Orphan Drugs Market Size and Forecast – 2026 – 2033
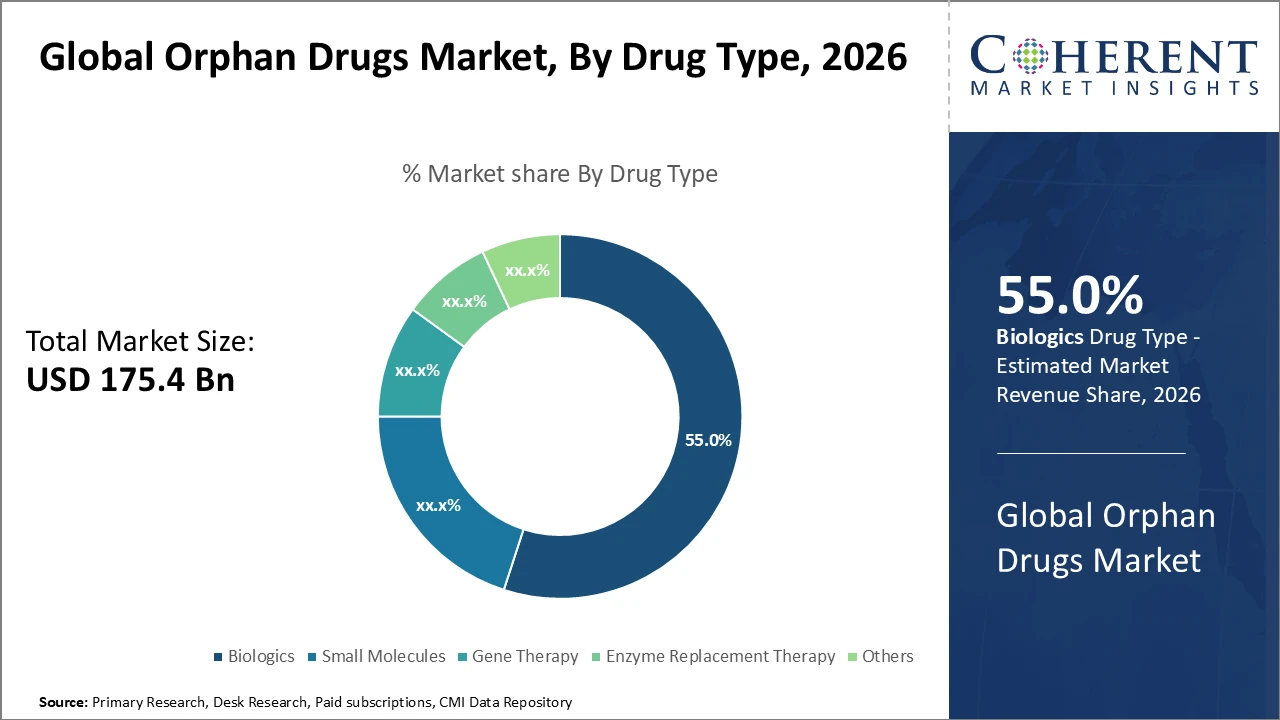
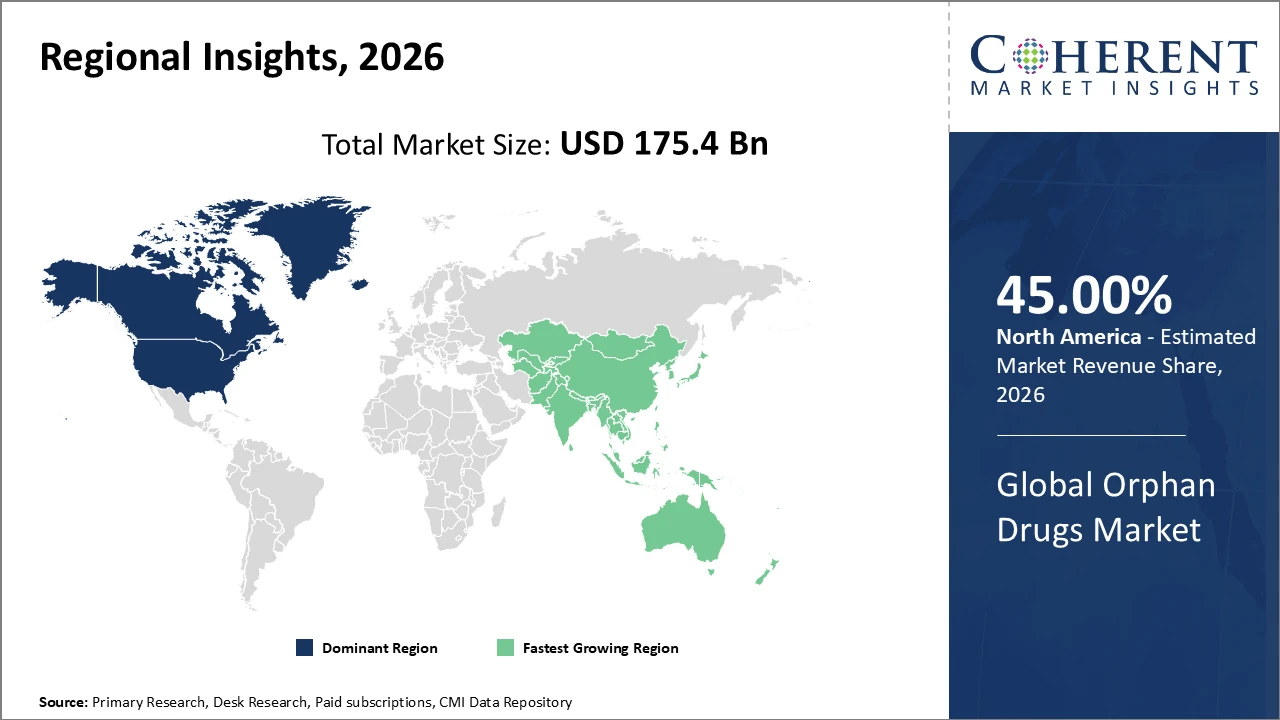
The Global Orphan Drugs Market size is estimated to be valued at USD 175.4 billion in 2026 and is expected to reach USD 320.8 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 9.3% from 2026 to 2033.
Global Orphan Drugs Market Overview
The orphan drugs market focuses on developing treatments for rare diseases that affect small patient populations. Governments and regulatory bodies provide incentives such as tax credits, market exclusivity, and faster approvals to encourage pharmaceutical companies to invest in these therapies. The increasing prevalence of rare diseases, advancements in biotechnology, and growing awareness among healthcare providers are driving market growth.
Key Takeaways
Oncology remains the dominant therapeutic area, accounting for over 40% of the orphan drugs market share, driven by intensive R&D in rare cancers. Neurology is the fastest-growing segment, with a CAGR above 10%, supported by advancements in gene therapies targeting neurodegenerative disorders.
Biologics lead as the primary drug type, holding a 55% market share due to their technological advantages and clinical effectiveness. Gene therapies are the fastest-growing drug segment, propelled by revolutionary treatment approaches for rare diseases.
North America holds the largest regional market share at around 45%, supported by strong regulatory frameworks and high healthcare spending. Asia Pacific exhibits the fastest growth, with a CAGR exceeding 11%, driven by government initiatives and increased rare disease diagnosis rates.
Orphan Drugs Market Segmentation Analysis
To learn more about this report, Request Free Sample
Orphan Drugs Market Insights, By Drug Type
Biologics dominate the orphan drugs market, driven by their superior efficacy and an expanding pipeline targeting complex rare diseases. They form the cornerstone of orphan drug development, supported by extensive R&D investment. Gene therapy is the fastest-growing segment, fueled by breakthroughs in genetic modification and personalized medicine, with multiple recent regulatory approvals enhancing commercial viability. Small molecules continue to play a vital role due to established production processes and cost advantages, primarily addressing well-characterized rare diseases. Enzyme replacement therapies remain crucial for specific metabolic disorders, offering life-changing treatments despite a narrower application scope. The “Others” segment includes emerging modalities such as RNA therapies and innovative delivery systems.
Orphan Drugs Market Insights, By Therapeutic Area
Oncology dominates the orphan drugs market, driven by the increasing prevalence of rare cancers and significant unmet medical needs, with multiple breakthrough biologics and immunotherapies receiving regulatory approvals regularly. Neurology is the fastest-growing therapeutic area, fueled by gene therapies and RNA-based treatments targeting neurodegenerative disorders such as spinal muscular atrophy and Duchenne muscular dystrophy. Metabolic disorders show steady growth, focusing on enzyme replacement and substrate reduction therapies for rare inherited conditions. Cardiovascular rare diseases present niche opportunities, mainly addressed through small molecule interventions. The “Others” category encompasses rare autoimmune and inflammatory disorders with emerging pipeline candidates.
Orphan Drugs Market Insights, By Distribution Channel
Hospital pharmacies dominate the orphan drugs market, serving as primary dispensing points due to the specialized administration and monitoring required for many therapies. The fastest-growing distribution subsegment is online pharmacies, driven by the expansion of telemedicine and the post-COVID-19 shift toward convenient patient access, particularly in developed and emerging markets. Retail pharmacies continue to grow steadily, supported by increasing awareness of rare diseases and improved local availability. The “Others” segment includes specialty pharmacies and direct-to-patient delivery models, which are increasingly adopted by market players to enhance patient compliance, ensure timely treatment, and expand overall market penetration.
Orphan Drugs Market Trends
The orphan drugs market is increasingly driven by advanced biologics and gene therapies targeting previously untreatable rare diseases.
In 2025, gene editing therapies received accelerated regulatory approvals, reflecting a maturing pipeline and growing commercial viability.
Evolving regulatory frameworks in North America and Europe provide incentives that shorten time-to-market, boosting market share for innovative companies.
Expansion into emerging markets, particularly Asia Pacific, is a key trend, supported by government funding and awareness campaigns.
Rare disease diagnoses in the region increased by over 20% year-over-year from 2024 to 2026, further enhancing market growth opportunities.
Orphan Drugs Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Orphan Drugs Market Analysis and Trends
In North America, the orphan drugs market is dominated by strong healthcare infrastructure, substantial R&D investments, and supportive regulatory policies, including the Orphan Drug Act. The region holds approximately 45% of the global market share, reinforced by the presence of leading companies headquartered in the U.S. and Canada. Expedited FDA approvals and patient assistance programs have enabled faster commercialization of novel therapies, enhancing patient access and adoption. Additionally, continuous investment in clinical trials and innovative biologics and gene therapies strengthens the market landscape. These factors collectively sustain revenue growth, making North America a pivotal hub for orphan drug development and commercialization.
Asia Pacific Orphan Drugs Market Analysis and Trends
Meanwhile, the Asia Pacific orphan drugs market is experiencing the fastest growth, with a CAGR exceeding 11%, driven by rising healthcare expenditures, government initiatives supporting biopharmaceutical innovation, and increasing patient awareness of rare diseases. Countries such as China and India are rapidly expanding diagnostic infrastructure and reimbursement frameworks, enabling wider access to orphan therapies and improving market penetration. Additionally, major global and regional players are investing in local manufacturing facilities to benefit from cost efficiencies and meet growing demand. These developments position Asia Pacific as a key growth region, with significant opportunities for both established and emerging orphan drug companies.
Orphan Drugs Market Outlook for Key Countries
USA Orphan Drugs Market Analysis and Trends
The U.S. orphan drugs market thrives due to a highly advanced healthcare ecosystem and one of the world’s highest per capita healthcare expenditures, surpassing USD 12,000 in 2025. Regulatory support from the FDA, including active orphan drug programs with over 70 approvals in a single year, has accelerated market growth. Leading companies such as Biogen Inc. and Pfizer have increased investments and pursued strategic collaborations, particularly in neurological and metabolic rare diseases. Strong patient advocacy, comprehensive insurance coverage, and access programs further support adoption. These factors collectively position the U.S. as a central driver of global orphan drug market share and innovation.
Germany Orphan Drugs Market Analysis and Trends
Germany’s orphan drugs industry is bolstered by comprehensive national rare disease policies and strict pharmaceutical regulations that promote early market access. The market has seen accelerated adoption of biologics and enzyme replacement therapies, especially for rare metabolic disorders. Government-supported reimbursement frameworks and collaborations with innovative biotech firms have driven a 14% increase in orphan drug revenue between 2024 and 2026. Leading companies such as Novartis AG and Sanofi S.A. actively operate in the region, leveraging Germany’s robust healthcare infrastructure, strong R&D capabilities, and favorable regulatory environment to expand their presence and deliver advanced therapies to patients with rare diseases.
Analyst Opinion
The surge in clinical trials targeting rare genetic disorders is a major demand-side driver. Over 1,200 orphan drug clinical trials were initiated globally in 2024, a 15% increase from 2023, indicating robust pipeline expansion and growing biopharmaceutical engagement in previously underserved rare disease segments.
Supply-side trends show increased production capacities for innovative biologics and gene therapies. In 2025, a leading biopharmaceutical company expanded its biologics manufacturing facility by 30% to meet orphan drug demand, enabling smaller niche players to scale operations and capture market share.
Pricing structures remain complex due to high development costs and regulatory exclusivity. In 2026, orphan drugs accounted for nearly 25% of specialty pharmaceutical market revenue, highlighting premium pricing and substantial market contribution despite small patient populations.
Imports of orphan drugs in emerging economies like India and Brazil are rising, supported by improved healthcare infrastructure and government policies. Between 2024 and 2026, orphan drug imports in Latin America grew 18% annually, reflecting geographic diversification and market expansion.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 175.4 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.3% | 2033 Value Projection: | USD 320.8 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Biogen Inc., Novartis AG, Pfizer Inc., Sanofi S.A., Roche Holding AG, Amgen Inc., Horizon Therapeutics, Sarepta Therapeutics, Jazz Pharmaceuticals, Vertex Pharmaceuticals | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Orphan Drugs Market Growth Factors
The orphan drugs market is driven by multiple growth factors. Regulatory support from agencies like the FDA and EMA, including expedited review processes and extended market exclusivity, continues to encourage innovation, with FDA approvals reaching a record high of over 70 in 2025. Advances in gene therapy and personalized medicine are expanding treatment options for rare diseases, with gene therapies comprising nearly 20% of global orphan drug pipelines by 2026. Rising patient awareness and advocacy have increased demand, particularly in emerging regions, while growing healthcare expenditure in developed economies, such as the U.S. investing over USD 50 billion in orphan drug research in 2024, reinforces sustainable market growth.
Orphan Drugs Market Development
In February 2026, Lupin announced the launch of NaMuscla (mexiletine) in Germany and the United Kingdom. The drug is approved across the European Union for the symptomatic treatment of myotonia in adults with non-dystrophic myotonic (NDM) disorders, a group of rare inherited neuromuscular conditions characterized by difficulty relaxing muscles after voluntary contraction.
Key Players
Leading Companies of the Market
Biogen Inc.
Novartis AG
Pfizer Inc.
Sanofi S.A.
Roche Holdings AG
Amgen Inc.
Horizon Therapeutics
Sarepta Therapeutics
Jazz Pharmaceuticals
Vertex Pharmaceuticals
Leading orphan drug companies focus on strategic acquisitions to expand their portfolios and target rare disease indications. For example, in 2025, a major biopharmaceutical acquisition enhanced access to gene therapies for neuromuscular disorders, significantly increasing market share in North America and Europe. Additionally, collaborations with research institutions and smaller biotech firms to leverage advanced technologies, including CRISPR and RNA-based therapies, are increasingly common. These partnerships demonstrate proactive growth strategies, enabling companies to maintain competitive advantage, accelerate innovation, and address challenges in the orphan drugs market, ultimately supporting broader patient access to specialized therapies.
Orphan Drugs Market Future Outlook
The orphan drugs market is expected to witness robust growth over the coming years, driven by advances in gene therapy, biologics, and personalized medicine targeting rare diseases. Regulatory incentives, such as expedited approvals and extended market exclusivity, will continue to encourage innovation and pipeline expansion. Rising patient awareness, advocacy, and improved healthcare access in emerging regions will further support adoption. Additionally, increasing investments by pharmaceutical companies in research and development, strategic collaborations, and local manufacturing will enhance market reach. With ongoing technological breakthroughs and expanding global patient populations, the orphan drugs market is poised for sustained growth and innovation.
Orphan Drugs Market Historical Analysis
The orphan drugs market has experienced significant growth over the past decade, driven by rising awareness of rare diseases and supportive regulatory frameworks such as the U.S. Orphan Drug Act and similar policies in Europe. Initially focused on enzyme replacement therapies and small molecules, the market gradually expanded to include biologics and gene therapies, addressing previously untreatable conditions. Increased R&D investments, coupled with patient advocacy and specialized reimbursement programs, fueled adoption across developed and emerging markets. Growing clinical trials and technological advancements in personalized medicine further strengthened the pipeline. These historical developments laid the foundation for the current rapid expansion and innovation in the orphan drugs market.
Sources
Primary Research Interviews:
Hospitals, specialty clinics, and rare disease treatment centers
Pharmaceutical and biopharmaceutical manufacturing facilities
Research laboratories and academic institutions focusing on rare diseases
Medical and biotech consultants specializing in orphan drug development
Magazines:
Pharmaceutical Executive – Rare Disease and Orphan Drug Trends
BioPharma Dive – Orphan Drugs Innovation and Market Insights
Fierce Biotech – Gene Therapy and Rare Disease Coverage
HealthTech Magazine – Biologics and Rare Disease Treatments
Journals:
Orphanet Journal of Rare Diseases
Molecular Therapy
Gene Therapy
Journal of Rare Diseases Research & Treatment
Clinical Therapeutics
Newspapers:
The Wall Street Journal – Biopharmaceuticals & Rare Diseases
Financial Times – Specialty Pharmaceuticals
Reuters – Orphan Drugs and Rare Disease Markets
Bloomberg – Biopharma and Healthcare Industry Updates
Nikkei Asia – Biopharmaceutical and Rare Disease Coverage
Associations:
National Organization for Rare Disorders (NORD)
European Organization for Rare Diseases (EURORDIS)
American Society of Gene & Cell Therapy (ASGCT)
International Society for Rare Diseases and Orphan Drugs (ISROD)
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients