltenyi Biotec; IQVIA Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.4% | 2033 Value Projection: | USD 15.23 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, Inc., Danaher, Charles River Laboratories, Promega Corporation, Agilent Technologies, Inc., Curia Global, Inc., Dassault Systemes, Catalent, Inc., Labcorp, Eurofins Scientific, Miltenyi Biotec, IQVIA Inc. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients