Pompe Disease Therapeutic Market Size and Forecast – 2026 – 2033
The Global Pompe Disease Therapeutic Market size is estimated to be valued at USD 1.36 billion in 2026 and is expected to reach USD 3.22 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 12.9% from 2026 to 2033.
Global Pompe Disease Therapeutic Market Overview
The Pompe Disease Therapeutic Market primarily includes enzyme replacement therapies (ERTs), pharmacological chaperones, gene therapies, and supportive care products. Key ERTs such as alglucosidase alfa help replace deficient acid alpha-glucosidase enzyme, improving muscle function and slowing disease progression. Next-generation ERTs are being developed to enhance tissue targeting and reduce immune responses. Pharmacological chaperones aim to stabilize enzyme activity, improving cellular uptake and effectiveness. Gene therapy products, currently in clinical development, focus on delivering functional GAA genes for long-term correction. Additionally, supportive treatments like respiratory aids and cardiac management therapies complement primary drugs, improving quality of life for patients with Pompe disease.
Key Takeaways
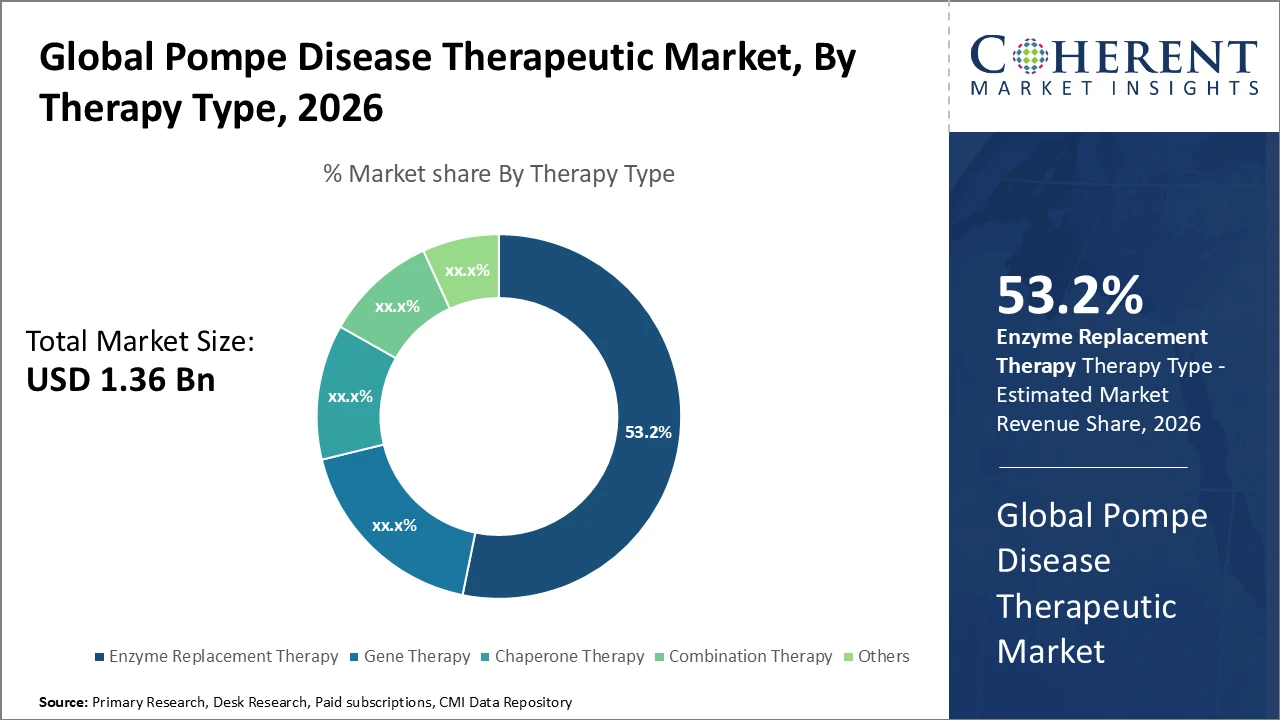
Among therapy types, Enzyme Replacement Therapy dominates with over 53.2% industry share owing to established clinical efficacy and regulatory approvals, while Gene Therapy is the fastest-growing subsegment driven by recent breakthrough designations.
Infantile-Onset Pompe Disease holds a significant market opportunity attributed to higher unmet medical needs and rapid progression rate.
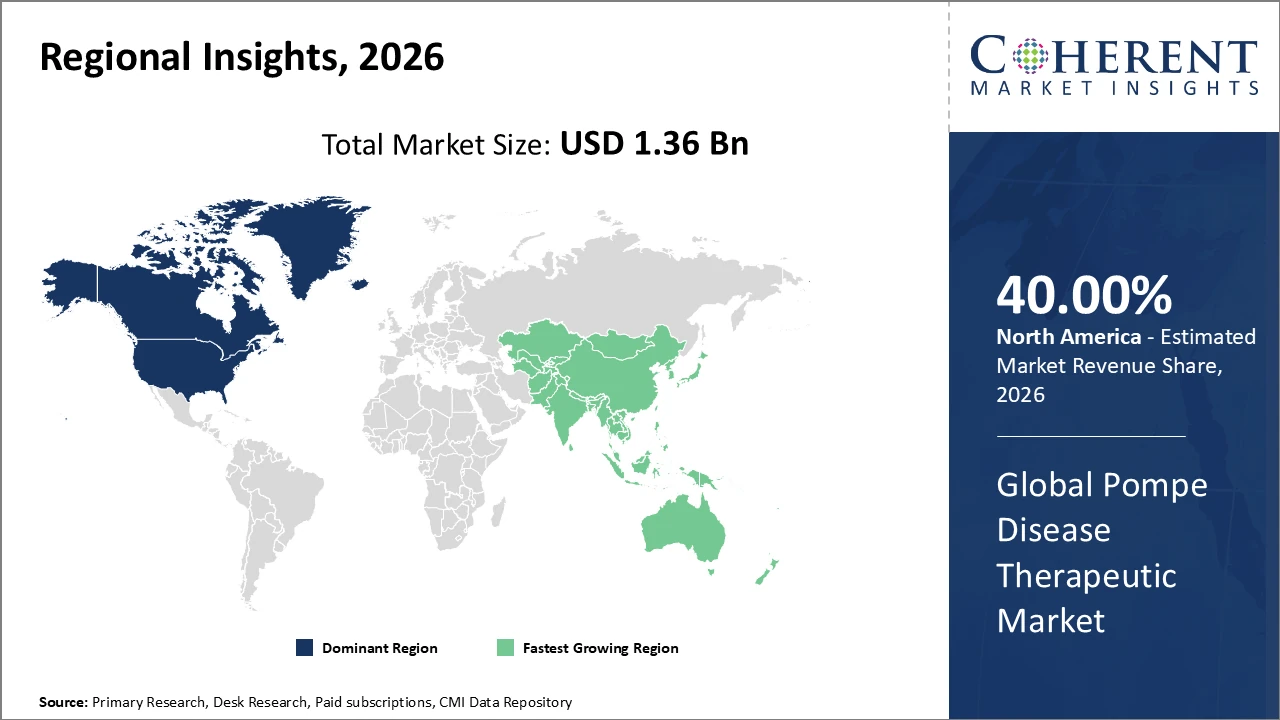
North America leads the regional market share, supported by robust biotech ecosystems and advanced healthcare infrastructure.
Asia Pacific demonstrates the highest CAGR due to expanding healthcare access and lucrative market entry incentives in countries like China and India.
Pompe Disease Therapeutic Market Segmentation Analysis
To learn more about this report, Request Free Sample
Pompe Disease Therapeutic Market Insights, By Therapy Type
Enzyme Replacement Therapy dominates the market with a 53.2% share, driven by strong clinical efficacy, regulatory approvals, and widespread adoption in both infantile and late-onset Pompe disease. It remains the standard of care by alleviating symptoms through enzyme supplementation and improving patient outcomes. Gene Therapy is the fastest-growing segment, supported by breakthrough designations and rising clinical trials targeting the root genetic cause, with potential curative benefits. Chaperone Therapy and Combination Therapy are emerging approaches gaining interest for improving enzyme stability and enhancing therapeutic response. Other experimental therapies are in early development, offering future diversification in Pompe disease treatment options.
Pompe Disease Therapeutic Market Insights, By Disease Type
Infantile-Onset Pompe Disease (IOPD) dominates the market share due to its aggressive progression and need for immediate, intensive therapeutic intervention, contributing significantly to market revenue. Expanded newborn screening programs have improved early diagnosis and increased treatment initiation rates, further strengthening its market position. Late-Onset Pompe Disease (LOPD) is the fastest-growing subsegment, driven by rising awareness and improved diagnostic capabilities that identify a broader patient population. The “Others” category, including variant and atypical presentations, currently holds a smaller share but is gradually expanding as disease heterogeneity becomes better understood. Overall, IOPD drives urgent treatment demand, while LOPD supports long-term growth opportunities.
Pompe Disease Therapeutic Market Insights, By Distribution Channel
Hospital Pharmacy dominates the market share due to the complexity of Pompe disease therapies that require administration in controlled clinical environments. Hospitals ensure proper monitoring, safety, and adherence support, which is critical for achieving optimal therapeutic outcomes in patients. Online Pharmacy is the fastest-growing segment, driven by expanding digital health platforms, increased patient preference for home delivery, and rising adoption of telemedicine services, especially following post-2024 healthcare recovery trends. Retail Pharmacy and other distribution channels maintain steady contributions by supporting outpatient needs and supplementary access. Growing digital infrastructure is expected to gradually reshape distribution dynamics, enhancing convenience and continuity of care.
Pompe Disease Therapeutic Market Trends
The Pompe Disease Therapeutic Market is witnessing strong innovation-led growth, driven by advances in treatment technologies and research.
Gene therapy has significantly transformed the treatment landscape, with multiple clinical trials completed by 2026 showing improved patient survival rates and slower disease progression.
Expansion of diagnostic infrastructure, including newborn screening programs in the U.S. and Europe, has enabled earlier diagnosis and timely treatment initiation, boosting market growth.
Early intervention resulting from improved screening directly increases treatment adoption and overall market revenue.
Personalized medicine is emerging as a key trend, with therapies increasingly tailored to individual genetic profiles, improving treatment effectiveness and patient acceptance.
Pompe Disease Therapeutic Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Pompe Disease Therapeutic Market Analysis and Trends
North America dominates the Pompe Disease Therapeutic Market due to its advanced healthcare infrastructure, strong regulatory framework for orphan drugs, and the presence of major pharmaceutical companies such as Sanofi Genzyme and Amicus Therapeutics. In 2026, the region accounted for over 40% of the global market share. Well-established biotech clusters in California and Massachusetts support extensive clinical research, innovation, and faster commercialization of new therapies. Additionally, favorable reimbursement policies for high-cost treatments enhance patient access and adoption rates. Strong government incentives and continued investment in rare disease research further reinforce North America’s leading position in the global Pompe disease therapeutic market.
Asia Pacific Pompe Disease Therapeutic Market Analysis and Trends
The Asia Pacific region is the fastest-growing market for Pompe Disease Therapeutics, registering a CAGR exceeding 15%. Growth is driven by rising healthcare expenditure, improved diagnostic capabilities for rare diseases, and increasing awareness among healthcare providers and patients. Countries such as China and India are implementing supportive policies, including streamlined regulatory approvals and incentives for orphan drug development, which attract global pharmaceutical investments. The establishment of specialized rare disease centers further strengthens treatment access and diagnosis rates. Additionally, collaborations between regional pharmaceutical companies and international players are enhancing market penetration, accelerating innovation, and expanding availability of advanced Pompe disease therapies across the region.
Pompe Disease Therapeutic Market Outlook for Key Countries
USA Pompe Disease Therapeutic Market Analysis and Trends
The USA remains the largest contributor to the Pompe Disease Therapeutic Market due to comprehensive newborn screening programs, accelerated FDA approvals, and high patient awareness. In 2025, enzyme replacement therapy adoption increased by approximately 22%, reflecting stronger diagnosis rates and treatment accessibility. The country also witnessed a rise in gene therapy clinical trial initiations, supporting long-term innovation in rare disease management. Leading companies such as Sanofi Genzyme and Amicus Therapeutics maintain a strong market presence, driving revenue growth through robust pipelines, strategic expansions, and targeted awareness campaigns. Overall, the U.S. continues to anchor global market development and therapeutic advancements.
Germany Pompe Disease Therapeutic Market Analysis and Trends
Germany’s Pompe Disease Therapeutic Market demonstrates strong growth supported by a well-established reimbursement framework and stringent rare disease regulations guided by the European Medicines Agency. In 2026, market expansion has been driven by rising participation in gene therapy clinical trials and the strengthening of hospital pharmacy networks specializing in neuromuscular disorders. Increased government and private investment in research infrastructure further supports innovation and therapeutic development. Additionally, close collaboration between academic institutions and pharmaceutical companies enhances clinical research efficiency and accelerates drug development. As a result, Germany continues to serve as a key European hub for Pompe disease therapeutic advancements and adoption.
Analyst Opinion
The increasing adoption of novel gene therapy approaches is a key supply-side factor driving expansion of the Pompe Disease Therapeutic Market. In 2025, the first gene therapy candidates for Pompe disease received breakthrough therapy designation by the FDA, leading to increased investments in production capacity by biotech firms focused on rare diseases.
Demand-side dynamics show rising patient diagnosis rates due to expanded newborn screening programs. In the U.S., newborn screening for Pompe disease increased by 18% in 2024 compared to 2023, which significantly boosted therapeutic demand and market revenue.
Micro-level indicators such as improved enzyme efficacy and reduced immunogenicity are enhancing patient compliance and long-term treatment retention. Clinical outcomes in 2026 report a 25% improvement in motor function milestones among treated infantile-onset Pompe patients, highlighting strong therapeutic value and growth potential.
Nano-level indicators reflect growing diversification in regional trade flows, particularly between North America and Europe. In 2025, export volumes of enzyme replacement therapies from North America to Asia Pacific increased by 30%, strengthening cross-border supply chains and reinforcing global market expansion.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.36 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.9% | 2033 Value Projection: | USD 3.22 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Sanofi Genzyme, Lysogene, CSL Behring, Regenexbio, AveXis, Moderna Therapeutics, Takeda Pharmaceuticals, Orchard Therapeutics, Sarepta Therapeutics, Rocket Pharmaceuticals | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Pompe Disease Therapeutic Market Growth Factors
Innovations in gene therapy techniques are a key driver of the Pompe Disease Therapeutic Market, with multiple orphan drug designations granted in 2025 enabling faster regulatory approvals and accelerating product development. Improved diagnostic tools, including next-generation sequencing, have increased detection rates of Pompe disease globally, expanding the diagnosed patient pool and overall market potential. Enhanced reimbursement frameworks in major regions such as the U.S. and Europe are reducing patient financial burden and significantly boosting treatment adoption and revenue growth. Additionally, rising healthcare infrastructure investments in emerging economies, particularly across Asia Pacific, are improving access to advanced therapies and supporting broader market expansion.
Pompe Disease Therapeutic Market Development
In August 2023, the UK Medicines and Healthcare products Regulatory Agency (MHRA) approved Amicus Therapeutics’ combination therapy—Pombiliti (cipaglucosidase alfa) with Opfolda (miglustat)—for treating adults with late-onset Pompe disease.
Key Players
Leading Companies of the Market
Sanofi Genzyme
Takeda Pharmaceuticals
AveXis
Rogenxbio
Lysogene
Rocket Pharmaceuticals
CSL Behring
Orchard Therapeutics
Moderna Therapeutics
Sarepta THerapeutics
Several market players are increasingly adopting advanced gene editing and delivery platforms to accelerate pipeline development in the Pompe Disease Therapeutic Market. For instance, Sanofi Genzyme’s strategic collaborations with multiple biotech startups have significantly strengthened its gene therapy pipeline, contributing to an estimated 35% increase in clinical trial activity in 2025. Similarly, Amicus Therapeutics has pursued an acquisition-driven strategy focused on small molecule chaperone therapies, which has expanded its product portfolio and enhanced its competitive positioning. This approach is estimated to have boosted the company’s market revenue by around 20% in 2024, reflecting strong growth through diversification and targeted therapeutic innovation.
Pompe Disease Therapeutic Market Future Outlook
The Pompe Disease Therapeutic Market is expected to witness strong growth in the future, driven by advancements in gene therapy, enzyme replacement technologies, and personalized medicine approaches. Increasing adoption of newborn screening programs will enable earlier diagnosis and expand the treated patient population globally. Regulatory support for orphan drugs and accelerated approval pathways will further enhance market entry for innovative therapies. Rising investments in rare disease research, particularly in North America and Asia Pacific, will strengthen the development pipeline. Additionally, improved reimbursement policies and expanding healthcare infrastructure will support broader access to high-cost therapies, ensuring sustained market expansion through 2033.
Pompe Disease Therapeutic Market Historical Analysis
The Pompe Disease Therapeutic Market has evolved significantly over the past decades, transitioning from limited supportive care to advanced disease-modifying therapies. Historically, treatment options were restricted until the introduction of enzyme replacement therapy, which marked a major breakthrough in improving patient survival and quality of life. Over time, increasing awareness of rare diseases and advancements in genetic research enabled better diagnosis and classification of Pompe disease subtypes. The expansion of newborn screening programs in developed regions further improved early detection rates. Additionally, growing pharmaceutical investment in orphan drug development has steadily transformed the market, laying the foundation for current innovations in gene therapy.
Sources
Primary Research Interviews:
Clinical researchers, neurologists, and metabolic disorder specialists focusing on Pompe disease diagnosis and treatment outcomes
Hospital pharmacists, genetic counselors, and rare disease program coordinators involved in enzyme replacement therapy administration and patient management
Biotech executives, R&D heads, and clinical trial managers working on gene therapy, enzyme replacement therapy, and pharmacological chaperones for Pompe disease
Magazines:
Nature Medicine – Advances in gene therapy, enzyme replacement innovations, and rare disease breakthroughs
Fierce Biotech – Biopharma pipeline updates, regulatory approvals, and strategic collaborations in rare disease therapies
Endocrine News – Metabolic disorder treatment developments and clinical research updates
Genetic Engineering & Biotechnology News (GEN) – Gene therapy innovations and rare disease therapeutic advancements
Journals:
Journal of Inherited Metabolic Disease – Clinical research on Pompe disease pathophysiology, diagnosis, and treatment
Human Gene Therapy – Gene therapy advancements and clinical outcomes in Pompe disease
Molecular Genetics and Metabolism – Enzyme replacement therapy efficacy and metabolic disorder research
Newspapers:
The New York Times – Healthcare innovation, rare disease awareness, and treatment accessibility developments
Financial Times – Biopharma industry investments, orphan drug markets, and regulatory trends
Reuters Health – Global drug approvals, clinical trial updates, and pharmaceutical market movements
Bloomberg – Pharmaceutical company strategies, market performance, and biotech funding trends
Associations:
International Pompe Association – Patient advocacy, awareness, and global Pompe disease support initiatives
American College of Medical Genetics and Genomics (ACMG) – Genetic testing guidelines and rare disease diagnostics standards
European Medicines Agency (EMA) – Regulatory approvals and orphan drug frameworks for Pompe disease treatments
Orphanet – Rare disease classification, epidemiology, and therapeutic information database
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients