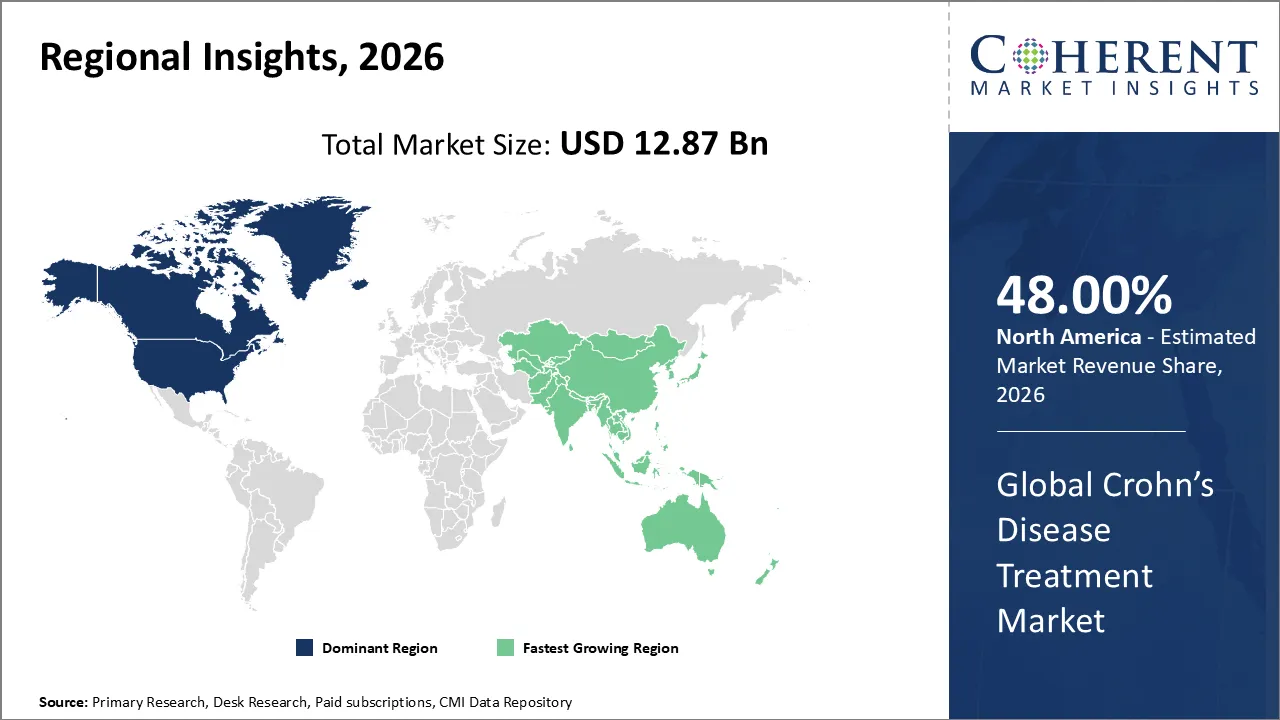
The global Crohn’s disease treatment market was valued at USD 12.87 Bn in 2026 and is forecast to reach a value of USD 17.16 Bn by 2033 at a CAGR of 4.2% between 2026 and 2033.
Crohn’s disease is a form of inflammatory bowel disease (IBD) that refers to a chronic inflammatory condition of the gastrointestinal tract causing inflammation in the digestive tract. The disease is confined to the colon of the large intestine and ileum of the small intestine. The disease is often painful and debilitating and can lead to life-threatening complications in some cases. These complications may comprise of one or more factors that includes bowel obstruction, fistulas, ulcers, anal fissures, colon cancer, and other health issues. Crohn’s disease can be caused due to factors such as stress, diet, heredity and malfunctioning of the immune system. The disease can occur at any age. However, is likely to develop at young ages among majority of the individuals
|
Current Event |
Description and its Impact |
|
Regulatory and Policy Changes in Drug Approval Processes |
|
|
Breakthrough Technological Advancements in Treatment Modalities |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Category |
Key Data (2025) |
Notes |
|
Prevalence |
~3.5 million people worldwide living with Crohn’s disease |
Rising incidence in Asia-Pacific and Middle East compared to stable rates in North America/Europe. |
|
Biologic Use |
>65% of moderate-to-severe Crohn’s patients in the US are on biologics |
Anti-TNF agents (infliximab, adalimumab) still dominate, but IL-23 inhibitors (guselkumab, risankizumab) are rapidly growing. |
|
Treatment Costs |
Average annual biologic therapy cost: $25,000–$40,000 per patient |
Biosimilars (e.g., infliximab, adalimumab) reduce costs by 20–30% in EU markets. |
|
Hospital Burden |
Crohn’s accounts for ~200,000 hospitalizations annually in the US |
30% reduction in hospitalizations observed in patients on biologics vs conventional therapy. |
|
Surgery Rates |
20–25% of Crohn’s patients require surgery within 10 years of diagnosis |
Biologics have lowered surgical rates compared to pre-biologic era (>50%). |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug type, the immunomodulators segment is expected to hold 47.3% share of the market in 2026, due to their control the immune system to lower chronic inflammation. They are the best choice considering they can keep patients in remission, limit flare-ups, and improve long-term outcomes. This class keeps getting better, which makes them better than older treatments like corticosteroids.
For instance, in January 2025, the FDA approved Eli Lilly's Omvoh (mirikizumab) for Crohn's disease, which means it can be used for more than just ulcerative colitis. This milestone makes Omvoh the first IL-23p19 inhibitor approved for both major inflammatory bowel diseases. This gives patients a new treatment option and strengthens Lilly's immunology portfolio.
In terms of distribution channel, the hospital pharmacies segment is expected to lead the market with 58.1% share in 2026, because many immunomodulators need to be administered and monitored by a doctor. Hospitals have the infrastructure to make sure that deliveries are safe, patients follow through, and medical help is available right away. This guarantees successful treatment results and solidifies their leading status over retail and e-commerce channels.
For instance, in November 2025, The Scottish Medicines Consortium has approved Johnson & Johnson's TREMFYA (guselkumab) for use by NHS Scotland in Crohn's disease and ulcerative colitis. After NICE's recommendation, guselkumab is now available as a subcutaneous induction and maintenance option. This makes it easier for people with inflammatory bowel disease in the UK to get treatment.
To learn more about this report, Request Free Sample
North America is expected to dominate the Crohn’s disease treatment market with 48% share in 2026, driven by a high number of cases, a well-developed healthcare system, a strong use of biologics, supportive reimbursement policies, and the presence of major pharmaceutical companies. These things together maintain its dominant market share and growth expanding.
For instance, in November 2025, Polpharma Biologics declared that Tyruko (natalizumab) is now available in the U.S. It is the first and only FDA-approved biosimilar for multiple sclerosis. Tyruko gives patients an affordable option to branded therapy, which is an important step forward in making MS treatment more accessible and giving neurologists more biosimilar options.
Asia Pacific is expected to be the fastest growing region, due to increasing diagnosed cases, healthcare infrastructure is growing, people are becoming more aware of inflammatory bowel diseases, and rising investment into biologics and biosimilars. The market is growing even faster because of rapid urbanization and the growth of the pharmaceutical industry in China and India. APAC is the fastest-growing region.
For instance, in September 2025, the "Dual Control" campaign from Johnson & Johnson is meant to give people with inflammatory bowel disease more power in Asia Pacific. The goal of the initiative is to raise awareness, build resilience, and encourage people with IBD to follow their dreams while also managing their illness.
The U.S. Crohn's Disease Treatment Market remains highly competitive in 2026 because there are an enormous number of patients, the healthcare system is highly advanced, biologics are extremely popular, reimbursement systems are very strong, and there are a large number of leading pharmaceutical innovators. These things make sure that the company stays on top and keeps growing in the global market.
For instance, in February 2025, At ECCO 2025, Sanofi and Teva presented new Phase 2b RELIEVE UCCD study data that showed duvakitug works effectively and is safe for people with ulcerative colitis and Crohn's disease. Results showed that there were large clinical and endoscopic improvements in all subgroups. This supports duvakitug's potential to be the best in its class and move on to Phase 3 trials later in 2025.
The demand for Crohn's disease treatment in India is going to rise in 2026 as more people are learning about inflammatory bowel diseases, increasing diagnosis rate, healthcare infrastructure continues to improve, and more patients can get advanced biologics and biosimilars. Investment in pharmaceuticals and the growth of cities are also driving adoption, making India a key growth engine in the Asia-Pacific region.
For instance, in August 2025, Takeda India has made a new mobile app to help people with inflammatory bowel disease. The app has tools for managing diseases, maintaining to treatment plans, and teaching patients about their conditions. Its goal is to improve quality of life and give people the tools they need to better manage Crohn's disease and ulcerative colitis.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12.87 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.2% | 2033 Value Projection: | USD 17.16 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie Inc., Janssen Biotech, Inc., Bristol-Myers Squibb Company, Boehringer Ingelheim GmbH, Pfizer Inc., Eli Lilly and Company, Novartis AG, Sanofi, Takeda Pharmaceutical Company Limited, Mylan N.V., Amgen Inc., and AstraZeneca |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Millions of people in North America, Europe, and emerging markets are affected by Crohn's Disease, and the number of people with it is still growing. As more people get Crohn's disease, the demand for treatments rises. Healthcare systems and drug companies work hard to find effective treatments. More diagnoses, better awareness campaigns, and easier access to medical care are all making the need for advanced treatments even greater. As prevalence rises, the market keeps going strong, creating steady chances for new ideas and growth.
Biologics and targeted therapies, like anti-TNF agents, JAK inhibitors, and monoclonal antibodies, are changing the way we treat diseases by being more effective and having fewer side effects than regular drugs. Their use is greatly increasing the Crohn's Disease treatment market growth, as both patients and doctors are choosing these more advanced options more and more. Ongoing clinical trials and approvals from regulatory bodies are adding to the therapeutic pipeline, making sure that new solutions keep coming in. This progress in technology makes the market more competitive and helps it grow over the long term.
For instance, in July 2025, BIOCAD announced it would start Phase 2 clinical trials for its first Crohn's disease treatment. The study's goal is to test the new therapy's safety and effectiveness. This will lead to more innovative options for people with inflammatory bowel disease and strengthen BIOCAD's pipeline in immunology and gastroenterology.
Pharmaceutical companies are using mergers, acquisitions, and strategic alliances increasingly to make their Crohn's Disease portfolios stronger. These partnerships speed up the process of making new drugs, make it easier to get them to more people, and give patients better access to cutting-edge treatments. Companies can cut R&D costs and speed up the time it takes to get new treatments to market by sharing resources and knowledge. These kinds of partnerships also help businesses get into new areas more easily, which opens up new growth opportunities. Because of this, these strategies are very important in making the Crohn's Disease treatment market forecast.
The market for Crohn's disease treatments is growing as growing diagnosed patient population, better treatments are being used quickly. Epidemiological studies show that millions of people in major markets now have Crohn's disease. The number of people with the disease is steadily rising due to it is being diagnosed more accurately and people are becoming more aware of it. Biologic therapies are the most common type of treatment. Anti-TNF inhibitors, integrin receptor antagonists, and interleukin inhibitors are some of the drugs that are most frequently prescribed in clinical practice and holds the largest share of prescriptions. These biologics have demonstrated robust clinical efficacy in the management of moderate-to-severe disease and in decreasing relapse rates, thereby supporting their continued application in specialized care.
Distribution channels show that hospital pharmacies and outpatient infusion centers are still the most important places to get advanced therapy, while retail and online pharmacies help people get oral maintenance medication. There are still differences between regions. North America has a large share of treatment uptake because it has a well-developed healthcare system and a high rate of diagnosis. In Asia-Pacific and Europe, demand is still growing because healthcare spending is rising and treatments are becoming more accessible.
Overall, the market is moving toward personalized treatment plans and new targeted therapies. Ongoing clinical research and better diagnostic practices will shape the future of treatment.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients