Swine Respiratory Disease Treatment Market Size and Forecast – 2026 – 2033
The Global Swine Respiratory Disease Treatment Market size is estimated to be valued at USD 1.42 billion in 2026 and is expected to reach USD 2.34 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 6.8% from 2026 to 2033.
Global Swine Respiratory Disease Treatment Market Overview
The swine respiratory disease treatment market is driven by the rising prevalence of respiratory infections such as porcine reproductive and respiratory syndrome (PRRS) and swine influenza, which significantly impact livestock productivity. Increasing global demand for pork has intensified large-scale farming, elevating disease transmission risks. Growing awareness among farmers about animal health and economic losses from untreated infections further supports treatment adoption. Advancements in veterinary pharmaceuticals, including vaccines and antimicrobial therapies, are also boosting market growth. Additionally, government initiatives and improved veterinary infrastructure, especially in developing regions, are encouraging better disease management and driving demand for effective treatment solutions.
Top of Form
Bottom of Form
Key Takeaways
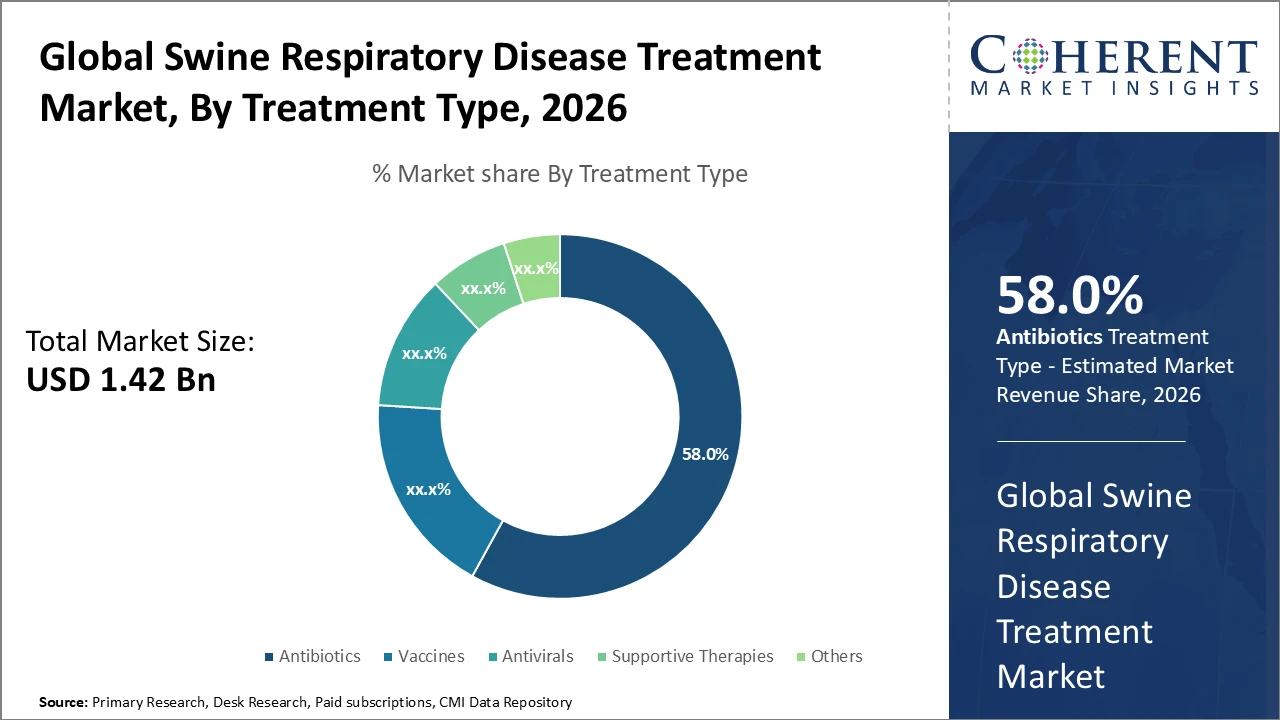
The antibiotics segment retains dominance with a market share exceeding 58%, driven by established efficacy and wide availability
Vaccines represent the fastest-growing segment due to rising awareness and preventive healthcare adoption
Swine influenza remains the predominant disease focus, contributing significantly to overall market revenue
Increasing attention is being given to managing Mycoplasma pneumonia, supporting segment expansion
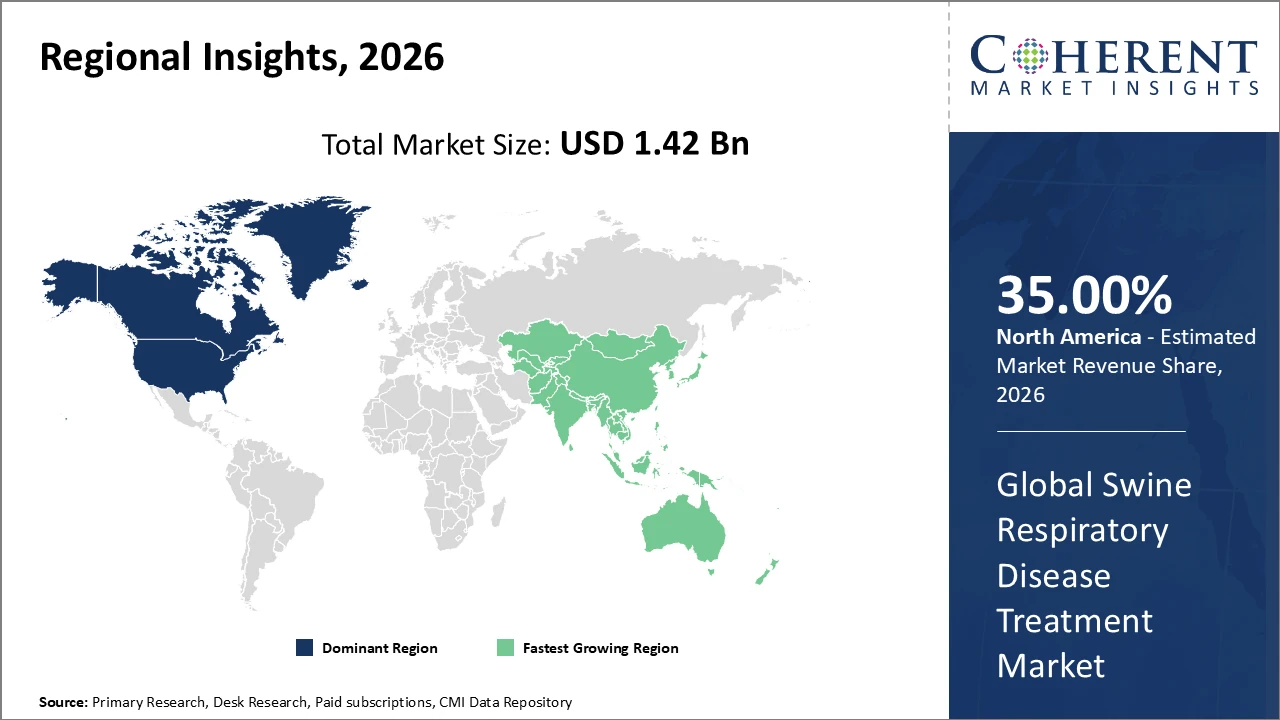
North America holds the largest market share, supported by advanced veterinary healthcare infrastructure and strong regulatory frameworks
Asia Pacific is the fastest-growing region, driven by increasing pork production, expanding farm规模, and government-led animal health initiatives in China and India
European markets are witnessing growing adoption of innovative treatment approaches, strengthening competitive dynamics
Swine Respiratory Disease Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Swine Respiratory Disease Treatment Market Insights, By Treatment Type
Antibiotics dominate the market due to their proven clinical efficacy, widespread availability, and cost-effectiveness, making them essential in managing acute respiratory diseases in swine. Their broad-spectrum action and long-standing acceptance among veterinarians further strengthen their position. Vaccines are the fastest-growing segment, driven by increased emphasis on preventive care and efforts to reduce antibiotic reliance, with advanced formulations targeting multiple pathogens. Antivirals and supportive therapies, though smaller in share, play an important role in enhancing immunity and addressing viral infections. The others segment includes alternative and emerging treatments, gradually gaining attention for specialized and niche applications.
Swine Respiratory Disease Treatment Market Insights, By Disease Type
Swine influenza dominates this segment due to its high prevalence and significant economic impact on global pig production, necessitating continuous treatment and control measures. Porcine Respiratory Disease Complex (PRDC) is the fastest-growing subsegment, driven by its multifactorial nature requiring combination therapies and integrated disease management practices, especially in large-scale farming. Mycoplasma pneumonia also holds a notable share, as it negatively affects growth performance and feed efficiency over time. Atrophic rhinitis and other respiratory conditions, though less common, remain important due to their ability to worsen overall herd health and increase vulnerability to secondary infections.
Swine Respiratory Disease Treatment Market Insights, By Application
Commercial swine farms hold the dominant market share due to their large-scale operations, which require structured health management practices, advanced respiratory treatments, and strict regulatory compliance to maintain productivity and control disease outbreaks. Smallholder farms are the fastest-growing segment, especially in emerging economies, where increasing pork demand is driving the adoption of improved veterinary care, supported by extension services and government initiatives. Research institutions, although smaller in share, play a crucial role in developing innovative therapies and refining treatment protocols. The others segment includes niche applications such as specialty swine breeds and academic or experimental use.
Swine Respiratory Disease Treatment Market Trends
The market is witnessing a strong shift toward sustainable and biologic therapies, driven by global efforts to combat antibiotic resistance
European countries have experienced around a 20% increase in vaccine adoption in 2025, supported by stringent regulatory policies
North America is leveraging digital health technologies to enhance early disease detection, reducing treatment initiation delays by nearly 30%
Asia Pacific’s expanding pork industry is driving higher demand for veterinary therapeutics across the region
China has reported approximately a 15% increase in veterinary drug usage in 2024, reflecting intensified livestock production and disease management efforts
Swine Respiratory Disease Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Swine Respiratory Disease Treatment Market Analysis and Trends
North America dominates the swine respiratory disease treatment market due to its advanced veterinary healthcare infrastructure, strict regulatory standards, and early adoption of innovative treatment solutions. The region accounts for over 35% of the global market share, supported by the presence of major industry players actively investing in research and development to expand their product portfolios. Strategic partnerships and acquisitions further strengthen market positioning. Additionally, strong government support through disease monitoring and control programs ensures effective management of swine health, contributing to consistent market growth and high adoption of advanced therapeutics across commercial farming operations.
Asia Swine Respiratory Disease Treatment Market Analysis and Trends
The Asia Pacific region is experiencing the fastest growth in the swine respiratory disease treatment market, with a CAGR exceeding 8%. This growth is primarily driven by the rapid intensification and modernization of pork production in countries like China and India. Increasing adoption of respiratory vaccines and antibiotics, along with the expansion of commercial farming operations, is significantly boosting demand for treatment solutions. Government support through subsidies and animal health awareness programs has further improved accessibility to veterinary care. These factors collectively contribute to strong market expansion and rising investments in advanced disease management practices across the region.
Swine Respiratory Disease Treatment Market Outlook for Key Countries
USA Swine Respiratory Disease Treatment Market Analysis and Trends
The USA leads the swine respiratory disease treatment market with a substantial share, supported by advanced veterinary healthcare infrastructure and strict animal health regulations enforced by agencies such as the USDA. The presence of leading companies investing in research and development has accelerated the introduction of innovative biologics and digital diagnostic solutions, improving disease detection and treatment efficiency. Market players are also expanding production capabilities; for instance, increased vaccine manufacturing capacity in 2026 enhanced supply, enabling wider implementation of herd-level immunization programs. These factors collectively drive higher adoption of advanced therapies and strengthen the country’s market position.
Germany Swine Respiratory Disease Treatment Market Analysis and Trends
Germany’s swine respiratory disease treatment market reflects broader European trends of increasing focus on preventive care, advanced diagnostics, and responsible therapeutic use. Swine influenza A and emerging viral pathogens remain significant concerns, driving demand for improved vaccines and rapid diagnostic tools supported by veterinary research institutes in the country. Regulatory emphasis on antimicrobial stewardship and animal welfare has accelerated vaccine adoption and reduced indiscriminate antibiotic use, influencing treatment strategies. Established animal health companies and local research collaborations are advancing tailored therapies and disease surveillance systems, enhancing overall respiratory disease management in Germany’s commercial pig sector.
Analyst Opinion
Precision in therapeutic formulations is a key supply-side driver, with advances in antibiotics and vaccines improving production capacity; for example, a leading pharmaceutical company reported a 15% increase in respiratory vaccine production efficiency in 2025.
Demand-side growth is driven by diversified applications across commercial and small-scale pig farms; rising veterinary drug imports in countries like Brazil, up 12% in 2024, indicate increased disease management awareness.
Micro-indicators, such as advanced diagnostic technologies, enable early pathogen detection, reducing disease progression; regulatory approvals of rapid detection kits in 2026 shortened treatment initiation by 30%, improving adoption.
Pricing dynamics influence market share, with cost optimization allowing access to price-sensitive markets; a 5–7% reduction in antibiotic prices in 2024 boosted volumes in Southeast Asia.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.42 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.8% | 2033 Value Projection: | USD 2.34 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Zoetis Inc., Virbac S.A., ZoZen Pharma, Nestle Purina, Bayer AG, Heska Corporation, Huvepharma EAD, Guandong Haid Group, Dechra Pharmaceuticals PLC, MSD Animal Health | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Swine Respiratory Disease Treatment Market Growth Factors
Rising demand for sustainable pork production is driving increased investment in swine respiratory disease treatments. Government incentives and subsidies have encouraged the adoption of vaccination programs, with Asia Pacific recording a 25% growth in vaccine uptake in 2024. Advances in veterinary diagnostics enable early disease detection, enhancing treatment effectiveness and reducing economic losses at the farm level. Heightened awareness of zoonotic risks has prompted regulatory authorities to enforce stricter health policies, further stimulating market growth. Additionally, expanding porcine populations, particularly in Latin America and Asia, offer significant opportunities for market players to capture incremental revenue by serving larger and modernizing farming operations.
Swine Respiratory Disease Treatment Market Development
In 2025, Elanco Animal Health introduced Pradalex (pradofloxacin injection), the first new antibiotic indicated for swine respiratory disease in nearly 20 years, receiving approval from the U.S. FDA. This provided pork producers with an effective option for controlling bacterial infections in swine herds.
Key Players
Leading Companies of the Market
Zoetis Inc.
Bayer AG
Heska Corporation
ZoZen Pharma
Nestle Purina
Huvepharma EAD
MSD Animal Health
Virbac S.A.
Dechra Pharmaceuticals PLC
Guandong Haid Group
Several market players in the swine respiratory disease treatment sector have implemented competitive strategies to strengthen their market position, including expanding R&D efforts to tackle drug resistance and forming global alliances to improve distribution networks. For instance, in 2025, Boehringer Ingelheim strategically acquired a biotech firm specializing in swine immunotherapies, resulting in a 20% increase in its R&D capabilities. This acquisition enabled the company to accelerate the development of innovative treatments, enhancing its presence and market share across North America and Europe while addressing the growing demand for effective respiratory disease management solutions in commercial pig farming.
Swine Respiratory Disease Treatment Market Future Outlook
The future outlook for the swine respiratory disease treatment market is positive, driven by increasing global demand for pork and the need for sustainable, efficient herd management. Advances in vaccines, biologics, and rapid diagnostic technologies are expected to enhance disease prevention and treatment efficacy. Regulatory focus on reducing antibiotic use and promoting animal welfare will further boost vaccine and alternative therapy adoption. Emerging markets in Asia Pacific and Latin America offer significant growth opportunities due to expanding commercial pig farming and supportive government initiatives. Overall, innovation, preventive care, and strategic investments will shape a resilient and steadily growing market in the coming years.
Swine Respiratory Disease Treatment Market Historical Analysis
The swine respiratory disease treatment market has historically been driven by the prevalence of infectious respiratory diseases such as swine influenza, PRRS, and Mycoplasma pneumonia. Antibiotics dominated early market growth due to their effectiveness and widespread availability, while vaccines gradually gained traction as preventive strategies became prioritized. North America and Europe led the market, supported by advanced veterinary infrastructure and regulatory frameworks. Over time, emerging economies in Asia and Latin America began increasing adoption of veterinary therapeutics in response to rising pork production and herd health concerns. Historical trends reflect a gradual shift from reactive treatments to preventive and integrated disease management approaches.
Sources
Primary Research Interviews:
Veterinary researchers, animal health specialists, and pharmaceutical consultants across swine farms and veterinary service companies
Operations managers, procurement heads, and distribution professionals in animal health and veterinary pharmaceutical companies
Livestock industry analysts and farm management consultants focusing on swine health, respiratory disease prevention, and treatment adoption
Magazines:
Pig Progress – Swine Health Management, Treatment Innovations, and Market Trends
World Pork – Veterinary Practices, Disease Prevention, and Farm Updates
Pig International – Respiratory Disease Management, Vaccine Adoption, and Technology in Swine Health
The Pig Site – Farm-Level Health Strategies, Drug Use, and Regulatory Updates
Journals:
Journal of Swine Health and Production – Research on Respiratory Diseases, Vaccines, and Herd Management
Preventive Veterinary Medicine – Disease Control, Therapy Optimization, and Farm Health Programs
Veterinary Record – Adoption Patterns, Treatment Efficacy, and Animal Welfare Guidelines
Newspapers:
Feed Navigator – Animal Health Developments, Veterinary Drug Launches, and Market Insights
Pig Progress News – Swine Industry Updates, Treatment Trends, and Regulatory Policies
World Animal Protection News – Livestock Health, Welfare Practices, and Market Changes
Farm Journal – Swine Production Trends, Pharmaceutical Developments, and Industry Reports
Associations:
American Association of Swine Veterinarians (AASV) – Research, Guidelines, and Training in Swine Respiratory Health
World Organisation for Animal Health (WOAH / OIE) – Standards, Disease Reporting, and Global Health Policies
National Pork Board – Animal Health Programs, Disease Prevention, and Farm Best Practices
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients