Specialty CROs Market Size and Forecast – 2026 – 2033
The Global Specialty CROs Market size is estimated to be valued at USD 14.7 billion in 2026 and is expected to reach USD 28.9 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 10.5% from 2026 to 2033.
Global Specialty Cros Market Overview
The Specialty CROs market offers a range of specialized services supporting clinical trials and drug development processes. Key offerings include clinical trial management, patient recruitment, data management, biostatistics, and regulatory consulting tailored to specific therapeutic areas such as oncology, cardiology, and rare diseases. Services also cover laboratory testing, pharmacovigilance, and real-world evidence generation. Advanced solutions like decentralized trials, digital monitoring, and AI-driven analytics enhance efficiency and accuracy. Specialty CROs focus on niche expertise, enabling pharmaceutical and biotechnology companies to streamline development timelines, reduce costs, and improve success rates while meeting stringent regulatory requirements across global markets.
Key Takeaways
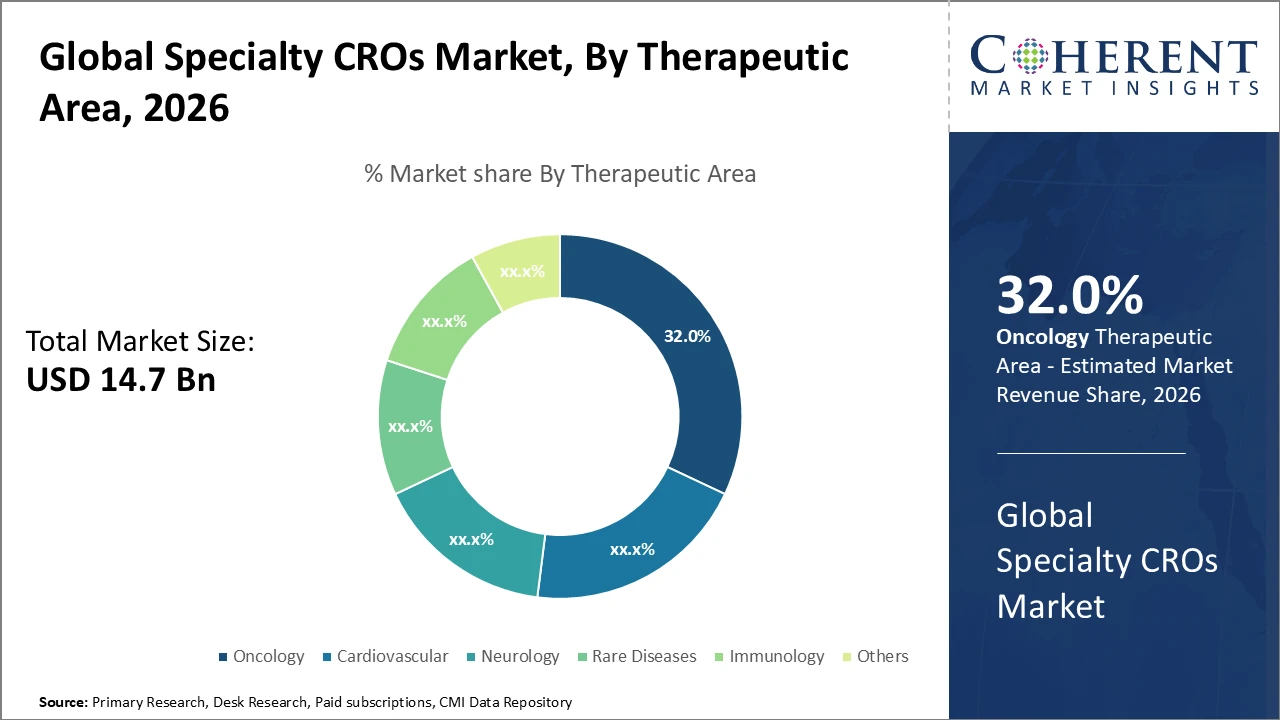
In the therapeutic area segment, oncology leads with around 32% market share, driven by rising cancer prevalence and increased investment in oncology drug development.
In the service type segment, clinical trial management dominates with approximately 38% share, supported by demand for comprehensive operational execution.
Pharmacovigilance is the fastest-growing service segment, driven by stringent regulatory requirements and increased emphasis on drug safety monitoring.
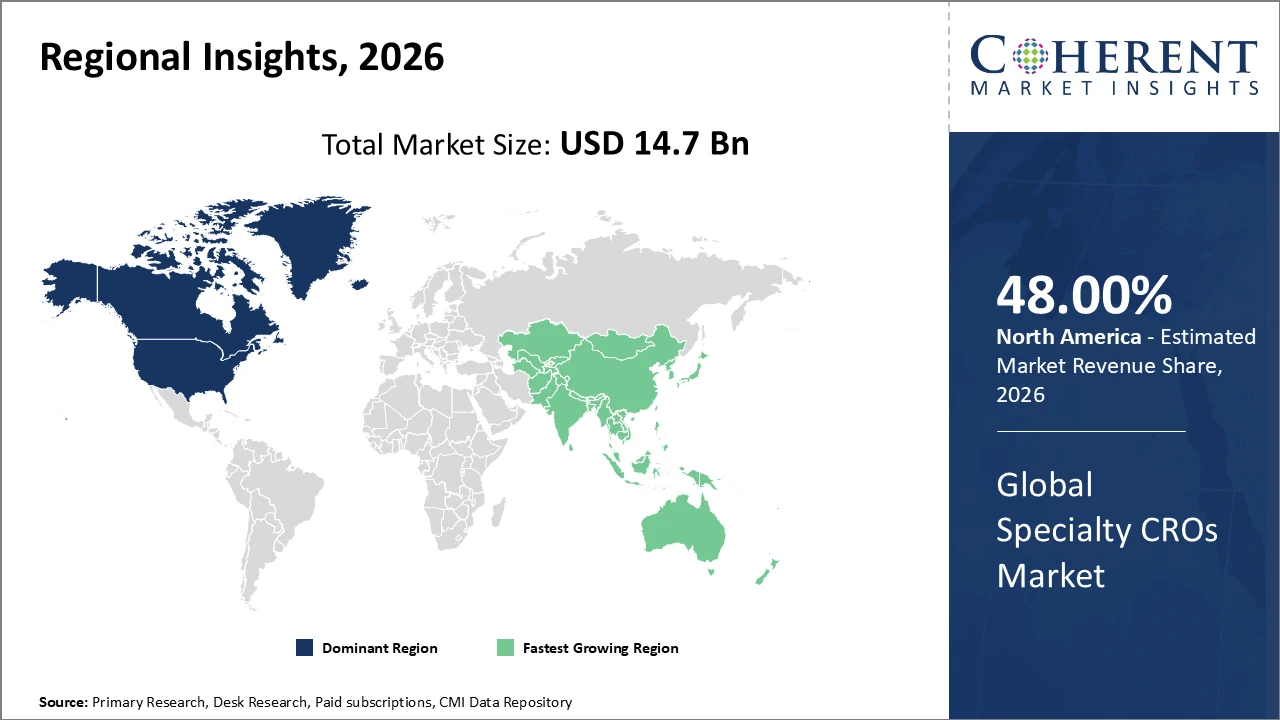
North America holds the largest regional share at about 48%, supported by a strong pharmaceutical ecosystem and high clinical trial activity.
Asia Pacific is the fastest-growing region, with a CAGR exceeding 14%, fueled by expanding clinical trial infrastructure and favorable government initiatives in countries like China and India.
Specialty CROs Market Segmentation Analysis
To learn more about this report, Request Free Sample
Specialty CROs Market Insights, By Therapeutic Area
Oncology dominates the Specialty CROs market with a 32% share, driven by strong R&D investments and a robust pipeline of targeted therapies. Specialized oncology CROs support complex biomarker-driven trials and adaptive study designs, making them highly in demand. Rare diseases represent the fastest-growing subsegment due to increasing orphan drug approvals and supportive regulatory incentives, creating demand for expertise in small patient populations. Cardiovascular remains a stable segment due to the chronic nature of related conditions and ongoing device trials. Neurology services are expanding with new treatments for Alzheimer’s and Parkinson’s, while immunology gains traction through advancements in autoimmune disease therapies.
Specialty CROs Market Insights, By Service Type
Clinical Trial Management dominates the Specialty CROs market with a 38% share, driven by the need for integrated coordination across patient recruitment, site management, and monitoring, particularly in complex specialty trials. Increasing adoption of digital tools has enhanced operational efficiency and data accuracy. Pharmacovigilance is the fastest-growing subsegment, fueled by stricter post-market surveillance requirements and accelerated drug approval timelines. Regulatory Affairs continues steady growth, supporting navigation of multi-jurisdictional approvals and evolving regulatory pathways. Data Management services form the backbone of clinical trials, with growing emphasis on big data analytics and real-world evidence. Other services include consulting and training, complementing core offerings.
Specialty CROs Market Insights, By End User
Pharmaceutical companies dominate the Specialty CROs market, driven by large R&D budgets and extensive drug pipelines that increase reliance on outsourced specialized services. Their focus on therapeutic expertise and global clinical trials further strengthens demand. Biotechnology companies are the fastest-growing segment, fueled by advancements in gene and cell therapies that require customized trial designs and specialized monitoring. Medical device manufacturers contribute steady growth as regulatory requirements for device trials become more complex and innovation accelerates. The “Others” segment, including academic institutions and government research organizations, maintains a consistent presence, supporting niche studies and contributing to overall market revenue growth.
Specialty CROs Market Trends
Innovation and specialization are key drivers shaping the Specialty CROs market, with increasing focus on therapeutic expertise and advanced technology integration.
The growing pipeline of biologics and rare disease drugs has pushed CROs to develop niche capabilities and specialized services.
By 2025, over 30% of specialty CRO revenues were generated from oncology and immunology segments, highlighting strong demand in these areas.
Decentralized clinical trials gained traction, accounting for nearly 25% of projects in 2024, driven by digital transformation and remote trial capabilities.
North America dominates the market with approximately 48% share, supported by a mature pharmaceutical industry, strong clinical trial infrastructure, and high R&D spending.
The United States leads regionally with high clinical trial volumes and a supportive regulatory framework for innovation.
Asia Pacific is the fastest-growing region, with a CAGR exceeding 14%, driven by rising clinical trials in China and India.
Government incentives, expanding patient pools, and increasing clinical trial sites in Asia Pacific further boost market growth.
Leading companies are expanding capacity and forming strategic partnerships in high-growth regions to capitalize on emerging opportunities.
Specialty CROs Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Specialty CROs Market Analysis and Trends
In North America, specialty CROs benefit from the strong presence of pharmaceutical giants and leading biotechnology hubs, which continuously drive demand for specialized clinical trial services. The region’s dominance is reinforced by stringent regulatory frameworks and innovation-driven drug development pipelines. U.S.-based specialty CROs contribute nearly 60% of the regional market revenue, supported by high contract volumes and significant investments in advanced clinical data management and analytics systems. Additionally, widespread adoption of decentralized trials and digital technologies enhances operational efficiency. These factors collectively strengthen North America’s leadership position and ensure sustained growth in the specialty CROs market.
Asia Pacific Specialty CROs Market Analysis and Trends
Asia Pacific is the fastest-growing region in the specialty CROs market, driven by the expansion of clinical trial sites and increasing outsourcing by multinational pharmaceutical companies. Favorable government policies in countries like China and India support clinical research activities and encourage foreign investments. Ongoing healthcare reforms and rising R&D expenditures improve trial infrastructure, enhance patient recruitment efficiency, and reduce operational costs. The availability of large and diverse patient populations further strengthens the region’s appeal for global trials. These combined factors contribute to a strong double-digit CAGR, positioning Asia Pacific as a key growth hub in the specialty CROs market.
Specialty CROs Market Outlook for Key Countries
USA Specialty CROs Market Analysis and Trends
The U.S. specialty CROs market is strengthened by a high concentration of pharmaceutical R&D centers and a regulatory environment that supports innovation. Over 45% of global specialty CRO revenues are generated from U.S.-based projects, driven largely by oncology, CNS, and rare disease trials. Leading companies such as Syneos Health and IQVIA promote competitive innovation through advanced service offerings. There is also a growing trend toward outsourcing early-phase trials to accelerate drug development timelines. Additionally, increased investment in digital trial methodologies and real-world evidence enhances efficiency, supporting sustained market growth and reinforcing the United States’ leadership in the global specialty CROs market.
Germany Specialty CROs Market Analysis and Trends
Germany’s specialty CROs market is driven by a strong pharmaceutical and biotechnology ecosystem, robust clinical research infrastructure, and high R&D investment. The country’s regulatory framework ensures compliance and quality, encouraging outsourcing of complex clinical trials, particularly in oncology, rare diseases, and immunology. German CROs leverage advanced data management, biostatistics, and regulatory consulting services to support multinational trials, enhancing operational efficiency. Additionally, collaborations with hospitals and research institutions expand trial access and patient recruitment. Emerging trends include adoption of decentralized trials, digital monitoring, and AI-driven analytics, positioning Germany as a key European hub for specialized clinical research and CRO services.
Analyst Opinion
The rising trend of biologics and personalized medicine drives Specialty CROs market growth, as these therapies require highly specialized clinical trials. In 2025, over 40% of newly launched biotech drugs underwent trials managed by specialty CROs, highlighting their role in innovation pipelines.
Demand-side dynamics show increased outsourcing among mid-to-large pharmaceutical companies to reduce operational costs while maintaining data quality. In 2024, outsourcing rates for niche therapeutic area trials rose by 15%, reflecting evolving drug development priorities.
Micro-indicators like the expansion of precision oncology and rare disease portfolios increase trial complexity, necessitating specialized CRO expertise. In 2026, oncology-focused CROs accounted for approximately 32% of total market revenue.
Export activities for region-specific clinical trials have grown, with specialty CROs enhancing cross-border study capabilities. Asia Pacific reported a 20% rise in phase II and III trials outsourced to specialty CROs in 2025, showing significant global integration.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14.7 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.5% | 2033 Value Projection: | USD 28.9 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | ICON plc, Syneos Health, PPD, Inc., Celerion, Novotech, PRA Health Sciences, BioClinica Inc., Chiltern, TFS International, Linical Co. Ltd. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Specialty CROs Market Growth Factors
Three key factors are driving the Specialty CROs market growth. First, the rising complexity of clinical trials, particularly in oncology and rare diseases, has increased demand for specialized expertise and innovative trial designs, with complex protocols rising 28% between 2024 and 2026. Second, heightened global regulatory pressures for patient safety and data integrity require CROs capable of comprehensive compliance management. Third, pharmaceutical and biotech firms are focusing on reducing development timelines and costs, boosting outsourcing to end-to-end specialty CROs. Additionally, expanding clinical trial activities in emerging markets, supported by favorable government policies, have propelled Asia Pacific growth at a 14% CAGR.
Specialty CROs Market Development
In 2025, Syneos Health expanded its oncology-focused clinical trial services across North America, resulting in a 10% increase in contract volumes. The company leveraged advanced data management platforms and adaptive trial designs to enhance operational efficiency, supporting faster patient recruitment and improved protocol adherence in complex cancer studies.
Key Players
Leading Companies of the Market
ICON plc
Syneos Health
PPD Inc.
Celerion
Novotech
TFS International
PRA Health Sciences
Chiltern
BioClinica Inc.
Linical Co. Ltd.
Several leading companies in the specialty CROs market have pursued differentiation strategies emphasizing therapeutic specialization and geographic expansion. For instance, ICON plc’s strategic acquisition of a precision medicine CRO in 2025 led to a 12% increase in oncology-related revenues, strengthening its position in high-demand therapeutic areas. Similarly, Syneos Health expanded its clinical development services in the Asia Pacific region through joint ventures, which enhanced its regional market share by over 18% in 2024. These strategic initiatives demonstrate how targeted acquisitions and regional partnerships enable companies to capture niche opportunities and drive sustained growth in the competitive specialty CROs market.
Specialty CROs Market Future Outlook
The Specialty CROs market is expected to witness robust growth driven by increasing demand for complex and niche therapeutic trials, particularly in oncology, rare diseases, and gene therapies. Advances in digital technologies, decentralized trials, and AI-driven data analytics are set to enhance trial efficiency, patient recruitment, and real-world evidence generation. Rising outsourcing by pharmaceutical and biotechnology companies aiming to reduce costs and accelerate time-to-market will further fuel market expansion. Emerging markets in Asia Pacific and Latin America will continue to gain prominence due to favorable regulatory policies, expanding patient populations, and improved clinical trial infrastructure, supporting a strong long-term growth trajectory.
Specialty CROs Market Historical Analysis
The Specialty CROs market has experienced significant growth over the past decade, driven by increasing complexity in clinical trials and the rise of targeted therapies. Initially, market expansion was fueled by demand for outsourcing in oncology, CNS, and rare disease trials, as pharmaceutical and biotechnology companies sought specialized expertise and operational efficiency. The early 2010s saw the adoption of centralized trial management and regulatory support services, while the mid-2010s introduced digital data management and adaptive trial designs. By 2020, increasing biologics pipelines and emerging market participation further accelerated growth, establishing specialty CROs as critical partners in global drug development.
Sources
Primary Research Interviews:
Executives and operations managers at specialty CROs providing clinical trial services across oncology, rare diseases, and other niche therapeutic areas
Clinical operations directors, trial managers, and procurement officers at pharmaceutical and biotechnology companies outsourcing trial management and regulatory services
Technology providers offering clinical trial management software, data analytics platforms, and decentralized trial solutions
Magazines:
Clinical Leader – Innovations in Clinical Trial Management and CRO Services
Applied Clinical Trials – Advances in Specialty Clinical Research Operations
Outsourcing-Pharma – Trends in Contract Research Organizations
PharmaTimes – Updates on Clinical Trial Outsourcing and CRO Partnerships
Journals:
Journal of Clinical Research & Bioethics – Specialty CRO Operations and Clinical Trial Applications
Therapeutic Innovation & Regulatory Science – Innovations in Clinical Trial Design and Management
Drug Development and Industrial Pharmacy – Specialty CRO Case Studies and Efficiency Improvements
Contemporary Clinical Trials – Trial Management, Data Analytics, and Regulatory Compliance
Newspapers:
The Wall Street Journal – Pharmaceutical R&D and Clinical Outsourcing Markets
Financial Times – Specialty CRO Market Trends and Global Contract Research Activities
PharmaVoice – Developments in Clinical Research and CRO Services
Healthcare Innovation – Advancements in Outsourced Clinical Trials and Regulatory Services
Associations:
Association of Clinical Research Organizations (ACRO) – Guidelines and Best Practices for CRO Operations
Drug Information Association (DIA) – Clinical Trial Standards and Regulatory Compliance
Society for Clinical Research Sites (SCRS) – CRO Collaboration and Site Management Standards
European CRO Federation (EUCROF) – Specialty CRO Guidelines and Clinical Research Recommendations
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients