The Antipsychotic Drugs Market is anticipated to grow at a CAGR of 7.5% with USD 19.4 Bn share in 2026 and is expected to reach USD 32.24 Bn in 2033. The Antipsychotic Drugs Market growth is driven by the rising diagnosed burden of schizophrenia, bipolar disorder, and psychosis, along with wider acceptance of long-term pharmacological management. WHO’s schizophrenia fact sheet, published in 2025, states that schizophrenia affects around 23 million people globally and only 29% of people with psychosis receive specialist mental health care, showing a large treatment gap. Innovation is also supporting growth: the FDA’s 2026 Novel Drug Approvals list reported approval of Bysanti (milsaperidone) for schizophrenia and manic or mixed episodes associated with bipolar I disorder, strengthening treatment options for adults and reinforcing demand for newer atypical antipsychotic therapies.
Source: WHO
Schizophrenia is projected to account for the largest share of antipsychotic drugs in 2026, representing approximately 41.0% of the total volume. The schizophrenia segment remains the dominant indication within the antipsychotic drugs market primarily because schizophrenia is a chronic, lifelong psychiatric disorder that requires sustained pharmacological intervention, making antipsychotics the foundation of therapy from first‑episode diagnosis through maintenance phases. For instance, on October 6, 2025, according to the World Health Organization’s schizophrenia fact sheet, schizophrenia affects approximately 23 million people worldwide, underpinning a steady and high baseline demand for antipsychotic medications for symptom control and relapse prevention. Epidemiological analyses have consistently shown that schizophrenia’s global burden remains significant, with millions of new cases emerging annually and prevalence remaining stable over decades, reinforcing its centrality to market demand. Clinical guidelines and studies indicate that antipsychotic therapy is the first‑line standard of care for virtually all diagnosed schizophrenia patients, given its effectiveness in reducing psychotic symptoms and managing long‑term outcomes.
Based on drug, risperidone dominates the market, accounting for a significant 30.0% share in 2026, driven by a combination of clinical effectiveness, broad regulatory approvals, varied formulations, and an established safety‑tolerability profile that supports wide therapeutic use.
Furthermore, Risperidone’s position as the dominant segment in the antipsychotic drugs market is underpinned by longstanding and broadly recognized clinical utility, wide regulatory approvals, and formulation diversity that enhances treatment adherence and outcomes. For instance, on November 10, 2024, according to StatPearls (NIH), risperidone is an FDA‑approved second‑generation antipsychotic for schizophrenia, bipolar I disorder (including maintenance therapy), and autism‑associated irritability with both oral and long‑acting injectable (LAI) options that support flexible clinical use and improved symptom control, backed by guideline recommendations for antipsychotic treatment. The availability of multiple LAI formulations including newer options evaluated in 2026 clinical practice research (e.g., Risperidone‑ISM showing effectiveness in hospitalized acute patients) helps address adherence challenges that are common in chronic psychotic disorders. Real‑world evidence and long‑acting formulations like UZEDY (data reported November 1, 2023) demonstrate significant prolongation of time to relapse in schizophrenia, reinforcing clinicians’ preference for risperidone LAIs in routine care. These factors credible regulatory endorsements, extensive formulation choices, and adherence‑enhancing delivery systems collectively support the segment’s dominance.
To learn more about this report, Request Free Sample
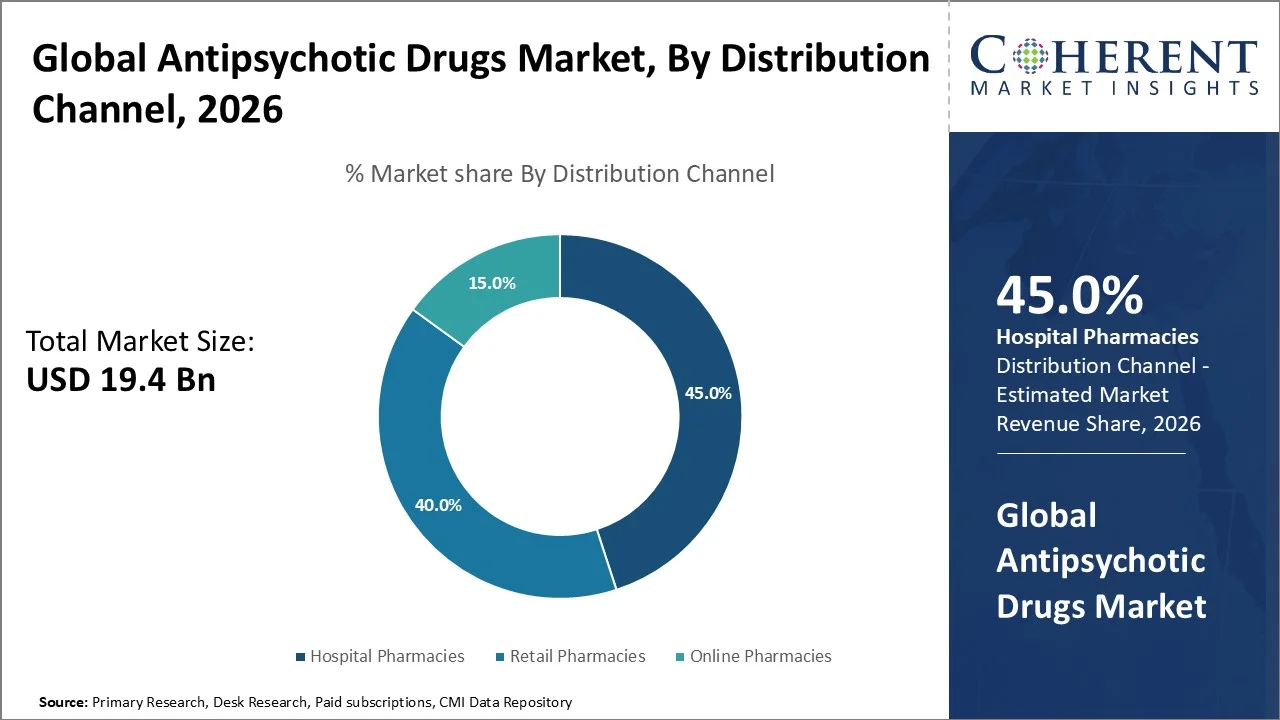
Hospital Pharmacies account for the largest share of 45.0% in 2026 due to their critical role in providing specialized, controlled medications under clinical supervision. They ensure proper dosing, monitor treatment efficacy, and manage potential side effects, particularly with long‑acting injectables and polypharmacy regimens. According to the WHO and PubMed, hospital pharmacies support mental health care through comprehensive medication management and adherence programs, offering a higher level of care than retail pharmacies, especially for inpatient and complex psychiatric treatments. In antipsychotic therapy, ensuring correct, supervised administration and monitoring is critical because these medications are central to managing severe psychotic disorders such as schizophrenia and bipolar spectrum conditions, where non‑adherence dramatically increases relapse risk and poorer outcomes; the World Health Organization’s mhGAP guidelines from 2023 emphasize the clinical significance of depot (long‑acting injectable) antipsychotics in improving treatment adherence and stabilizing patients under specialist care a process that typically requires institutional pharmacy support such as that provided in hospital settings rather than community retail pharmacies.
Scientific studies consistently show that medication regimens initiated or adjusted during hospitalization require closely coordinated dispensing and monitoring services, making hospital pharmacies the central locus for antipsychotic distribution and safety oversight particularly where polypharmacy and therapeutic drug interaction risks are high. Moreover, pharmacy practice research published in 2025 highlights the importance of pharmacists’ roles in medication management within mental health services, reinforcing those institutional settings with trained clinical teams, such as hospital pharmacies, are essential for optimizing antipsychotic outcomes and minimizing adverse events.
The increasing prevalence of mental health disorders including schizophrenia, bipolar disorder, and other serious psychiatric conditions is a fundamental driver of the antipsychotic drugs market growth because it directly expands the pool of individuals requiring pharmacological treatment and long‑term symptom management. According to the World Health Organization (WHO), approximately 970 million people globally were living with a mental disorder as of 2019, highlighting the immense and persistent global burden of these conditions (data published on the WHO website in 2023). More recent behavioral health analyses show that the prevalence of any mental illness continued to rise into 2024, especially among young adults, driving greater demand for psychiatric services and associated medications (2026 Behavioral Health Report). This sustained rise in diagnosed cases increases prescription rates and encourages development of advanced antipsychotic therapies, including long‑acting injectables and newer agents with improved safety profiles, as healthcare systems and pharmaceutical manufacturers respond to higher demand. For example, in February 2026, the US FDA approved a new atypical antipsychotic (Bysanti) for schizophrenia and bipolar I disorder, underscoring ongoing industry response to prevalent psychiatric conditions.
Source: WHO; Trilliant Health
The expanding geriatric population is a key driver of growth in the antipsychotic drugs market because older adults are more susceptible to neuropsychiatric and age‑related mental health conditions that require pharmacological management. For instance, in October 2025, according to the data published by the World Health Organization’s fact sheet on ageing and health it reports that the global population of people aged 60 years and older is projected to reach 1.4 billion by 2030, and continuing to grow rapidly, particularly in low‑ and middle‑income countries, signifying a substantial rise in the elderly demographic that health systems must serve. As individuals age, the likelihood of cognitive disorders such as dementia, Alzheimer’s disease, and associated psychosis increases, often necessitating antipsychotic drug therapy to manage behavioral and psychological symptoms, thereby enlarging the addressable patient base. Additionally, older adults often experience multiple co‑morbidities and complex clinical presentations, prompting broader antipsychotic utilization in integrated treatment plans. A January 2026 clinical analysis highlighted growing long‑term antipsychotic prescribing in elderly dementia patients, underscoring real‑world demand in this age segment. This demographic shift boosts demand for both existing and novel antipsychotic therapies as healthcare providers strive to meet the escalating needs of an aging population.
Source: WHO; News-Medical.net
Innovations in antipsychotic therapy are reshaping the market by improving efficacy, safety, and patient adherence. For instance, in 2026, the U.S. FDA accepted Teva’s New Drug Application for an extended‑release, once‑monthly olanzapine injectable a formulation designed to enhance long‑term treatment stability and adherence without intensive post‑injection monitoring, addressing a major unmet need in schizophrenia care. Similarly, in 2026, the FDA approved a supplemental application for CAPLYTA (lumateperone), demonstrating significantly reduced relapse risk and improved long‑term stability in schizophrenia patients. The same year, the FDA approved Bysanti (milsaperidone) for schizophrenia and bipolar disorder, adding a novel atypical antipsychotic option. These regulatory advancements reflect technological progress in drug formulations and relapse management, expanding treatment choices, improving outcomes, and thus driving broader adoption and market growth.
|
Current Event |
Description and its Impact |
|
FDA and EMA Propel Psychiatric Drug Innovation Through Regulatory Approvals in 2026 |
|
|
WHO Expands Mental Health Services Under Global Action Plan |
|
|
WHO Special Initiative for Mental Health Enhances Treatment Access |
|
|
Indian Government Strengthens Mental Healthcare Access |
|
|
Increasing Initiatives by Regulatory Bodies |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Recent clinical evidence confirms that long‑acting injectable antipsychotics are increasingly used due to improved adherence and reduced relapse in schizophrenia spectrum disorders. A 2026 PubMed‑indexed expert consensus highlights LAIs as beneficial even early in treatment, especially when adherence with oral therapy is inconsistent; they have been linked to reduced hospitalization rates and stable drug exposure, underscoring their growing clinical role in psychiatric care.
The World Health Organization’s 2023 mhGAP guidance on psychosis and bipolar disorders reaffirms the importance of supervised antipsychotic treatment under specialist care. The guidance validated and in use through 2025–2026 prioritizes balancing efficacy with side‑effect profiles and monitoring, encouraging individualized therapeutic approaches for complex psychiatric conditions like schizophrenia and bipolar disorder.
Emerging studies published in 2026 demonstrate that machine learning and digital phenotyping are being explored to predict individual patient responses and relapse risk for antipsychotic treatment. This suggests a trend toward personalized treatment algorithms that could tailor antipsychotic choice and dosing to individual profiles, improving long‑term outcomes and minimizing adverse effects.
To learn more about this report, Request Free Sample
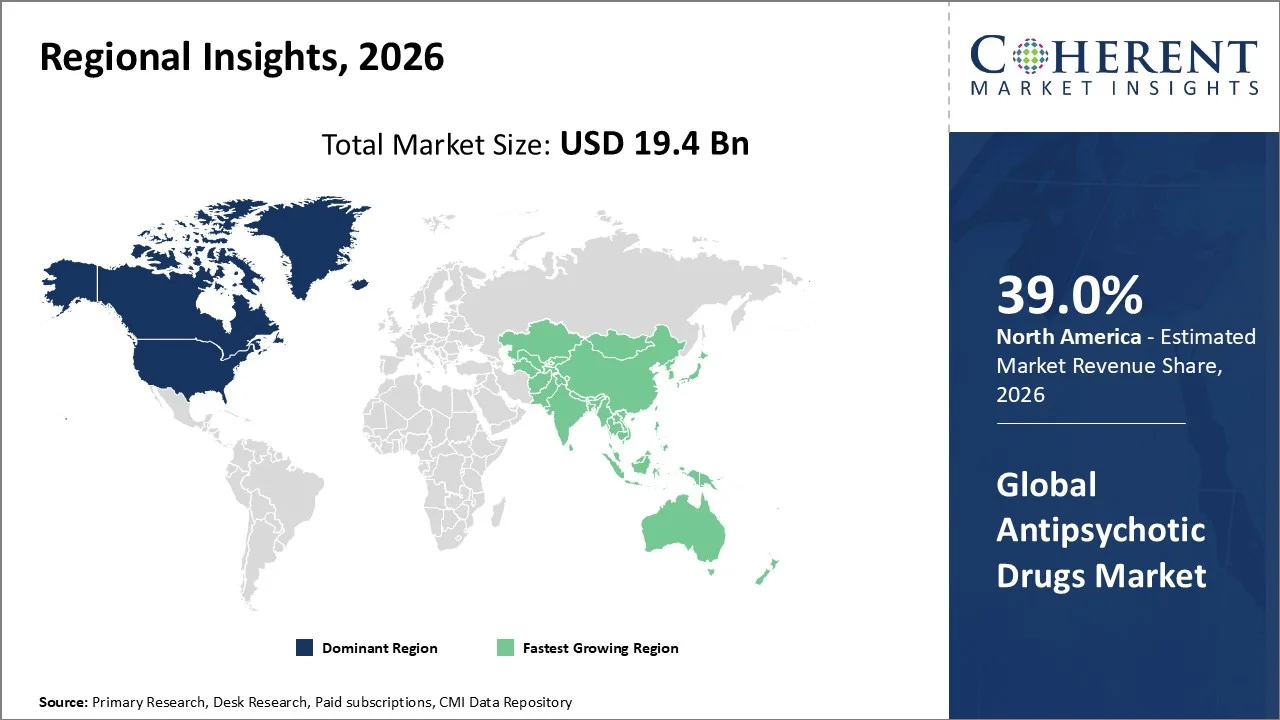
North America account 39.0% market share in 2026, supported by a constellation of structural, clinical, and regulatory factors that reinforce the region’s leadership in 2026. Established high prevalence of psychiatric conditions such as schizophrenia, bipolar disorder and other serious mental illnesses in the U.S. and Canada creates sustained clinical demand for antipsychotic therapies. For instance, in June 2025, according to the U.S. Centers for Disease Control and Prevention (CDC) (data published June 9, 2025), about 23 % of U.S. adults live with a mental health condition, and about 6 % (nearly 1 in 18) have a serious mental health condition, which includes disorders like schizophrenia and bipolar disorder. This indicates a substantial population potentially requiring specialized psychiatric care. Robust healthcare infrastructure and high healthcare expenditure facilitate widespread diagnosis and management of mental health disorders and support rapid adoption of advanced drug classes, including second‑ and third‑generation antipsychotics. The presence of leading biopharmaceutical companies, strong R&D investment, and an active regulatory environment highlighted by recent U.S. FDA approvals of novel antipsychotic and psychiatric treatments in 2026 ensure a continuous flow of innovative therapies to market. Additionally, extensive insurance coverage and public health initiatives improve patient access and uptake, further amplifying North America’s market size relative to other regions.
Furthermore, increasing approval and launch of the new antipsychotic drugs by the key players operating in the North America region has further driven the growth of the market in the region. For instance, on February 2026, the U.S. Food and Drug Administration approved the treatment of schizophrenia and manic or mixed episodes associated with bipolar I disorder. It is an atypical antipsychotic oral medication being positioned for a U.S. launch in 2026 (expected in the third quarter).
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to several interrelated epidemiological, healthcare and socio‑economic factors. For instance, according to the World Health Organization’s Mental Disorders fact sheet published on 30 September 2025, mental health conditions such as schizophrenia, bipolar disorder and depression affect large populations globally, and a substantial proportion of these individuals reside in Asia Pacific countries where treatment gaps remain high. A November 2024 OECD/WHO Health at a Glance: Asia/Pacific report revealed that mental and neurological conditions affect approximately 475 million people in the region, with significant unmet needs due to limited-service coverage and under‑resourced care systems. These public health burdens have driven governments in China, India, Japan and Southeast Asian countries to strengthen mental health policy frameworks, improve diagnostic capacity and expand access to psychiatric care, thereby expanding demand for antipsychotic therapies. Market analysis reports from 2025‑26 further note rising mental health awareness, increased healthcare expenditure, urbanization‑linked stress and improved healthcare infrastructure as key growth drivers for antipsychotic drugs in Asia Pacific, supported by both domestic and multinational pharmaceutical activity in the region.
The U.S. antipsychotic drugs market dominates North America due to increasing regulatory approvals, government initiatives and rising prevalence in the U.S. country. For instance, according to CDC mental health data reviewed in February 2026, about 12.1% of U.S. adults report regular feelings of anxiety and 4.8 % report regular feelings of depression, based on the 2024 National Health Interview Survey. CDC surveys from 2024 also show that 19% (nearly 1 in 5) of U.S. adults have ever been diagnosed with a depression disorder, and 12% report regular anxiety symptoms, highlighting substantial common mental health burden. Further, the country’s advanced healthcare infrastructure and extensive insurance coverage ensure broad access to psychiatric medications and specialist care, enabling earlier diagnosis and long‑term management. Furthermore, the U.S. is home to many major biopharmaceutical companies investing heavily in R&D and new drug launches, creating a dynamic pipeline of innovative antipsychotic therapies that reinforces market leadership. For instance, on April 27, 2026, the manufacturer announced updated U.S. FDA data supporting long‑term relapse prevention in schizophrenia, reinforcing its therapeutic positioning in the antipsychotic space, though this is a label expansion rather than a brand‑new molecule.
China has become the biggest player in the Asia Pacific antipsychotic drugs market in 2026. China’s dominance in the Asia Pacific antipsychotic drugs market stems from a unique combination of demographic scale, healthcare reforms, manufacturing strength and policy support. China leads the region largely because of its vast patient population with growing recognition and diagnosis of psychiatric conditions, which fuels demand for both generic and branded antipsychotic therapies. China’s robust domestic pharmaceutical manufacturing base ensures competitive pricing and supply scale, attracting multinationals to form collaborations and local production partnerships, accelerating therapy availability. Additionally, continuous investments in R&D and streamlined clinical trial and approval processes, supported by regulatory reforms, have strengthened China’s pharmaceutical ecosystem, enabled faster introduction of advanced antipsychotic formulations and contributed to its leadership in the Asia Pacific market. For instance, in April 2026, Luye Pharma Group Ltd., a key Chinese pharmaceutical company actively advancing investigational CNS/antipsychotic therapies. In April 2026, Luye announced that the first subjects were enrolled in Phase II clinical trials in China for its innovative compounds such as LY03020 for schizophrenia and LY03017 for Alzheimer’s disease‑related psychosis, showing ongoing clinical work in antipsychotic‑relevant CNS conditions. Luye’s broader CNS pipeline (including LY03015, LY03020, LY03017 and others) was highlighted in its April 2026 corporate investor announcement.
Source: Luye Pharma Group
Some of the major key players in antipsychotic drugs market are H. Lundbeck A/S; Otsuka Pharmaceutical Co., Ltd.; Janssen Pharmaceuticals, Inc.; Eli Lilly and Company; AbbVie, Inc.; Teva Pharmaceutical Industries Ltd.; Dr. Reddy’s Laboratories Ltd.; Sumitomo Pharma Co., Ltd.; Alkermes; Bristol-Myers Squibb Company
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 19.4 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 32.24 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
H. Lundbeck A/S; Otsuka Pharmaceutical Co., Ltd.; Janssen Pharmaceuticals, Inc.; Eli Lilly and Company; AbbVie, Inc.; Teva Pharmaceutical Industries Ltd.; Dr. Reddy’s Laboratories Ltd.; Sumitomo Pharma Co., Ltd.; Alkermes; Bristol-Myers Squibb Company |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients