U.S. nasal spray market is estimated to be valued at USD 11.97 Bn in 2026 and is expected to reach USD 19.61 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.3% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The U.S. nasal spray market has witnessed steady growth owing to rising prevalence of allergic rhinitis and nasal congestion. There has been rising demand for nasal sprays over the forecast period due to growing preference for over-the-counter medications and increasing penetration of generic drugs. Self-administration of nasal sprays has made them very convenient to treat various nasal conditions without medical oversight, thus, boosting its adoption. With pharmacological advancements leading to availability of multidose nasal spray formulations with fewer side effects, specialty drugs are likely to dominate the category and fuel competition in the in the near future.
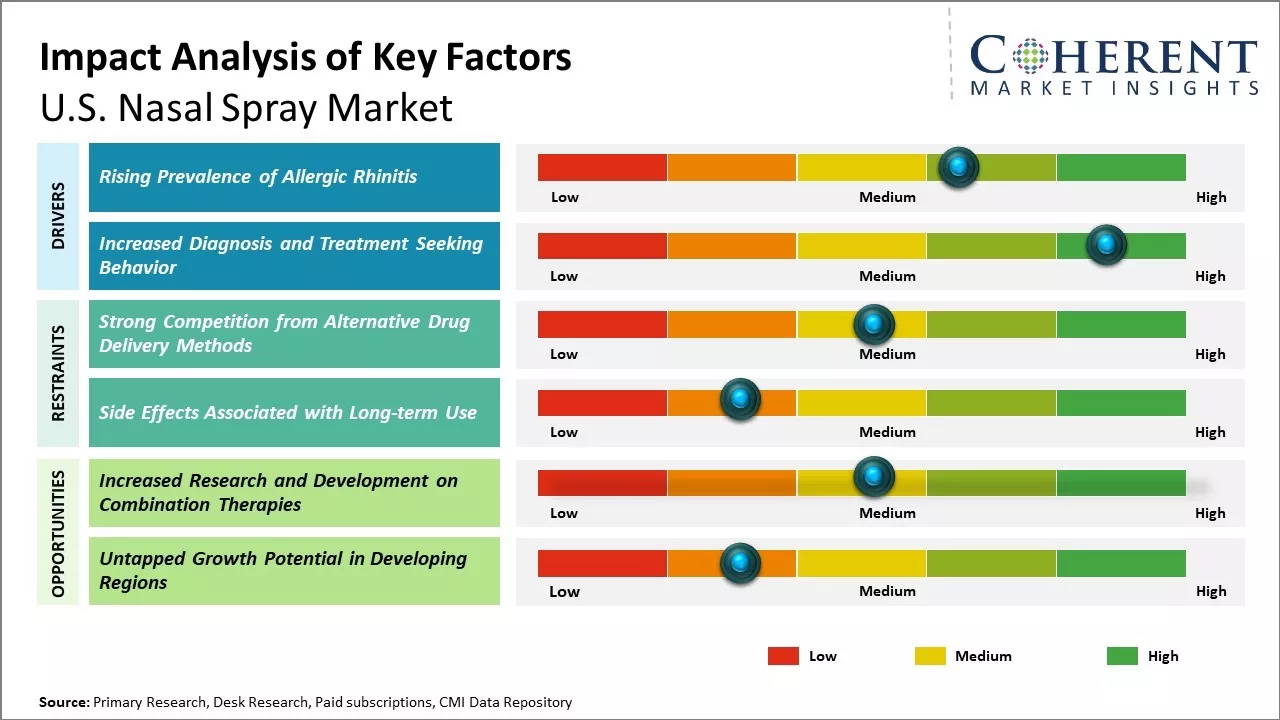
Market Driver – Rising Prevalence of Allergic Rhinitis
With changing environmental conditions and lifestyle patterns, the incidence of allergic rhinitis has increased substantially over the past few decades. Commonly known as hay fever, allergic rhinitis causes symptoms such as sneezing, nasal congestion and nasal irritation. It is one of the most prevalent chronic respiratory diseases worldwide. For instance, according to Medscape, the prevalence of allergic rhinitis in the U.S. was 7.7% in 2022. Rising prevalence of allergic rhinitis can be attributed to increased exposure to indoor and outdoor allergens such as dust mites, animal dander, and pollen grains. Widespread use of air conditioning has led to dry indoor conditions conducive for dust mite growth. The people in the U.S. are now spending more time outdoors involved in activities such as gardening, hiking, camping, and others, thus, exposing themselves to various types of pollen. Changes in lifestyle and diet have also weakened immunity, leading to increased susceptibility. With such a large patient base suffering from bothersome symptoms throughout the year, the demand for effective nasal spray medications for quick relief is growing steadily.
Get actionable strategies to beat competition: Request Free Sample
Market Driver – Increased Diagnosis and Treatment Seeking BehaviorIncreased diagnosis of allergic rhinitis and other similar conditions in the U.S. and greater awareness about nasal allergies among both patients and physicians has changed the treatment seeking behavior. People no longer consider symptoms such as sneezing or nasal congestion as a minor irritant but rather seek medical advice to get an accurate diagnosis and appropriate treatment. This is due to intense medical marketing by pharmaceutical companies and as patients now understand that leaving the condition untreated can affect their quality of life and productivity. Once diagnosed, the majority of the people prefer easy to use nasal spray medications over oral antihistamines due to the faster onset of action and topical delivery of drug.

To learn more about this report, Request Free Sample
Market Challenges – Strong Competition from Alternative Drug Delivery MethodsOral medications remain a cost-effective alternative for many conditions typically treated with nasal sprays. Furthermore, the rise of advanced inhalers provides stiff competition in respiratory applications such as allergies and asthma. Manufacturers must clearly demonstrate the clinical advantages of nasal sprays to justify their higher costs. Another challenge is the lack of insurance coverage for some new nasal spray products, which can limit market potential.
Market Opportunities – Increased Research and Development on Combination Therapies
The U.S. nasal spray market has immense scope for growth through increased focus on combination therapies. Most nasal spray products are targeted towards individual conditions like congestion, allergies or dryness. However, nasal issues often occur concurrently requiring treatment of multiple symptoms. Combination therapies present a huge opportunity to address this clinical need comprehensively. Nasal sprays containing more than one active ingredient can provide relief from multiple bothersome nasal problems with just one product. This improves patient convenience and compliance significantly. For example, a spray treating congestion as well as dryness will eliminate the need to use two different bottles. Combining medications also allows delivering lower doses of each component, reducing risks of side effects.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
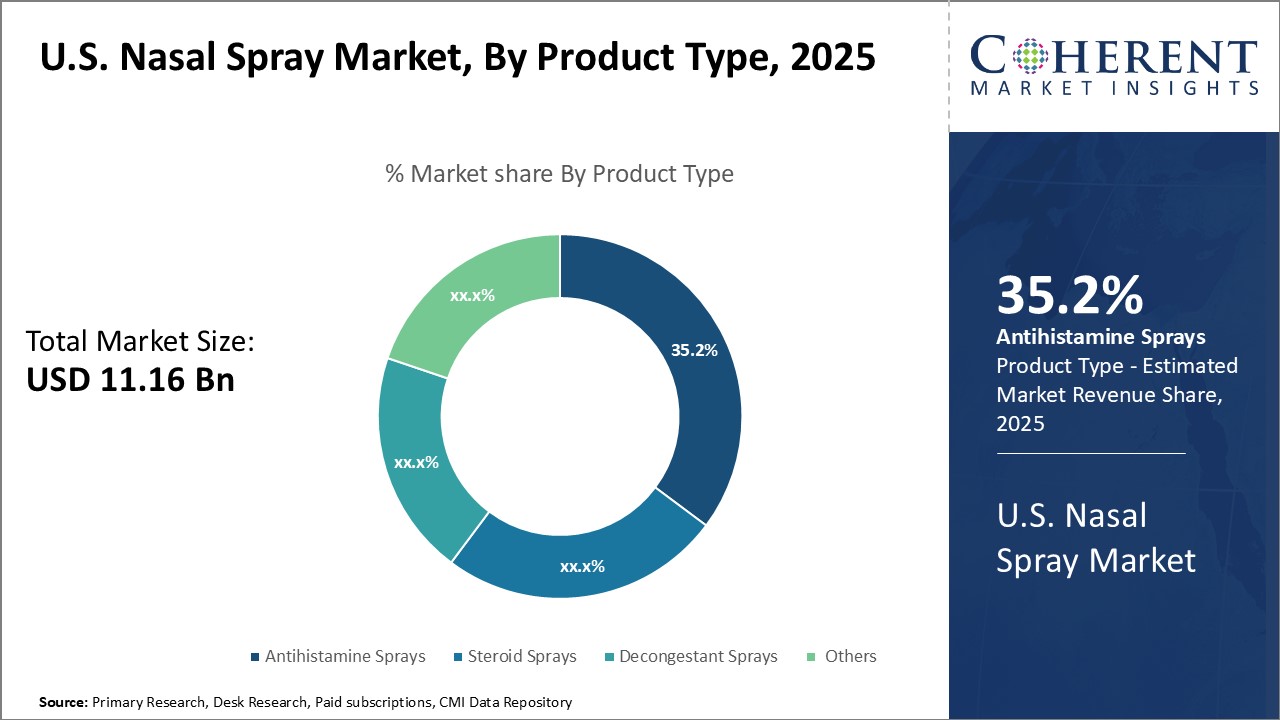
Insights, By Product Type: Convenience and efficacy properties boosts antihistamine sprays segment growthProduct Type segment is sub-segmented into steroid sprays, antihistamine sprays, decongestant sprays, others. Antihistamine sprays segment is expected to hold 36.2% of the market share in 2026, due to its convenience and efficacy benefits. As a topical treatment delivered directly to the nasal passages, antihistamine sprays provide fast acting, targeted relief from symptoms of allergies like sneezing, itching and congestion. The convenient "spray and go" portable format makes antihistamine sprays ideal for on-the-go use. Their quick absorption into nasal tissues also means antihistamine sprays start working more rapidly than oral antihistamines that must pass through the digestive system first. This faster relief offered by antihistamine sprays address the need of many allergy sufferers for relief as symptoms arise. The localized delivery of antihistamine medication directly where it is needed also minimizes the risk of potential drowsiness side effects associated with some oral antihistamines.
To learn more about this report, Request Free Sample
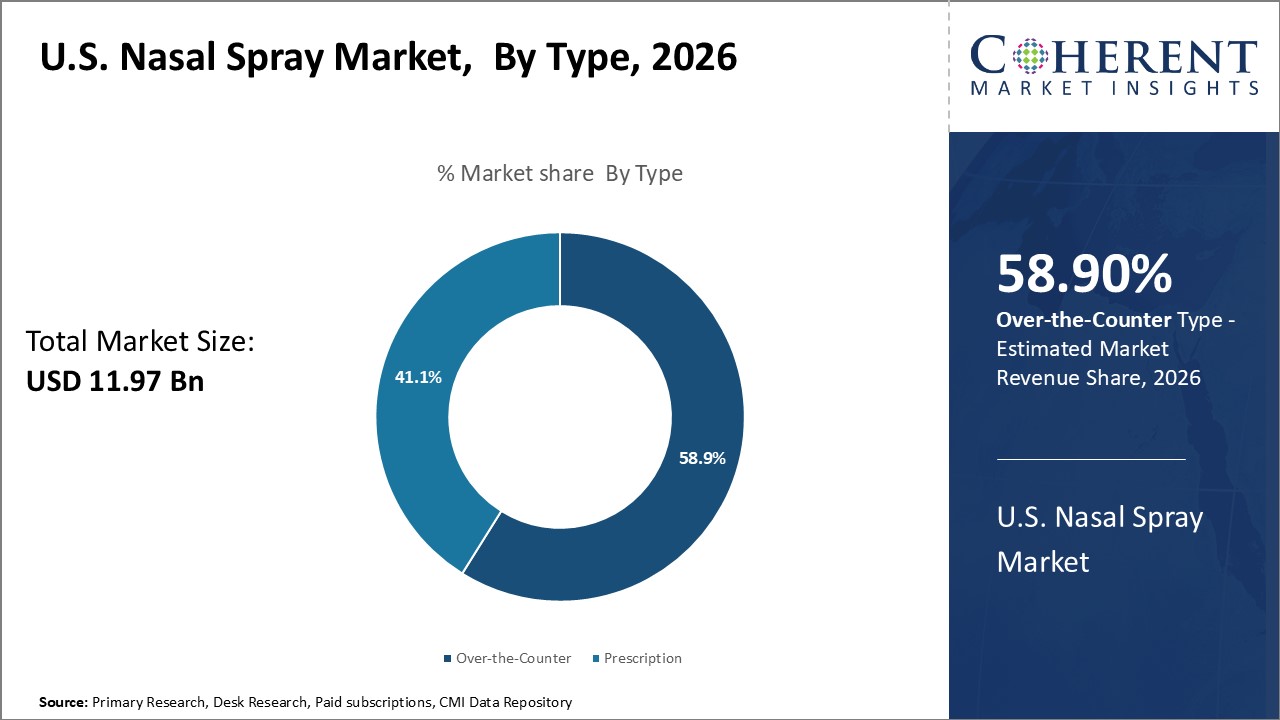
Insights, By Type: Self-care flexibility boosts over-the-counter segment growth
Type segment includes is sub-segmented into prescription and over-the-counter. Over-the-counter segment is expected to hold 58.9% of the market share in 2026, owing to its flexibility and accessibility advantages over prescription medications. Unlike prescription nasal sprays which require a doctor's visit for assessment and continued renewals, over-the-counter sprays empower individuals to easily purchase medications whenever symptoms flare up without a prescription. This self-care flexibility allows over-the-counter users full control over managing mild to moderate allergy and congestion cases, according to their personal needs and schedules. By removing barriers posed by medical appointments and prescription authorizations, over-the-counter availability broadens the potential market reach and makes relief more practical to obtain. Ease of acquisition from retail pharmacy shelves means over-the-counter sprays are not constrained by a prescriber's limited operating hours or prescription coverage limitations.
Insights, By Indication: Increasing Prevalence of Allergic Rhinitis
Indication segment is sub-segmented into allergic rhinitis, nasal allergies, nasal congestion, others. Allergic rhinitis segment is expected to hold 32.7% of the market share in 2024, owing to increasing prevalence of allergic rhinitis. Allergic rhinitis is a condition characterized by inflammation of the nasal passages due to an allergic response. The allergic response is triggered by exposure to allergens such as pollen, dust mites, animal dander, mould, and certain foods. The symptoms of allergic rhinitis include sneezing, itching, runny nose, and nasal congestion. These symptoms can significantly impact a person's quality of life, leading to limitations in daily activities, social behavior, and emotional well-being.
U.S. Nasal Spray Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 11.97 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.3% | 2033 Value Projection: | USD 19.61 Bn |
| Segments covered: |
|
||
| Companies covered: |
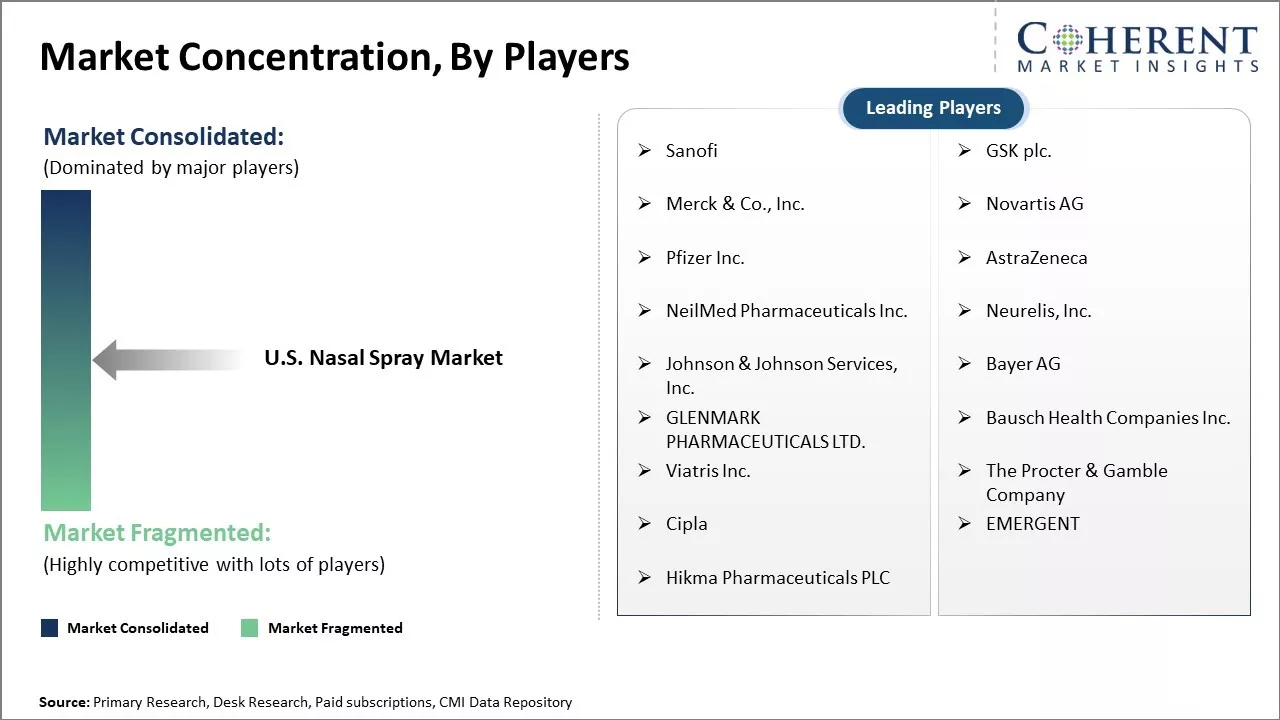
Sanofi, GSK plc., Merck & Co., Inc., Novartis AG, Pfizer Inc., AstraZeneca, NeilMed Pharmaceuticals Inc., Neurelis, Inc., Johnson & Johnson Services, Inc., Bayer AG, GLENMARK PHARMACEUTICALS LTD., Bausch Health Companies Inc., Viatris Inc., The Procter & Gamble Company, Cipla, EMERGENT, Hikma Pharmaceuticals PLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients