Urolithiasis Management Devices Market is estimated to be valued at USD 1,954.8 Mn in 2026 and is expected to reach USD 2,750.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.0% from 2026 to 2033. There are multiple types of kidney stones, however, 80% of stones are composed of calcium oxalate or phosphate, other stone types include uric acid (9%), struvite (10%), and cystine (1%) stones and are significantly less common than stones composed of calcium oxalate or phosphate (80%). The different types of stones occur due to varying risk factors such as diet, prior personal and family history of stones, environmental factors, medications. Moreover, an increase in government and non-government organizations’ initiatives to improve access to affordable urinary care and an increase in awareness among people regarding urinary-related diseases are expected to drive market growth.
Analysts’ Views on Global Urolithiasis Management Devices Market:
The global urolithiasis management devices market growth can be driven by the increasing prevalence of urolithiasis. For instance, according to the data provided by ScienceDirect, a website which provides access to bibliographic database of scientific and medical publications, in January 2022, urolithiasis is among the most common urologic diagnosed globally, the estimated prevalence of urolithiasis ranges from 1% to 13% in different regions, globally.
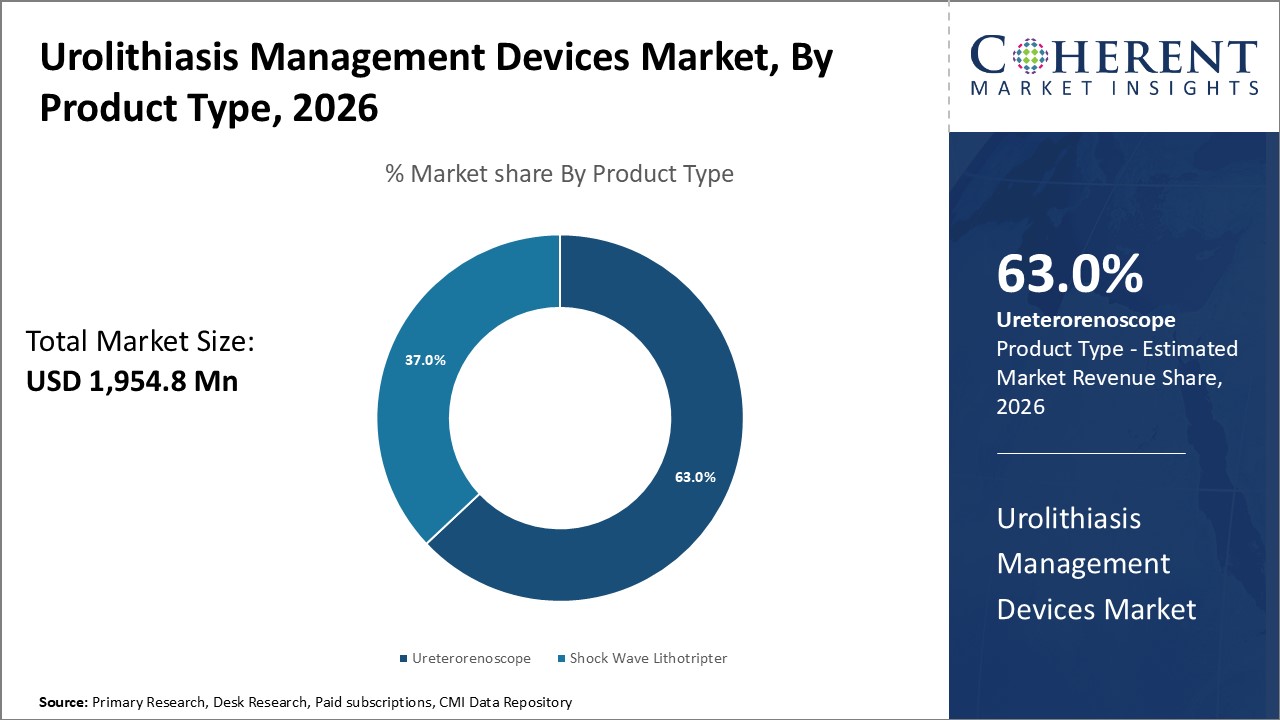
Figure 1. Global Urolithiasis Management Devices Market Share (%), by Product Type, 2026
To learn more about this report, Download Free Sample
Global Urolithiasis Management Devices Market- Driver
Rise in awareness of the importance of urinary health
Globally, kidney stone disease prevalence and recurrence rates are increasing owing to the increased prevalence of urolithiasis in both developed and developing countries over the last few decades. This growing trend is believed to be associated with lifestyle changes, such as a lack of physical activity. For instance, in May 2022, Urology Care Foundation, a foundation that supports individuals and projects that provide direct urologic patient care, announced a medical student innovation fellowship in collaboration with Boston Scientific Corporation, a biomedical devices manufacturing company, which will support the education of medical students from racial and ethnic backgrounds underrepresented in urologic research.
Increase in inorganic strategies collaboration and partnership by key market players
Key market players are adopting growth strategies such as partnerships and collaborations to launch new products. For instance, in September 2021, Advanced Medtech, a pharmaceutical and medical devices manufacturer, acquired Shenzhen Huikang Medical Apparatus Co, Ltd., a medical devices manufacturing company. This acquisition is done to strengthen the market position of Advanced Medtech, and develop various solutions to treat urological and orthopedic conditions such as kidney stones, and others, which is expected to drive the market growth over the forecast period.
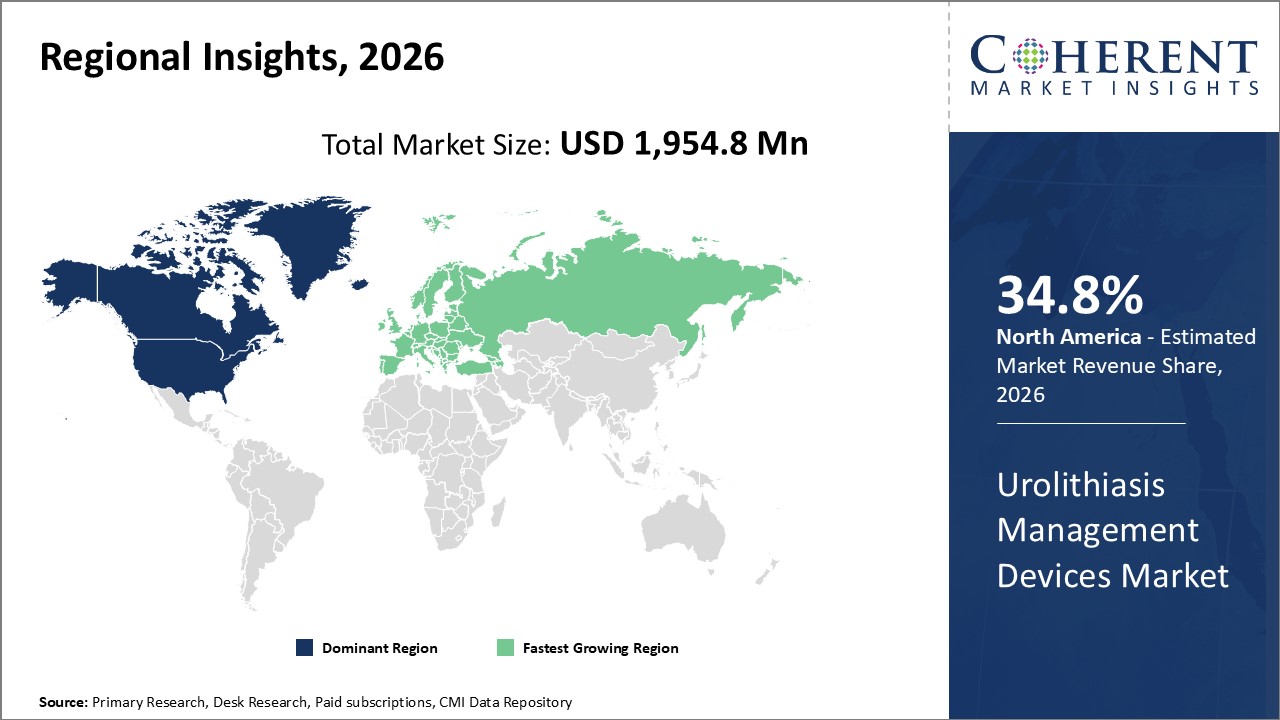
Figure 2. Global Urolithiasis Management Devices Market Share (%), by Region
To learn more about this report, Download Free Sample
Global Urolithiasis Management Devices Market - Regional Analysis
Among all regions, North America is expected to dominate the global market over the forecast period. This is attributed to North America holding a 34.8% market share and an increase in the acquisition by the key market players in this region.
The Europe region is expected to be the second dominating region over the forecast period, due to an increase in product launches in Europe. For instance, the launch of new satellite offices for urology care is likely to fuel the growth of the Global urolithiasis management devices market. For instance, in July 2020, Advanced Urology, a urology consultant which provides lab work, testing, and surgery, started new satellite offices in Buckhead, Canton, Braselton, and cumming in Georgia to offer urology care.
For instance, in November 2020, data was published by the National Center forBiotechnologyAccording to the survey, 49% of physicians reported that their regular therapeutic practice had changed by more than 90%. Throughout the crisis, 72.3% of physicians utilized telemedicine.
Urolithiasis Management Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,954.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.0% | 2033 Value Projection: | USD 2,750.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Lumenis Ltd., Boston Scientific Corp, BD, Cook Medical, Karl Storz, Allengers Medical Systems, Inc, Olympus Corporation, Electro Medical Systems SA, Dornier MedTech GmbH, Direx Group., HealthTronics, Inc., CONMED Corporation., Applaud Medical, Inc., Potent Medical., and other prominent players. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Urolithiasis Management Devices Market Segmentation:
The global urolithiasis management devices market report is segmented into Product type and End users.
Based on Product Type, the market is segmented into Shockwave lithotripter and ureterorenoscope. Shockwave lithotripter is sub-segmented into intracorporeal lithotripter and extracorporeal lithotripter, and ureterorenoscope sub-segmented into rigid ureterorenoscope, semi rigid ureterorenoscope and flexible ureterorenoscope. Out of which the ureterorenoscope is expected to dominate the global urolithiasis management devices market during the forecast period.
Based on End Users, the market is segmented into hospitals, clinics, and ambulatory surgical centres, out which the ambulatory surgical centres are dominated
Based on Region , North America, Latin America, Europe, Asia Pacific, Middle East and Africa, among all regions the North America region is dominated.
Global Urolithiasis Management Devices Market Cross Sectional Analysis:
In product type segment, Intracorporeal Lithotripter segment held a dominant position in the Asia Pacific region due to the increasing number of product launches by key market players. For instance, according to the National Center for Biotechnology Information (NCBI), the cost of medical management of kidney stones is expected to reach US$ 2,357.7 Mn in the Asia Pacific by 2030. Cost containment for the patients has been made possible by technological development in terms of shorter duration hospital stays and more minimally invasive surgical procedures.
Global Urolithiasis Management Devices Market: Key Developments
Investment in research and development of non-invasive solutions for kidney stones is expected to propel the growth of the global urolithiasis management devices market over the forecast period. For instance, in January 2022, Applaud Medical, Inc. (Applaud), an emerging leader in the treatment of kidney stones, announced that its Acoustic Enhancer technology an acoustic enhancer is a microbubble consisting of a gas core with a lipid shell technology thatreceived the breakthrough devices designation from the U.S. Food and Drug Administration (FDA).
Global Urolithiasis Management Devices Market: Key Trend
Increasing inorganic strategies like investments which is expected to drive the market over the forecast period. For instance, in January 2021, Advanced MedTech, one of the global medical device manufacturers, announced that it had funded US$ 3 Mn in Devicare, a medical device manufacturing company, to support its development of innovative urological treatments.
Global Urolithiasis Management Devices Market: Restraint
High risk of damage to the urinary tract is expected to hinder the growth of the global urolithiasis management devices market over the forecast period. For instance, in March 2021, Cleveland Clinic, a non-profit academic medical center, according to which shock wave lithotripter can cause risks which include blockage in the ureter, infection, painful urination, blood in the urine or bleeding around the kidney, and others.
Definition: A small and hard deposit that forms in the kidneys and is often painful when passed. Kidney stones are hard deposits of minerals and acid salts that stick together in concentrated urine.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients