Allergic Rhinitis Treatment Market is estimated to be valued at USD 17.79 Bn in 2026 and is expected to reach USD 25.37 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.2% from 2026 to 2033.
To learn more about this report, Download Free Sample
The Allergic Rhinitis Treatment Market Size is significant and still growing due to environmental causes, growing allergy prevalence, and new treatment options. The growing pollution levels across major cities due to rapid urbanization and industrialization have led to increased instances of allergies and other diseases. Furthermore, the rising patient awareness regarding the availability of various treatment options for allergic rhinitis is also contributing to the market growth.
For instance, in April 2025, in a significant move to address the rising burden of allergies, Inamdar Multispeciality Hospital launched a specialized Allergy Department. The initiative marks a major leap forward in early diagnosis, evidence-based treatment, and patient education-critical components often missing in routine care.
|
Event |
Description and Impact |
|
Rising Air Pollution and Climate Change |
|
|
Surge in Online Pharmacies and Digital Health |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The allergic rhinitis treatment pipeline in Europe features innovative therapies targeting improved efficacy and patient compliance. Key developments include monoclonal antibodies for e.g., REGN1908-1909 and novel immunotherapies like Revelation Biosciences' intranasal REVTx-99b, which completed Phase Ib trials for symptom management by modulating eosinophil recruitment.
Additionally, pentosan polysulfate sodium (Rhinosul) is advancing through Phase IIa trials in Sweden as a repurposed anti-inflammatory for allergic rhinitis. These therapies aim to shift treatment from symptomatic relief to long-term disease modification, with several candidates expected to launch by 2025–2026.
Europe's patent landscape for allergic rhinitis drugs is characterized by complex portfolios protecting formulations, delivery mechanisms, and indications. For instance, fluticasone furoate a widely used corticosteroid faces patent expirations in the early 2030s, opening avenues for generics.
However, secondary patents covering nasal spray formulations or combination therapies may extend market exclusivity. Companies like GSK and AstraZeneca leverage patent thickets to delay competition, though this strategy invites scrutiny under EU antitrust regulations. Post-expiry, biosimilars and generic alternatives are poised to reduce treatment costs significantly.
The treatment type segment includes immunotherapy, anti-histamines, decongestants, leukotriene receptor antagonists, corticosteroids, and others. The immunotherapy sub-segment is estimated to hold 32.2% of the market share in 2026 owing to its targeted approach in addressing the root cause of allergic rhinitis.
Immunotherapy, also known as desensitization or hypo-sensitization, counteracts the allergic reaction that an individual suffers due to allergens such as pollen, dust mites, and animal dander. Unlike symptomatic treatments like antihistamines that only offer relief from the symptoms, immunotherapy aims to reduce sensitivity to allergens over time and provide long lasting relief. It works by exposing the patient to gradually increasing doses of the allergen(s) that cause their symptoms, in order to induce tolerance.
This 'tolerance induction' affords the individual protection against future reactions upon re-exposure to these allergens. As a consequence, patients may find long term reduction or elimination of symptoms on immunotherapy. Its targeted mechanism catering to the cause rather than the symptoms alone has made it the treatment of choice for individuals seeking a long-term solution to their allergic rhinitis.
To learn more about this report, Download Free Sample
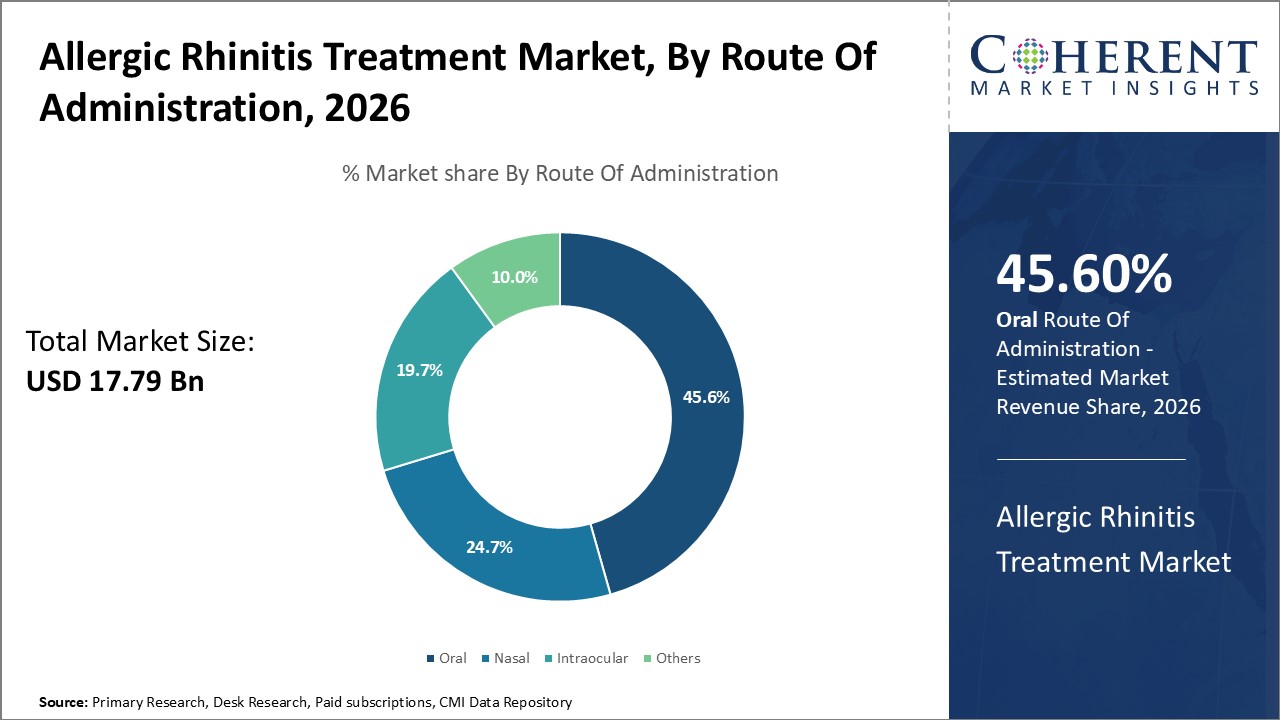
The route of administration segment includes oral, nasal, intraocular, and others. The oral sub-segment is estimated to hold 45.6% of the market share in 2026 due to the various benefits of oral administration over other modes. Oral medication is the most convenient route for self-administration as tablets or capsules can be taken without needing assistance. This makes adherence to treatment simpler.
Additionally, oral drugs typically have higher bioavailability compared to other modes meaning higher amounts of the drug reach the target sites in the body resulting in better efficacy. The avoidance of repeated invasions of the nasal or other mucosa seen with other administration modes also adds to patient comfort with oral drugs.
Furthermore, oral drugs usually have lower production and distribution costs compared to other complex drug delivery systems. Together, the convenience, effectiveness, and lower costs associated with the oral route have cemented its status as the preferred administration method for allergic rhinitis treatment.
The distribution channel segment includes hospital pharmacies, retail pharmacies, and e-commerce. The hospital pharmacies sub-segment is estimated to hold 59.3% of the market share in 2026 owing to the specialized services they provide. As healthcare experts, hospital pharmacists are well-versed in allergy management and able to offer specialized guidance to patients. They ensure each patient receives tailor-made treatment best suited to their condition and lifestyle.
Patients regularly visiting hospitals also benefit from continuity of care as all their treatment needs can be addressed under one roof. Hospital pharmacies also stock a wider range of medications including specialty drugs for complex cases. This enables more comprehensive management of allergic rhinitis.
The oversight of hospital staff further helps address any drug-related issues timeously. The specialized know-how and integrated services preferentially drive customers to hospital pharmacies over other channels for their allergic rhinitis treatment needs.
To learn more about this report, Download Free Sample
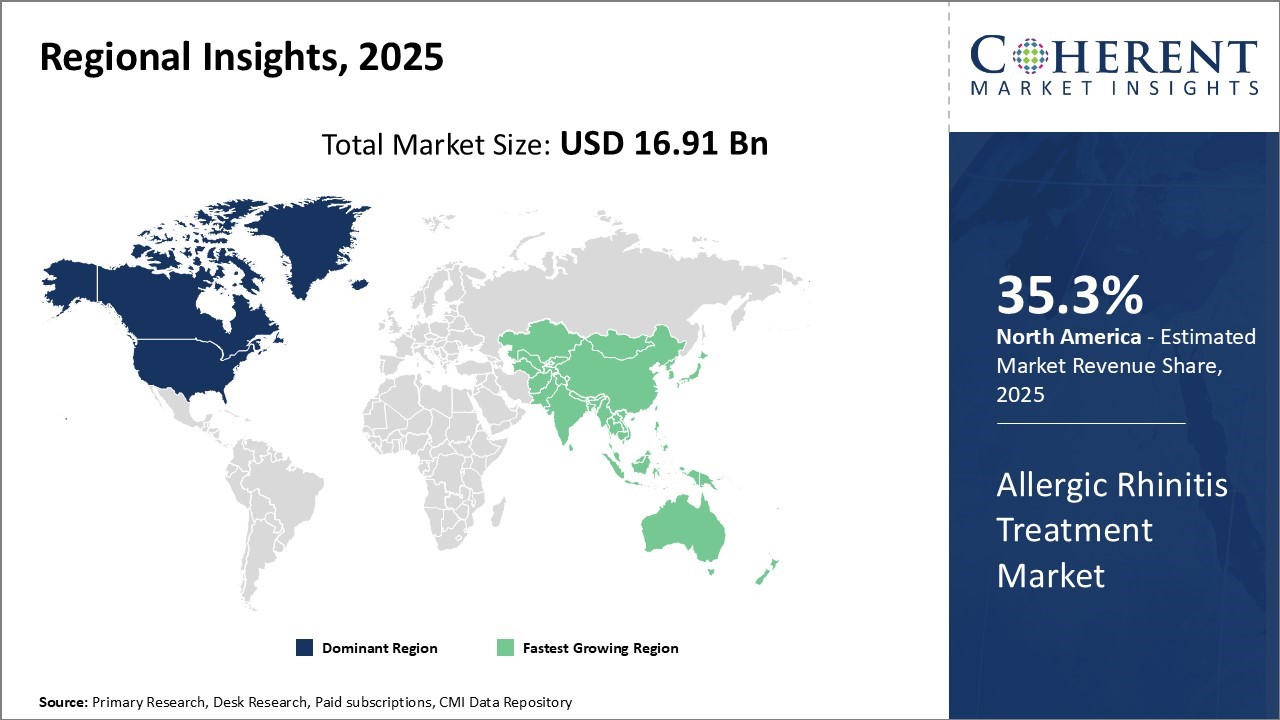
North America remains the dominant region in the global allergic rhinitis treatment market and is estimated to hold 35.3% of the market share in 2026 owing to its developed healthcare infrastructure and strong presence of leading pharmaceutical manufacturers in the U.S. With awareness among consumers regarding allergy diagnosis and management being fairly high, there is a considerable demand for prescription and over-the-counter medication options across major countries in the region.
Furthermore, the presence of several well-established contract research organizations (CROs) and research universities encourages clinical trials for new drug formulations.
Asia Pacific is poised to be the fastest growing region owing to rising environmental pollution levels and changing lifestyle patterns in densely populated nations like India and China. This has significantly increased the prevalence of allergic reactions among general population. At the same time, improving access to private and public healthcare along with increasing spending on specialty medicines is fueling market revenue growth.
While pricing of branded drugs remains relatively high in the region compared to generics, industry players are actively focusing on affordable product offerings to tap rising demand from the massive patient pool. Partnerships with local pharmaceutical companies have also enabled global vendors to strengthen supply chain and distribution networks across emerging Asia Pacific countries.
Europe has one of the highest rates of allergies in the world; up to 45% of people between the ages of 20 and 45 suffer from allergic rhinitis, and this number is predicted to continue to increase in the years to come. With a strong healthcare system and quick acceptance of cutting-edge treatments, Germany dominates the European market. The UK also shows high demand, helped by the NHS and public health campaigns.
Factors such as increasing air pollution, widespread access to healthcare, and the adoption of combination therapies including antihistamine and corticosteroid sprays—have contributed to this leadership position. The U.S. market is also characterized by rapid uptake of innovative treatments, including biologics and immunotherapies, which are expected to further drive market growth in the coming years.
India’s allergic rhinitis treatment market is expanding rapidly, propelled by rising urbanization, increasing pollution levels, and growing health awareness among the population. The country is witnessing a substantial increase in the incidence of allergic rhinitis, especially in urban centers where air quality is deteriorating.
China is emerging as a significant player in the allergic rhinitis treatment market, with growth fueled by rapid industrialization, urbanization, and escalating environmental pollution. The country has seen a marked increase in allergic rhinitis cases, particularly in densely populated urban regions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 17.79 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 25.37 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
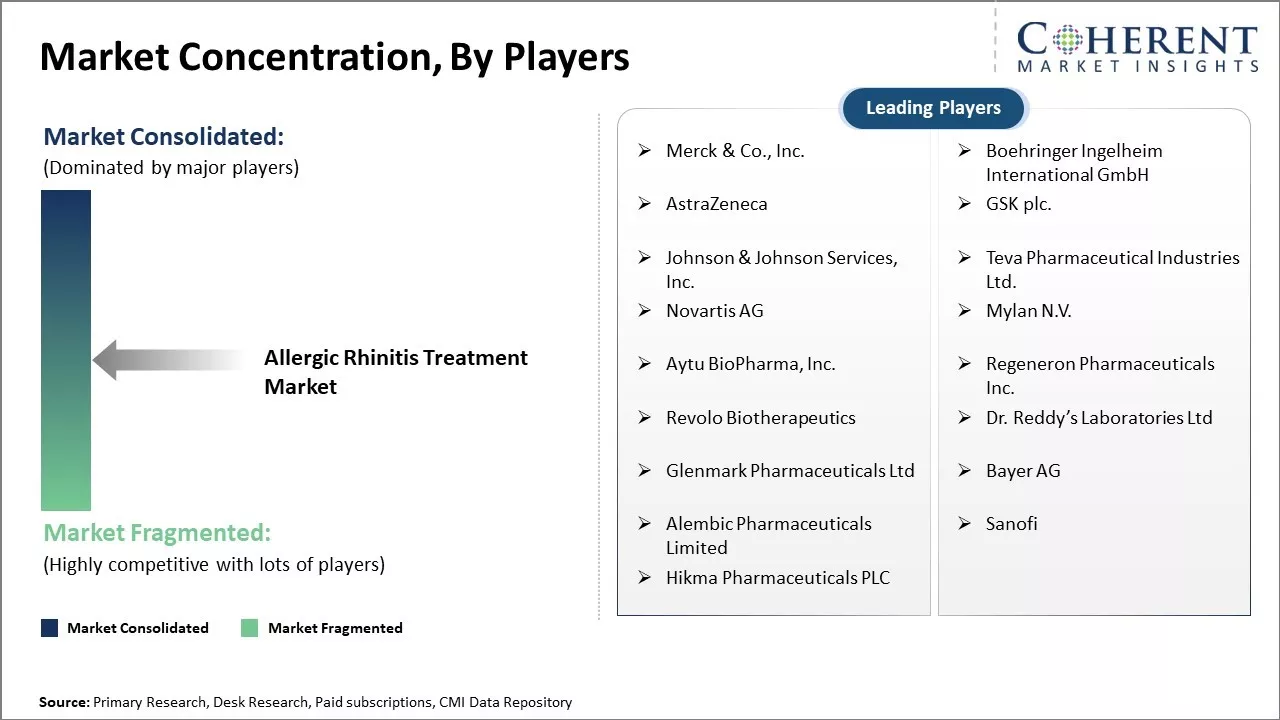
Merck & Co., Inc., Boehringer Ingelheim International GmbH, AstraZeneca, GSK plc., Johnson & Johnson Services, Inc., Teva Pharmaceutical Industries Ltd., Novartis AG, Mylan N.V., Aytu BioPharma, Inc., Regeneron Pharmaceuticals Inc., Revolo Biotherapeutics, Dr. Reddy’s Laboratories Ltd, Glenmark Pharmaceuticals Ltd, Bayer AG, Alembic Pharmaceuticals Limited, Sanofi, and Hikma Pharmaceuticals PLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Download Free Sample
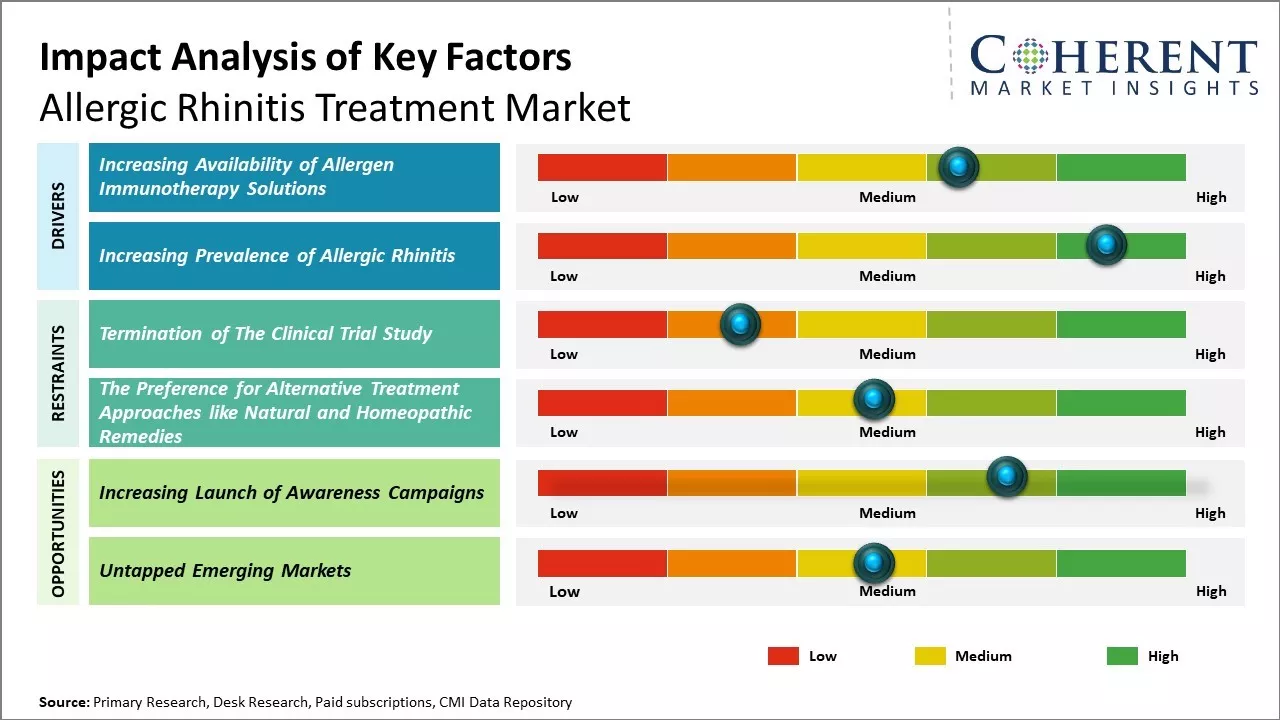
The increasing availability of allergen immunotherapy solutions is one of the factors that is expected to drive the market growth during the forecast period. For instance, in June 2021, Stallergenes Greer, one of the global leaders in allergy immunotherapy, announced that the Alusta, subcutaneous allergen immunotherapy (SCIT) solution for different allergies involving rhinitis, conjunctivitis, rhino conjunctivitis, or asthma is now available in Spain and Italy.
Growing industrialization and urbanization have led to higher concentrations of outdoor allergens like pollen, mold, dust, and animal dander in the air. At the same time, people are spending more time indoors where indoor allergens from furnishings, cleaning products, and other materials are present. This has increased individual's exposure and susceptibility to develop allergic rhinitis.
Thus, the increasing prevalence of allergic rhinitis boosts the demand for effective treatment which is expected to drive the market growth over the forecast period. For instance, according to the World Allergy Organization report in October 2020, over 400 million people suffer from allergic rhinitis around the world, which to a large extent remains underdiagnosed and undertreated. In the U.S., it affects between 10-30% of the adult general population and up to 40% of children, making it the fifth most common chronic disease.
Easy access to prescription and over-the-counter medications through physical and online pharmacies is contributing to a shift in patient knowledge and proactive treatment. Digital health platforms and tools for identifying allergens are examples of technological innovations that are improving diagnosis and individualized treatment.
The market is challenged, nevertheless, by the availability of substitute therapies and the lapse of patents on important medications, which could affect competition and pricing. Overall, it is anticipated that the market for treatments for allergic rhinitis will continue to increase steadily due to innovation, external factors, and changing patient needs.
Increasing adoption of inorganic growth strategies such launch of awareness campaigns by key market players is expected to offer lucrative growth opportunities in the market. For instance, in June 2023, Alkem, a global pharmaceutical company, announced that they had launched "Stop Sneeze to Wheeze" Campaign to Raise Awareness on Allergic Rhinitis and Asthma Coexistence during World Allergy Week 2023. The programme highlights the idea of "One Airway, One Disease" and seeks to increase awareness of the coexistence of allergic rhinitis and asthma.
Allergic Rhinitis Treatment Market: Key Developments
*Definition: Allergic rhinitis also called as hay fever, is an inflammation of the inside of the nose due to an allergic reaction to tiny particles in the air called allergens. Allergic rhinitis is also called cold-like symptoms because the symptoms resemble to cold symptoms such as sneezing, itchiness, blocked or runny nose, etc. The immune system reacts to a particular type of allergen, which causes allergic rhinitis. This reaction releases a different number of chemicals inside of mucous membrane of the nose due to which the nose becomes swollen and forms an excessive amount of mucus. Various allergens that cause allergic rhinitis include pollen, spores, house dust mites, flakes of skin, or droplets of saliva or urine from a certain animal. When allergic rhinitis is caused by pollen, it is called hay fever.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients