Pigmented Lesion Treatment Market Size and Forecast – 2026 – 2033
The Global Pigmented Lesion Treatment Market is projected to grow significantly, rising from roughly USD 3.6 Billion in 2026 to about USD 7.6 Billion by 2033, expanding at a CAGR of 7.8 % from 2026 to 2033, driven by rising pigmentation concerns and technological innovations in treatments.
Global Pigmented Lesion Treatment Market Overview
The Pigmented Lesion Treatment market focuses on therapies addressing hyperpigmentation, age spots, freckles, melasma, and other skin discolorations caused by genetics, sun exposure, or hormonal changes. Treatments include topical agents, laser therapy, chemical peels, and cryotherapy, targeting melanin reduction and skin tone improvement. Rising aesthetic awareness, increasing dermatological procedures, and technological advancements in laser and light-based therapies are fueling market growth. The market is segmented by treatment type, end-user (clinics, hospitals, dermatology centers), and geography. North America and Asia-Pacific lead adoption due to high cosmetic procedure demand, while innovations in non-invasive therapies continue to drive global expansion.
Key Takeaways
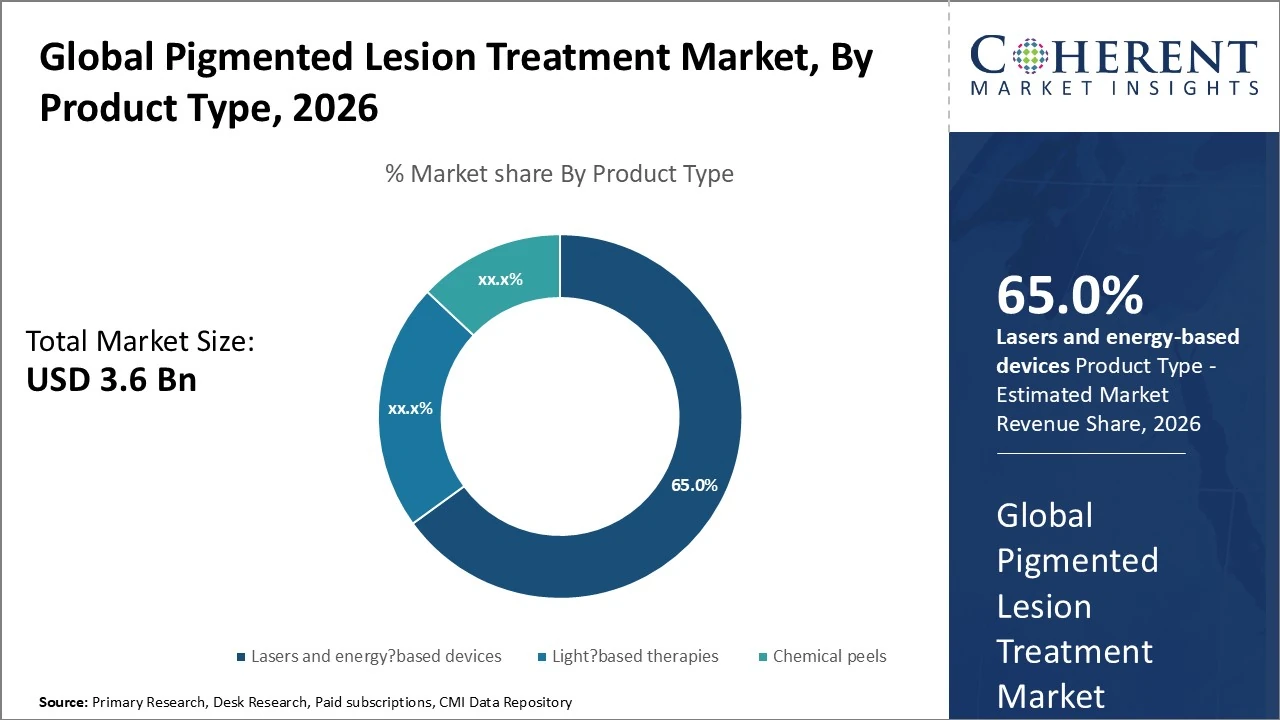
The pigmented lesion treatment market is dominated by lasers and energy‑based devices, holding the largest share at around 65 %.
In the pigmented lesion treatment market, Hospitals account for the largest share (65 %).
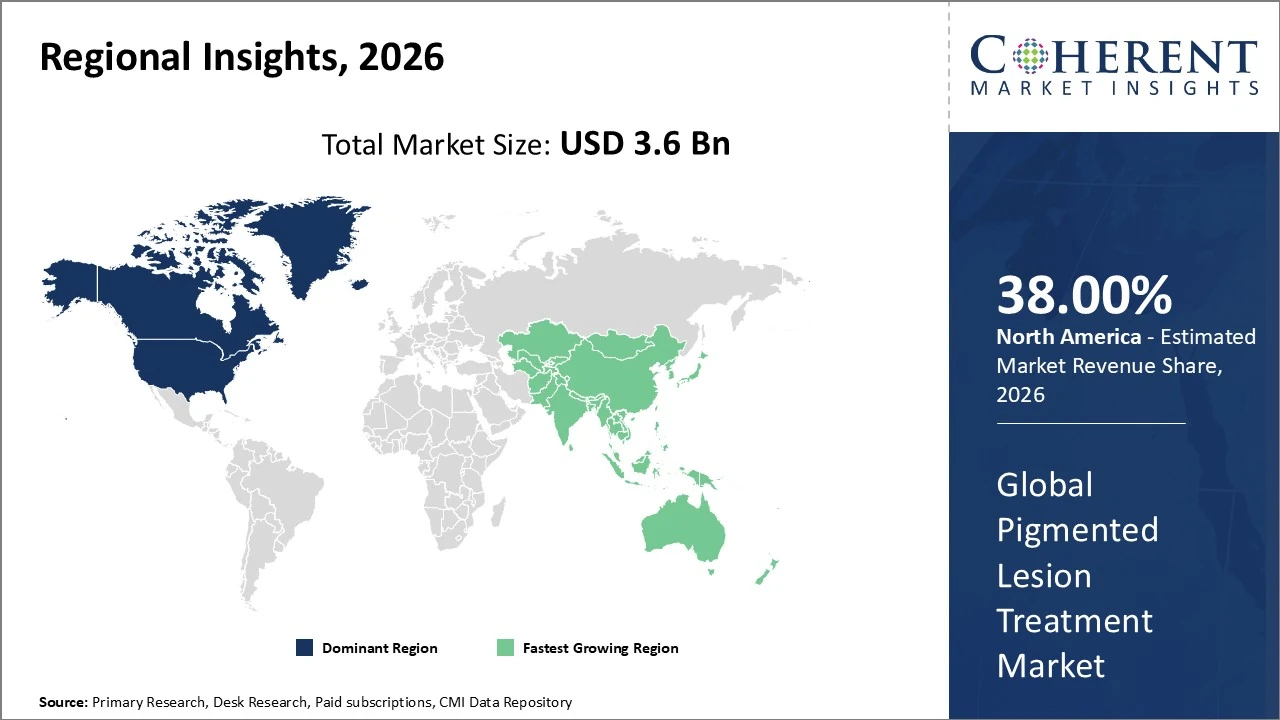
In North America, the pigmented lesion treatment market leads globally, holding 38 % share of total revenues.
In Asia Pacific, the pigmented lesion treatment market is expanding rapidly and held about 21% of global revenue in 2026.
Pigmented Lesion Treatment Market Segmentation Analysis
To learn more about this report, Download Free Sample
Pigmented Lesion Treatment Market Insights, By Product Type
The pigmented lesion treatment market is dominated by Lasers and energy‑based devices, holding the largest share at around 65 % due to high precision and effectiveness in treating melasma, lentigines, and freckles. Light‑based therapies (like IPL devices) and Chemical peels follow with smaller shares, roughly 22 % and 13 % respectively, driven by non‑invasive demand and versatility across skin types. Advanced devices such as radiofrequency and micro‑focused ultrasound are gaining traction, further diversifying the product mix and supporting steady growth in both dermatology clinics and aesthetic centers worldwide.
Pigmented Lesion Treatment Market Insights, By End-User
In the pigmented lesion treatment market, Hospitals account for the largest share (~65 %) due to comprehensive diagnostic and treatment infrastructure and advanced medical equipment. Dermatology clinics hold about ~22 %, offering specialized skin care and targeted pigment disorder therapies in outpatient settings. Homecare and other settings make up roughly ~13 %, driven by rising demand for at‑home devices and topical kits that support maintenance and mild condition management. This segmentation reflects the preference for professional clinical care for complex cases, while consumer‑oriented solutions grow steadily.
Pigmented Lesion Treatment Market Trends
Increasing preference for laser, IPL, and RF-based treatments over surgical options due to minimal downtime and faster recovery.
Emergence of picosecond lasers, combination therapies, and AI-assisted devices is enhancing precision and treatment efficacy.
Rising cosmetic consciousness, social media influence, and skin health awareness are driving adoption in dermatology clinics and aesthetic centers globally.
Pigmented Lesion Treatment Market Insights, By Geography
To learn more about this report, Download Free Sample
North America Pigmented Lesion Treatment Market Analysis and Trends
In North America, the pigmented lesion treatment market leads globally, holding 38 % share of total revenues in 2026, driven by advanced healthcare infrastructure, high aesthetic awareness, and widespread adoption of laser and energy‑based therapies. The U.S. dominates with robust demand for non‑invasive cosmetic procedures and strong clinician adoption of innovative technologies. Canada and Mexico show steady uptake supported by dermatology clinic growth. Trends include integration of AI‑assisted diagnostics, expanded medical spa networks, and rising consumer preference for precision treatments, supporting sustained regional growth through 2033.
Asia Pacific Pigmented Lesion Treatment Market Analysis and Trends
In Asia Pacific, the pigmented lesion treatment market is expanding rapidly and held about 21% of global revenue in 2026, with growth expected to outpace many regions due to rising disposable incomes, expanding dermatology clinics, and heightened aesthetic awareness in countries such as China, India, South Korea, and Japan. The region is projected to see the highest CAGR through the forecast period, driven by increased adoption of laser and non‑invasive therapies, growth in medical tourism, and broader access to advanced treatments. Local manufacturers and digital engagement in beauty care further bolster market momentum.
Pigmented Lesion Treatment Market Outlook for Key Countries
USA Pigmented Lesion Treatment Market Analysis and Trends
In the United States, the pigmented lesion treatment market accounts for the majority of the North American share (the U.S. represents 90 % of the North America hyperpigmentation segment). Growing awareness of skin pigmentation disorders and strong adoption of advanced, non‑invasive technologies like laser, IPL, and AI‑driven diagnostic tools are driving demand. The U.S. hyperpigmentation/pigmented lesion segment is expected to grow at a 5.8 % CAGR through 2033 across energy‑based therapies and topical treatments, supported by high disposable income, robust dermatology infrastructure, and expanding aesthetic clinics. Consumer preference for precision cosmetic care remains a core trend.
Germany Pigmented Lesion Treatment Market Analysis and Trends
In Germany, the pigmented lesion treatment market is growing steadily, supported by its advanced healthcare system and high consumer awareness of skin health. The country accounts for a significant portion of Europe’s share with demand for both clinical and aesthetic solutions. The market is projected to expand at a 7.0 % CAGR through 2026 as non‑invasive laser and topical therapies gain traction. Consumers favour evidence‑based procedures in dermatology clinics, and adoption of innovative energy‑based devices continues to rise, enhancing treatment outcomes.
Analyst Opinion
Analysts project the market to expand at around a 7.8 % CAGR, nearly doubling in value by 2033 due to rising demand for both medical and cosmetic treatments.
Innovation in diagnostic tools like AI‑assisted imaging and dermoscopy is enhancing early detection and treatment precision, boosting clinician confidence.
There’s a shift toward laser, IPL, and energy‑based therapies as patients prefer procedures with less downtime and better cosmetic outcomes.
Growing emphasis on flawless skin and preventive dermatology is pushing more consumers toward pigmented lesion treatments beyond purely medical needs.
Partnerships between device manufacturers and clinics are seen as key to scaling access, improving protocols, and driving personalized treatment solutions.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.6 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.8% | 2033 Value Projection: | USD 7.6 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Lumenis Ltd., Alma Lasers Ltd., Cynosure, Inc., Cutera Inc., Lutronic Corporation, Sciton, Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Generic Pigmented Lesion Treatment Growth Factors
Generic Pigmented Lesion Treatment Growth Factors are driven by multiple converging trends. Rising skin health and cosmetic awareness globally fuels demand for accessible pigmentation therapies. Technological advancements in laser, IPL, and non-invasive devices enhance efficacy, safety, and adoption in dermatology clinics. Increasing prevalence of hyperpigmentation disorders due to sun exposure, aging, and hormonal factors expands the patient pool. Affordable generic topical agents and over-the-counter products improve accessibility, especially in emerging markets. Medical tourism and rising disposable incomes in Asia-Pacific and Latin America support market growth. Combined, these factors create sustained expansion for both clinical and at-home treatment solutions worldwide.
Pigmented Lesion Treatment Market Development
The Pigmented Lesion Treatment Market is undergoing rapid development, driven by innovations in laser, intense pulsed light (IPL), radiofrequency, and combination therapies that improve precision and minimize downtime. Companies are investing in R&D for advanced devices and topical formulations, while regulatory approvals and clinical studies are expanding treatment options. The market is also evolving with digital dermatology tools, AI-assisted diagnostics, and teledermatology for personalized care. Increasing consumer demand for aesthetic procedures, along with rising prevalence of hyperpigmentation and aging-related skin conditions, is encouraging new entrants and partnerships. Emerging regions, particularly Asia-Pacific, are experiencing accelerated adoption, supporting global market expansion.
Key Players
Leading Companies of the Market
Lumenis Ltd.
Alma Lasers Ltd.
Cynosure Inc.
Cutera Inc.
Lutronic Corporation
Sciton, Inc.
Pigmented Lesion Treatment Market Future Outlook
The future outlook of the Pigmented Lesion Treatment Market is highly promising, driven by technological innovation, rising aesthetic awareness, and expanding healthcare access. Energy-based devices like picosecond lasers and advanced IPL systems are expected to dominate, offering precise, non-invasive treatments with minimal downtime. The market will benefit from growing adoption in emerging regions, increased dermatology clinic networks, and integration of AI-assisted diagnostics for personalized care. Demand for affordable topical therapies and combination treatments will continue to rise. Analysts anticipate steady CAGR growth (7.5 %) through 2033, positioning the market for sustained global expansion and innovation in both clinical and cosmetic segments.
Pigmented Lesion Treatment Market Historical Analysis
The historical analysis of the Pigmented Lesion Treatment Market shows steady growth over the past decade, driven by increasing prevalence of hyperpigmentation disorders, aging populations, and rising cosmetic awareness. Initially dominated by topical agents and chemical peels, the market shifted toward laser and energy-based devices after technological advancements improved treatment precision and reduced recovery time. North America and Europe led adoption, supported by advanced healthcare infrastructure and strong dermatology networks. Early market growth was also influenced by regulatory approvals, clinical research, and consumer education, setting the foundation for current innovations and the expanding global demand seen today.
Sources
Primary Research Interviews:
Dermatologists and Cosmetic Surgeons
Hospital and Clinic Administrators
Medical Device Manufacturers
Pharmaceutical Companies
Databases:
PubMed / MEDLINE
ClinicalTrials.gov
World Health Organization (WHO)
Journals:
Journal of the American Academy of Dermatology
Dermatologic Surgery
Lasers in Surgery and Medicine
International Journal of Dermatology
Newspapers:
The Wall Street Journal
Financial Times
The Economic Times
The New York Times
Associations:
American Academy of Dermatology (AAD)
European Academy of Dermatology and Venereology (EADV)
International Society for Dermatologic Surgery (ISDS)
Society for Investigative Dermatology (SID)
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients