Intrauterine Contraceptive Devices Market Size and Forecast – 2026 – 2033
The Global Intrauterine Contraceptive Devices Market size is estimated to be valued at USD 4.3 billion in 2026 and is expected to reach USD 7.8 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033.
Global Intrauterine Contraceptive Devices Market Overview
Intrauterine contraceptive devices (IUDs) are long-acting, reversible birth control products inserted into the uterus to prevent pregnancy. These devices work either by releasing hormones or by creating a local environment that inhibits fertilization. IUDs are designed to provide effective contraception for several years with minimal maintenance. They are widely used in clinical settings and are valued for their reliability, convenience, and low user dependency.
Key Takeaways
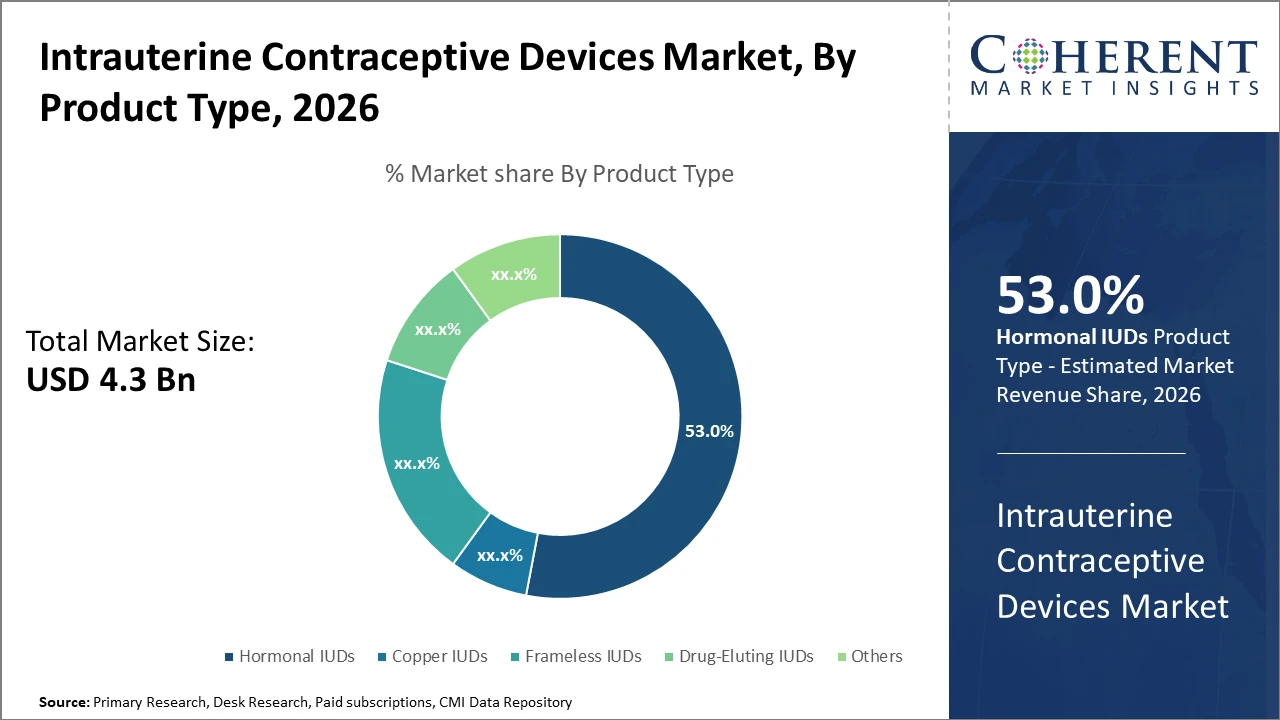
The hormonal IUD segment dominates the product market share, accounting for 53%, driven by superior efficacy and user convenience. Copper IUDs, although slower growing, remain essential due to their non-hormonal profile catering to specific user preferences.
Distribution channels such as hospital pharmacies hold significant importance, facilitating around 45% of revenue streams, while online pharmacies are emerging rapidly, especially in urban centers.
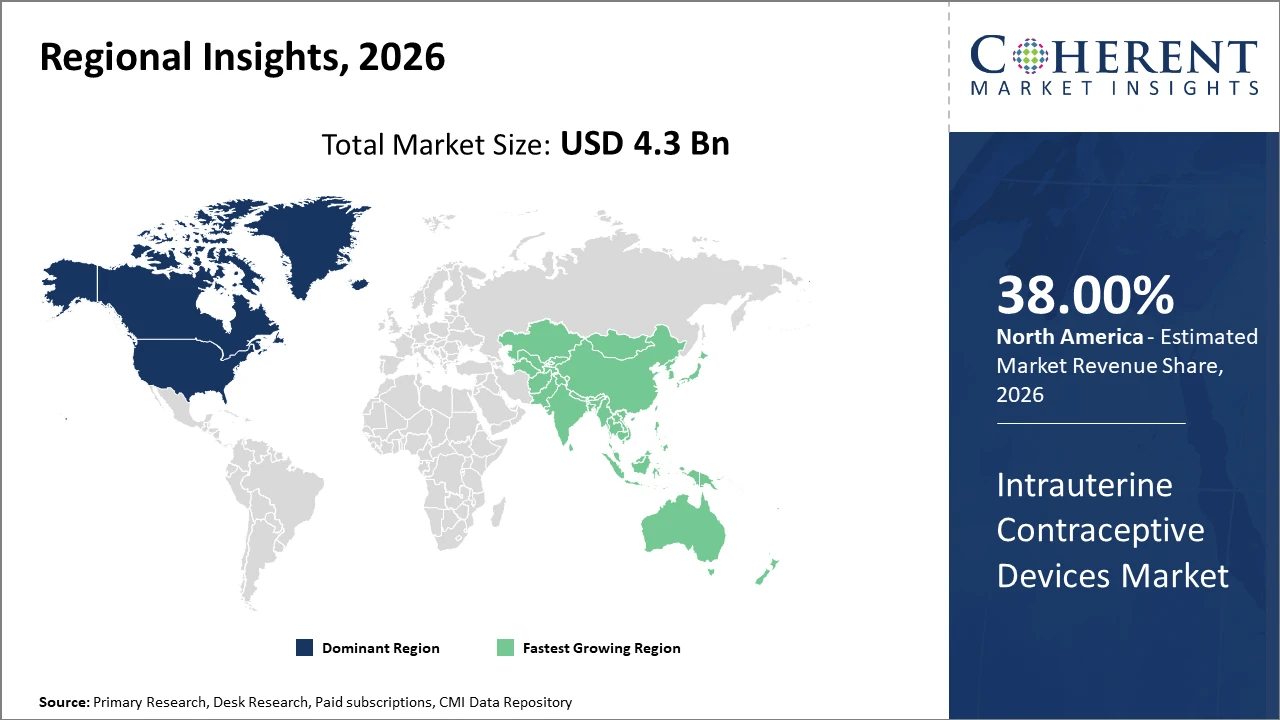
North America leads the regional landscape with a commanding market share of approximately 38%, attributed to advanced healthcare infrastructure and high patient awareness. Asia Pacific, exhibiting the fastest CAGR, benefits from large unmet needs, government initiatives, and expanding healthcare access.
The expanding family planning centers segment reflects increased government investments and NGO participation, propelling broader reach in underpenetrated markets.
Intrauterine Contraceptive Devices Market Segmentation Analysis
To learn more about this report, Request Free Sample
Intrauterine Contraceptive Devices Market Insights, By Product Type
Hormonal IUDs dominate due to their high efficacy, extended action period, and favorable side effect profile. Their growing demand is driven by recent product innovations incorporating levonorgestrel and polymer matrices, which have enhanced hormone release uniformity and minimized adverse events. Copper IUDs remain vital, particularly favored by women preferring hormone-free methods; this segment exhibits slower growth but steady demand due to its affordability and long history of safe usage. Frameless IUDs, though a niche segment, attract interest owing to flexible designs offering better uterine compatibility and reduced expulsion rates. Drug-Eluting IUDs, with emerging bioactive drug combinations, represent a fast-growing niche driven by technological advances.
Intrauterine Contraceptive Devices Market Insights, By End-User
Hospitals maintain dominance due to their capacity for specialized insertion procedures and postoperative care, often enabling higher volumes of surgical intrauterine device insertions. Clinics provide accessible care in urban and suburban settings, capitalizing on affordability and minimal wait times, making them the fastest-growing subsegment owing to expansion in outpatient services and increasing patient preference. Family Planning Centers play a crucial role in public health outreach, especially in lower-income regions, where dedicated counseling and subsidized access promote user acceptance.
Intrauterine Contraceptive Devices Market Insights, By Distribution Channel
Hospital Pharmacies dominate, holding the leading market share by being the primary conduit for intrauterine contraceptive device accessibility and integration into formal healthcare workflows. This segment benefits from bulk procurement and trusted provider recommendations. Retail Pharmacies sustain steady growth supported by improving awareness and metropolitan penetration, catering to convenience-focused consumers and enabling direct purchase options. Online Pharmacies represent the fastest-growing channel as digitization reshapes healthcare delivery; these platforms witnessed over 30% growth in 2026 by facilitating discretion, ease of access, and targeted marketing, especially among younger demographics.
Intrauterine Contraceptive Devices Market Trends
The market has witnessed a notable trend towards the development of multipurpose devices that integrate contraception with therapeutic benefits, such as localized hormone time-release and antimicrobial effects, exemplified by clinical trial successes in 2026.
Additionally, sustainability considerations led to the introduction of biodegradable IUDs, a pioneering trend accelerating adoption particularly among environmentally conscious consumers.
Another dominant trend involves leveraging digital platforms, enabling enhanced patient education and customized contraceptive choices; telehealth consultations related to reproductive health saw a growth rate exceeding 25% in 2025, facilitating market penetration in remote regions.
Intrauterine Contraceptive Devices Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Intrauterine Contraceptive Devices Market Analysis and Trends
In North America, the dominance in the market is attributed to well-established healthcare frameworks, high awareness of contraceptive options, and robust reimbursement schemes. The region accounted for roughly 38% of the industry share as of 2026, supported by stringent regulatory standards that ensure device safety and efficacy. Advanced R&D initiatives and presence of major market players consolidate this regional lead.
Asia Pacific Intrauterine Contraceptive Devices Market Analysis and Trends
Meanwhile, the Asia Pacific exhibits the fastest growth with a CAGR exceeding 10% from 2026 to 2033. Factors such as an expanding reproductive-age population, proactive government family planning policies in countries like India and China, and improving healthcare infrastructure underpin this acceleration. Noteworthy contributions stem from regional manufacturers increasing production capacities and targeted awareness campaigns.
Intrauterine Contraceptive Devices Market Outlook for Key Countries
USA Intrauterine Contraceptive Devices Market Analysis and Trends
The USA’s market benefits from high healthcare access, reimbursement coverage, and advanced technological integration. Manufacturers have focused on improving hormonal IUD offerings, boosting adoption rates to approximately 28% of contraceptive users in 2026. Collaborations between medical centers and key companies foster innovative product launches and educational initiatives, reinforcing market leadership. Regulatory readiness for next-generation devices and consumer preference shifts toward long-lasting reversible contraception fortify continued business growth within the country.
India Intrauterine Contraceptive Devices Market Analysis and Trends
India’s market experiences rapid expansion fueled by government-supported family welfare programs targeting increased contraceptive prevalence. The adoption rate of intrauterine devices surged by around 20% between 2024 and 2026, propelled by rural healthcare outreach and subsidized pricing models. Domestic manufacturers are scaling production, which has significantly lowered costs, enabling widespread market penetration. Government initiatives such as Mission Parivar Vikas accelerated market revenue growth, placing India among the fastest evolving countries in this sector.
Analyst Opinion
The production capacity for advanced inert and hormonal IUDs has expanded by approximately 15% in 2025 compared to 2024, facilitating wider availability. For instance, a leading manufacturer reported a 20% increase in global exports, especially to Asia Pacific, underscoring supply-side enhancements as a pivotal factor influencing the market share growth within the market.
Utilization of intrauterine contraceptive devices across diverse demographic segments, including teenagers and women in the postpartum period, increased by nearly 18% in 2026. This demand-side indicator reflects a larger trend in healthcare accessibility and preference for long-term contraception solutions, which substantially supports the overall market revenue expansion.
Competitive pricing in emerging economies has led to an 11% rise in device adoption during 2025, particularly in Latin America and parts of Asia. Cost-effective product variations combined with government-subsidized healthcare initiatives remain integral to the evolving market landscape and segmentation strategies driving higher penetration rates.
The integration of polymer-based materials and hormone-release mechanisms enhanced product efficacy and reduced side effects by 25% in clinical trials completed in 2026. Such product innovation has fortified consumer confidence, pushing market players to intensify R&D efforts while further expanding intrauterine contraceptive devices market revenue and business growth.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.3 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 7.8 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Bayer AG, CooperSurgical, Inc., Essity Aktiebolag, MedTech Company, Merck KGaA, Ferring Pharmaceuticals, Becton Dickinson and Company, Laboratoire Effik, Teva Pharmaceutical Industries Ltd., Bharat Serums and Vaccines Limited. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Intrauterine Contraceptive Devices Market Growth Factors
Rising awareness on contraception and reproductive health drives adoption substantially, with public health campaigns increasing user uptake by over 20% in multiple regions during 2026. Technological advancements, such as bio-compatible materials and hormone delivery systems, have enhanced device longevity and efficacy, boosting market growth strategies focused on product differentiation. Furthermore, expanding healthcare infrastructure and increasing affordability through government subsidies have removed significant market restraints in emerging economies, motivating continuous market expansion. Lastly, the growing emphasis on women’s empowerment and family planning policies practiced worldwide acted as crucial market drivers, sustained by epidemiological shifts and demographic trends recorded through 2024 and 2025.
Intrauterine Contraceptive Devices Market Development
In February 2025, Sebela Women’s Health received FDA approval for MIUDELLA, marking the first new hormone-free copper intrauterine device (IUD) introduced in the U.S. in more than four decades. MIUDELLA features a flexible nitinol frame and contains less than half the copper used in traditional copper IUDs, a design intended to maintain contraceptive effectiveness while potentially reducing side effects such as cramping and heavy bleeding. Approved for up to three years of use, the device represents a significant advancement in non-hormonal contraception, offering women a modern, lower-copper alternative with improved comfort and flexibility.
In January 2025, 49Care launched Yanae in Canada, a French-invented copper IUD designed to simplify and improve the insertion experience. Yanae incorporates a flexible inserter system that allows for easier, less painful placement and eliminates the need for additional surgical instruments, making the procedure more accessible in outpatient and primary care settings. The launch reflects a growing focus on patient-centric contraceptive innovation, emphasizing comfort, ease of use, and broader adoption of non-hormonal birth control options.
Key Players
Leading Companies of the Market
Bayer AG
CooperSurgical, Inc
Essity Aktiebolag
MedTech Company
Merck KGaA
Ferring Pharmaceuticals
Becton Dickinson and Company
Laboratoire Effik
Teva Pharmaceutical Industries Ltd.
Bharat Serums
Vaccines Limited.
These companies aggressively pursue growth strategies, including product innovation, geographic expansion, and strategic partnerships. For instance, Bayer AG expanded its product portfolio in 2025 by launching next-generation hormonal IUDs exhibiting improved insertion ease, resulting in a 12% sales increase in North America. Similarly, CooperSurgical’s collaboration with regional healthcare centers in Southeast Asia led to a 14% volume growth in 2026.
Intrauterine Contraceptive Devices Market Future Outlook
The future outlook for the Intrauterine Contraceptive Devices Market is driven by rising demand for reliable, long-term, and low-maintenance contraceptive solutions. Ongoing innovation focuses on improving comfort, minimizing side effects, and extending device lifespan. Increased emphasis on women’s reproductive health, combined with supportive healthcare policies, is expected to further expand access to IUDs across both developed and emerging regions. Manufacturers are also exploring new materials and hormone delivery mechanisms to enhance user experience. As education and awareness continue to improve, IUDs are likely to remain a cornerstone of modern contraceptive care.
Intrauterine Contraceptive Devices Market Historical Analysis
The Intrauterine Contraceptive Devices Market has undergone a substantial transformation over several decades, shaped by medical advancements, public health policies, and changing societal attitudes toward family planning. Early IUD designs faced challenges related to safety, comfort, and user acceptance, which limited widespread adoption. Over time, improvements in materials, device geometry, and clinical insertion techniques significantly enhanced safety and effectiveness. The introduction of hormonal IUDs further expanded the market by offering additional benefits such as reduced menstrual bleeding and long-term contraception. Government-supported family planning initiatives and growing awareness of long-acting reversible contraceptives played a critical role in increasing adoption globally.
Sources
Primary Research Interviews:
Gynecologists
Family planning specialists
Reproductive health NGOs
Hospital procurement managers
Device manufacturers
Databases:
WHO Reproductive Health Data
UNFPA Statistics
CDC Contraceptive Use Data
OECD Health Data
Magazines:
Contemporary OB/GYN
Women’s Health Magazine (Clinical)
Medical Device Network
Healthcare Weekly
Global Health Now
Journals:
Contraception Journal
Obstetrics & Gynecology
International Journal of Gynecology & Obstetrics
BMJ Sexual & Reproductive Health
Human Reproduction
Newspapers:
The Guardian (Women’s Health)
Reuters Health
The New York Times (Health)
Financial Times (Healthcare)
BBC Health
Associations:
International Planned Parenthood Federation
American College of Obstetricians and Gynecologists
WHO
UNFPA
FIGO
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients