Global Cell Therapy Manufacturing Platform Market Size and Forecast – 2026-2033
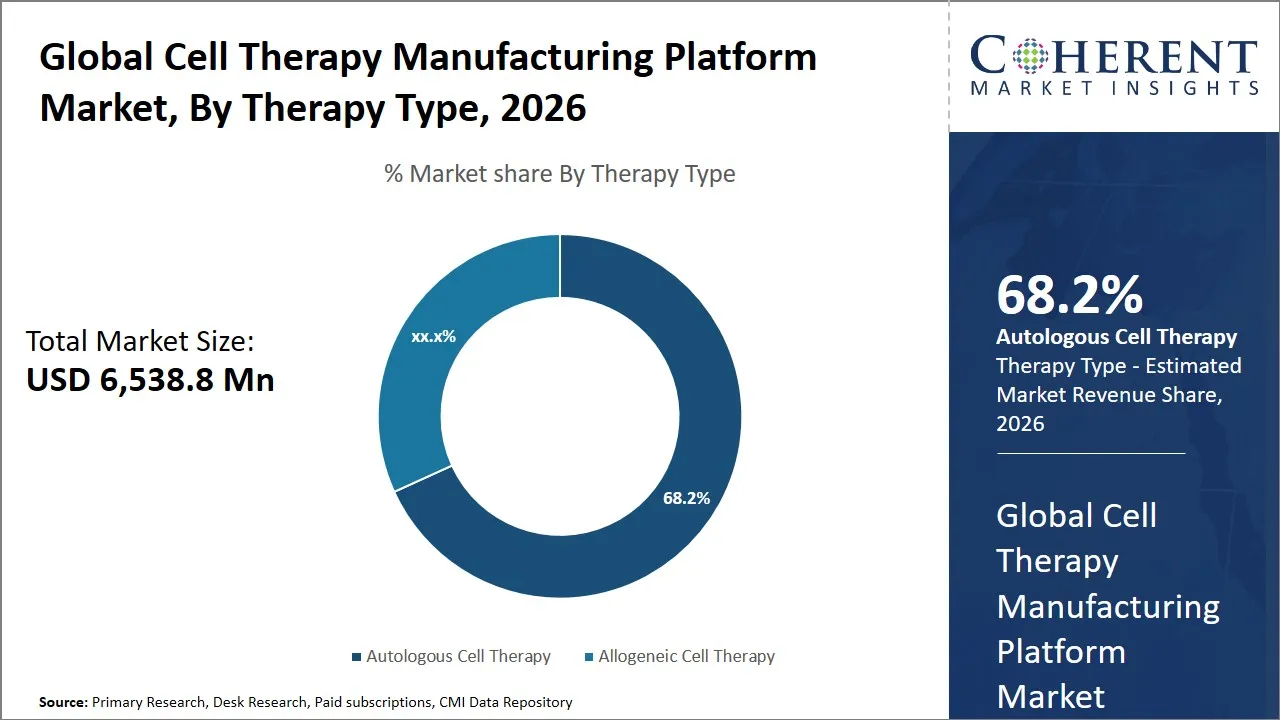
The global cell therapy manufacturing platform market is estimated to be valued at USD 6,538.8 Mn in 2026 and is expected to reach USD 13,667.5 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 15.2% from 2026 to 2033. This significant growth reflects increasing investments in cell therapy research, rising adoption of advanced manufacturing technologies, and the growing prevalence of chronic diseases that can be addressed by cell-based therapies.
Key Takeaways of the Global Cell Therapy Manufacturing Platform Market
- Autologous cell therapy segment is expected to lead the global cell therapy manufacturing platform market, capturing 68.2% share in 2026.
- Somatic cell technology segment is estimated to represent 61.4% of the global cell therapy manufacturing platform market share in 2026.
- IPSC’s (Induced pluripotent stem cell) segment is projected to dominate with 29.7% of the global cell therapy manufacturing platform market share in 2026.
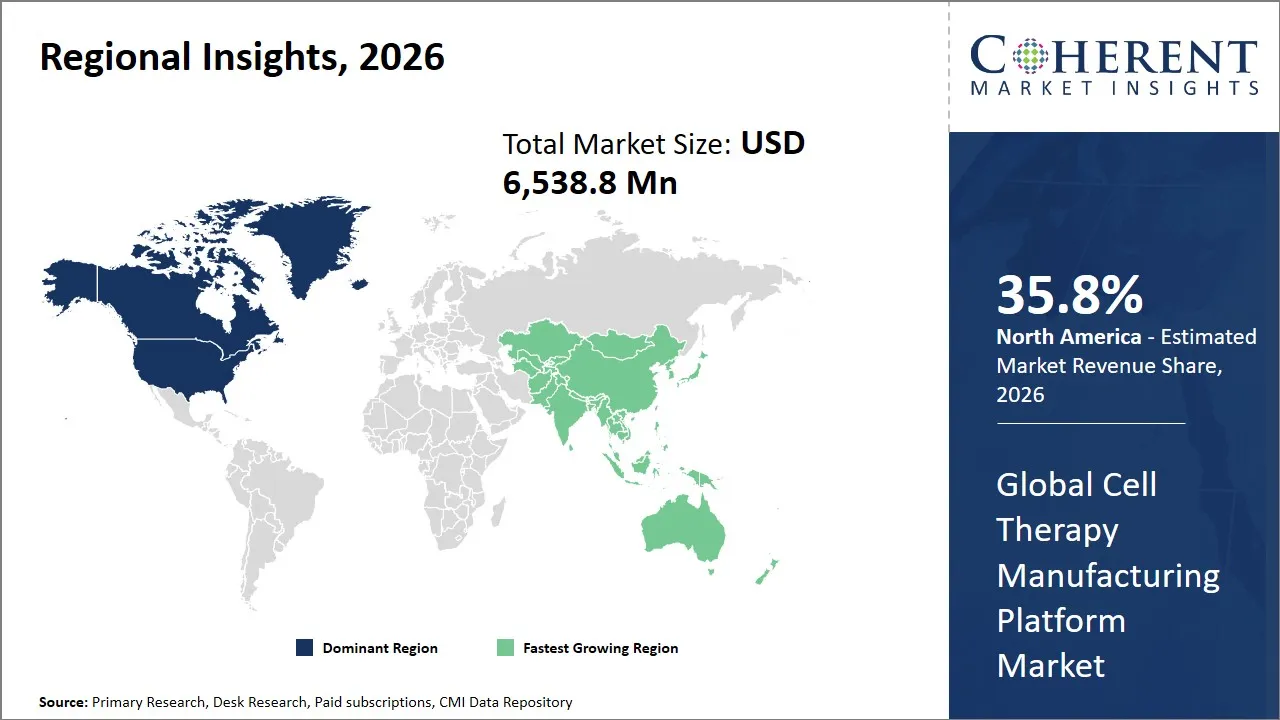
- North America is expected to lead the market, holding a share of 35.8% in 2026. Asia Pacific is anticipated to be the fastest-growing region, with 28.3% share in 2026.
Market Overview
- The market is shifting from manual, labor-intensive production to automated, closed, and digitally managed manufacturing platforms to improve consistency, throughput, and commercial readiness.
- Regulatory support is becoming a strong growth catalyst, with the U.S. FDA’s Advanced Manufacturing Technologies Designation Program now explicitly covering automated manufacturing methods for cell-based therapies.
- The biggest market demand is coming from the need to lower batch failure risk, reduce cost per dose, standardize workflows, and support scale-up for both autologous and allogeneic therapies.
- Platform innovation is accelerating, with companies such as Ori Biotech, Cellares, and Sartorius launching or advancing integrated systems designed for scalable GMP cell therapy production.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA support for advanced manufacturing platforms in cell therapy |
|
|
Large-scale investment in automated manufacturing capacity |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Autologous Cell Therapy Segment Dominate the Global Cell Therapy Manufacturing Platform Market in 2026?
The autologous cell therapy segment is expected to hold the largest market share of 68.2% in 2026, due to its foundation in personalized medicine and immunocompatibility benefits. In autologous therapy cells are taken out of a patient who will be receiving a treatment and this would go a long way in ensuring fewer chances of immune rejection. This natural compatibility reduces the use of immunosuppressive drugs which normally have negative side effects hence making autologous methods safer and more effective to the patient.
The increased cases of the chronic and degenerative diseases have led to a high demand of personalized treatment modalities that are able to focus on the profile of the patients. The personalized solution of autologous cell therapies suits this need perfectly with the isolation, genetic manipulation, cultures, and replacement of diseased tissues in patients. Such individualized manipulation improves the effectiveness of therapy and creates patient-centered treatment paradigms that have been spread among both healthcare providers and patients.
For instance, on January 6, 2026, Autolus Therapeutics announced it would evaluate Cellares’ automated Cell Shuttle platform for manufacturing AUCATZYL (obe-cel), an autologous CAR-T therapy, to support expansion into new indications. This reflects growing demand for automated, scalable manufacturing platforms in autologous cell therapy, where consistency, turnaround time, and cost control are critical.
(Source- Cellares)
Why Does the Somatic Cell Technology Segment Dominate the Global Cell Therapy Manufacturing Platform Market in 2026?
The somatic cell technology segment is expected to hold 61.4% of the market share in 2026, owing to a mature scientific foundation and broad applicability across therapy types. Instead of addressing ethical and technical issues of using embryonic stem cells, somatic cell technology is the manipulation of differentiated cells with therapeutic purposes. This strategy complies with the regulatory preferences of various countries that will allow to develop and commercialize faster.
The flexibility of somatic cell technologies is that they can be used in a broad spectrum of therapeutic applications such as tissue repair, immune regulation and as treatment of genetic diseases. In contrast to the emerging technologies which remain mostly experimental, somatic cell platforms have already become clinical in various indications, and have demonstrated a positive history of efficacy and safety. This is a real-life validation that builds trust amongst the stakeholders and expedites the assimilation into the current health systems.
For instance, in October 2025, Healios and Minaris Advanced Therapies entered a commercial manufacturing partnership to support late-phase cell therapies for acute respiratory distress syndrome and other indications. This is relevant to somatic cell technology because it highlights how therapy developers are relying on specialized manufacturing partners to scale complex cell-based products from clinical development toward commercialization.
(Source- Minaris)
The IPSC’s (Induced pluripotent stem cells) Segment Dominates the Global Cell Therapy Manufacturing Platform Market
The IPSC’s (Induced pluripotent stem cells) segment is expected to hold the largest market share of 29.7% in 2026, due to their extraordinary regenerative potential combined with ethical acceptability. The IPSCs are obtained by converting adult somatic cells back to a state of pluripotency to allow them to develop into virtually any type of cell. This adaptability heralds uncountable prospects of repairing the destroyed tissues and treating a wide range of conditions including neurodegenerative and heart diseases.
One of the greatest benefits of IPSCs compared to embryonic stem cells is that, unlike embryos, it does not require any destruction of embryos, and this has made it easier to be accepted by the regulatory agencies, the researchers and the general population. This level of ethics will fasten research partnerships, funding, and policy backing that, when combined, would improve the penetration of IPSC-based manufacturing frames.
Rising GMP Inspections and Regulatory Approvals Strengthening Cell Therapy Manufacturing Infrastructure
- The count of GMP checks and compliance approvals on the manufacturing of cell therapy is on the increase as regulators have restricted control over the production of advanced therapy and broadened the system of decentralized manufacturing. U.S. FDA reports it oversees compliance of manufacturers with the existing Good Manufacturing Practice (GMP) by conducting inspections, and the MHRA/EudraGMDP GMP register of cell therapies displays over 1,100 GMP entries across nations and lists specific cell therapy facilities as at the University of Birmingham Cell Therapy Facility, which represents a wider range of inspection around the infrastructure of advanced therapies.
- On the approvals side, the regulatory pipeline is also moving in the same direction, supporting the expansion of qualified cell therapy manufacturing infrastructure and facilities. In the European Union, the European Medicines Agency (EMA) requires all Advanced Therapy Medicinal Products (ATMPs) to undergo centralized authorization. In its quarterly report, the Committee for Advanced Therapies (CAT) indicated 49 ATMP marketing authorization applications and 32 positive draft opinions cumulatively as of November 2025, representing 31 Advanced Therapy Medicinal Products.
Regional Insights
To learn more about this report, Request Free Sample
North America Cell Therapy Manufacturing Platform Market Analysis and Trends
The North America region is projected to lead the market with a 35.8% share in 2026, driven by a well-established and highly sophisticated market ecosystem that integrates cutting-edge biotechnology research with advanced manufacturing capabilities. There is also good government patronage of the region through funding programs such as the National Institutes of Health (NIH) and good regulatory policies that have been put in place by the FDA that promote innovation but also safeguard product safety and effectiveness. North America boasts of a rich concentration of research institutes, leading contract manufacturing organizations (CMOs), and groundbreaking biotech firms that serve as a strong industry node that drives the market leadership.
Specific players like Lonza, Catalent, and Miltenyi Biotec are of major importance as they offer end to end development and manufacturing system, which enables quicker progress and commercial scale production of clinical trials. The widespread presence of healthcare facilities in the region and high rate of acceptance toward new treatment methods also make it dominant in the market.
For instance, in April 2025, U.S.-based Cellares received FDA Advanced Manufacturing Technology designation for its Cell Shuttle platform, an automated end-to-end system for cell therapy production.
(Source- Cellares)
Asia Pacific Cell Therapy Manufacturing Platform Market Analysis and Trends
The Asia Pacific region is expected to exhibit the fastest growth in the cell therapy manufacturing platform market contributing 28.3% share in 2026, due to expanding healthcare access, increasing government investments in biotech infrastructure, and rapidly growing patient populations that demand innovative treatment options. The supportive regulatory policies and national strategies that promote the development of cell therapies and the localization of manufacturing capacities have been developed by countries like China, Japan and South Korea. The area is very competitive in terms of manufacturing costs and growing partnerships among local biotechs and other international companies. Significant firms such as WuXi AppTec, FUJIFILM Cellular Dynamics and Samsung Biologics are leading the trends in the field of cell therapy manufacturing, using their technological potential and expertise in their respective areas to effectively scale up operations. Also, the positive trade conditions and regional alliances support the ease of supply chains and the expansion of the market.
For instance, on March 14, 2026, Japan approved two induced pluripotent stem cell derived therapies manufactured at SMaRT, described as the world’s first commercial facility for iPS cell-based treatments.
(Source- Wired)
Cell Therapy Manufacturing Platform Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Cell Therapy Manufacturing Platform Market?
The cell therapy manufacturing platform market in the U.S. is a leader in the cell therapy manufacturing as a result of its strong biotech ecosystem and extensive regulatory support. Key suppliers such as Lonza and Catalent have large manufacturing divisions that take care of cell and gene therapies, allowing them to easily shift to commercial levels. The US also enjoys good venture capital climate where innovations and startups which specialize in new platform technologies are being developed. The fact that the market has leading academic medical centers and a high level of clinical trials activity enhances the evolution of the market.
How is China Helping in the Growth of the Cell Therapy Manufacturing Platform Market?
The cell therapy manufacturing platform market in China is growing at a rapid rate, and the promotion of these products is backed by the active governmental policy, such as the scheme of Made in China 2025, which focuses on the development of biopharmaceuticals. Large companies like WuXi AppTec and Cellular Biomedicine Group are quickly expanding manufacturing systems to be used with cell therapies. The growing regulatory convergence of the country with the international standards and the high number of patients in the country accelerate the process of R&D and manufacturing. The increased partnership with foreign biotech firms by China only increases its market.
Key Drivers for the Growth of the Germany Cell Therapy Manufacturing Platform Market
Germany has remained the leader in the Europe cell therapy manufacturing platform market, with a high industrial base and favorable regulatory frameworks within the European Medicines Agency (EMA). Principles of CMOs like Miltenyi Biotec and Thermo Fisher Scientific have their presence in the country as their local subsidiaries are very crucial in promoting manufacturing technologies. The intense quality and precision manufacturing of Germany is appealing to partnerships and funding, which promotes cell therapy production innovation.
Japan Cell Therapy Manufacturing Platform Market Trends
The market of Japan is characterized by its active regulatory policies facilitated by the Pharmaceuticals and Medical Devices Agency (PMDA) to speed up the process of approval of regenerative medicine. The government is also promoting the idea of industry-academic cooperation enhancing innovation in the manufacturing platform of cell therapies. Others of relevance such as FUJIFILM Cellular Dynamics and Takeda Pharmaceutical are more concerned with the development of advanced manufacturing processes and quality assurance methods. The aging population of Japan is also a contributing factor that increases the need to find good cell-based therapies.
Cold Chain and Cryopreservation Complexity Creating Major Operational Bottlenecks
- This is one of the significant operational challenges in the global market of cell therapy manufacturing platform as these treatments are very sensitive, customized, and in many cases, nonrenewable. Cryopreserved cell products normally require storing in conditions close to liquid nitrogen temperature, and the preservation requires a well-orchestrated formulation, fill-finish, freezing, storage, transportation, as well as thawing procedures. Even small execution errors may compromise the stability of the final product, and temperature swings, delays and handling variation during transit can compromise a whole therapy.
- The complexity is further compounded by the fact that, clinical and manufacturing plants will require special freezers, liquid nitrogen storage facilities, reserve capacity, oxygen security system, trained personnel and tested handling procedures. Cytiva observes that space and cost constraints are an issue in many sites and some have already exceeded the limits of storage capacity whilst standardization gaps in the cryopreservation and fill- finish industry is an established problem. Practically, it renders logistics not only a transportation capability but an essential aspect of platform design, scalability, and commercial preparedness to cell therapy manufacturing.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On February 3, 2026, Cellares announced a collaboration with Stanford Medicine to expand its automated Cell Shuttle manufacturing platform for gene-edited hematopoietic stem cell therapies. The partnership aims to develop standardized manufacturing processes and analytical workflows that can support multiple rare disease programs and improve scalability in advanced cell therapy manufacturing.
- In September 2025, Ori Biotech received U.S. Food and Drug Administration Advanced Manufacturing Technology (AMT) designation for its IRO cell and gene therapy manufacturing platform, a closed automated system designed to standardize and scale cell therapy production. The platform integrates automation, analytics, and digital workflows to improve throughput, reduce batch failures, and enable scalable commercial manufacturing of cell therapies.
- In March 2025, AstraZeneca signed an agreement to acquire EsoBiotec, a biotechnology company developing in-vivo cell therapy platforms using its Engineered NanoBody Lentiviral (ENaBL) technology. The acquisition is intended to accelerate the development and scalable manufacturing of innovative cell therapies for oncology and immune diseases.
Top Strategies Followed by Global Cell Therapy Manufacturing Platform Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players lead the market through strong research and development, automated platform innovation, and strategic partnerships that improve scalability, compliance, and manufacturing efficiency. They also expand into high-growth regions to capture rising demand from biotech and cell therapy developers. |
In January 2026, Cellares raised major funding and expanded its automated Cell Shuttle manufacturing network through a global agreement with Bristol Myers Squibb, strengthening its position in large-scale cell therapy manufacturing. |
|
Mid-Level Players |
Mid-level companies compete by offering cost-effective manufacturing platforms that cover core performance needs without the high price of large integrated systems. Their advantage is flexibility. They can adapt faster, work closely with technology partners and contract manufacturers, and serve smaller biotech clients that need scalable but affordable manufacturing support. |
Ori Biotech is a strong example of this approach. In June 2025, it partnered with MaxCyte to improve manufacturing efficiency and broaden adoption of autologous cell therapies, showing how mid-level players use collaboration to strengthen technology capability and market reach without building everything alone. |
|
Small-Scale Players |
Small-scale players stay competitive by focusing on niche innovations such as automation modules, single-use systems, and specialized platform components that solve specific manufacturing gaps rather than offering full-scale broad platforms. Their strength is speed, specialization, and close collaboration with startups, academic labs, and regional partners to bring targeted solutions to market faster. |
In January 2026, Cytiva partnered with U.K.-based Cellular Origins to combine Cytiva’s manufacturing technologies with Cellular Origins’ robotic platform for cell and gene therapy production. It is a good example of how smaller specialist innovators use partnerships with larger industry players to scale visibility and commercial reach. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Cell Therapy Manufacturing Platform Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6,538.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 15.2% | 2033 Value Projection: | USD 13,667.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, Lonza, Miltenyi Biotec, Sartorius, Cytiva, Terumo Blood and Cell Technologies, Fresenius Kabi, Cellares, Ori Biotech, ScaleReady, Wilson Wolf Manufacturing, Bio-Techne, Minaris Advanced Therapies, ElevateBio, and FUJIFILM Biosciences |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Cell Therapy Manufacturing Platform Market Dynamics
To learn more about this report, Request Free Sample
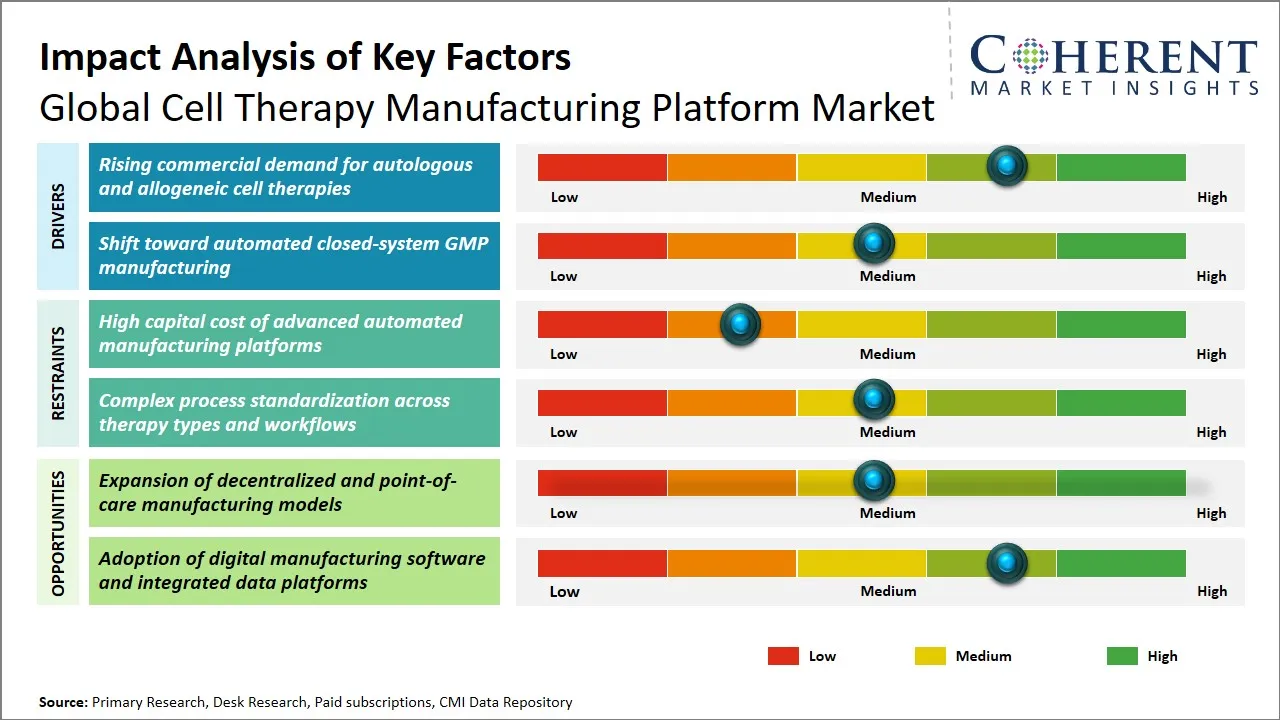
Global Cell Therapy Manufacturing Platform Market Driver - Rising Commercial Demand for Autologous and Allogeneic Cell Therapies
The increasing commercial pressures of autologous and allogeneic cell therapies are a major force that is driving the growth of the global cell therapy manufacturing platforms market. Autologous therapies, the ones that utilize patient own cells have become popular because of their personalized nature and also less chance of rejection by the immune system, which makes them very attractive in the treatment of different issues which include cancers and auto immune diseases. Simultaneously, allogeneic therapies are the off-the-shelf and scalable options, based on donor cells, which solve limitations of manufacturing time and cost of autologous treatments.
For instance, in April 2024, Cellares expanded its agreement with Bristol Myers Squibb to support a second CAR-T program on its automated Cell Shuttle platform. This reflects rising commercial demand for both autologous and broader next-generation cell therapies, as developers increasingly turn to scalable manufacturing platforms to handle higher volumes, improve consistency, and prepare for commercialization.
(Source- Cellares)
Global Cell Therapy Manufacturing Platform Market Opportunity - Expansion of Decentralized and Point-of-Care Manufacturing Models
Decentralized and point-of-care (POC) manufacturing models are one of the greatest opportunities in the global cell therapy manufacturing platform market. There are usually some issues with traditional centralized manufacturing methods in terms of scaling, logistical complexity, and long lead times, and this can slow down delivery of important cell-based treatments to patients. Decentralized manufacturing allows the localized production near the treatment site and thus, decreases transportation risks, contamination potentialities, and time-to-treatment. This model is especially suitable in the context of personalized cell therapies, like the model of autologous one, where the specific cells of patients must be delivered in a short time. The growing implementation of automated, modular and scalable manufacturing systems allows implementing POC manufacturing units in hospitals, clinics, and specialized treatment facilities. It is also supported by technological innovations of closed-system bioreactors, combined quality management and real-time measurement, which provides a stable quality of manufacturing and regulatory standards in the decentralized establishment.
For instance, in August 2024, the Champalimaud Foundation in Portugal adopted Lonza’s Cocoon Platform to support decentralized cell therapy manufacturing closer to the treatment site. This is relevant because it shows how point-of-care and near-patient production models are being used to shorten turnaround time, reduce logistics complexity, and improve access to advanced therapies outside a single centralized manufacturing hub.
(Source- Lonza)
Analyst Opinion (Expert Opinion)
- The global cell therapy manufacturing platform market is experiencing an increase in the market demands due to the increased cell therapy demands and manufacturers transitioning to automated, closed and scalable production models. Advantageous manufacturing is also getting better regulatory support which is enhancing confidence in the use of platforms. The primary directions of growth are commercial scale-up, process control through software, and quicker production of both autologous and allogeneic therapies. High platform costs, cold chain complexity, process standardization and lack of GMF-ready facilities have remained the greatest challenges.
- Recent industry conferences like ISCT, Cell and Gene Meeting on the Mesa, and Advanced Therapies Congress have assisted in getting the market moving forward with regulators, developers and technology providers coming together around manufacturing scale-up and commercialization. The more recent case of oriented biotech receiving an U.S FDA AMT designation and the growth of automated manufacturing by Cellares indicate the trend in the market towards platform-based cell therapy manufacturing instead of manual and fragmented processes.
Market Segmentation
- Therapy Type Insights (Revenue, USD Mn, 2021 - 2033)
- Autologous Cell Therapy
- Allogeneic Cell Therapy
- Technology Insights (Revenue, USD Mn, 2021 - 2033)
- Somatic Cell Technology
- Cell Immortalization Technology
- Viral Vector Technology
- Genome Editing Technology
- Cell Plasticity Technology
- 3D Technology
- Source Insights (Revenue, USD Mn, 2021 - 2033)
- IPSC’s (Induced pluripotent stem cell)
- Bone Marrow
- Umbilical Cord
- Adipose Tissue
- Neural Stem
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Musculoskeletal
- Cardiovascular
- Gastrointestinal
- Neurological
- Oncology
- Dermatology
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Thermo Fisher Scientific
- Lonza
- Miltenyi Biotec
- Sartorius
- Cytiva
- Terumo Blood and Cell Technologies
- Fresenius Kabi
- Cellares
- Ori Biotech
- ScaleReady
- Wilson Wolf Manufacturing
- Bio-Techne
- Minaris Advanced Therapies
- ElevateBio
- FUJIFILM Biosciences
Sources
Primary Research Interviews
Industry Stakeholders list
- Cell therapy manufacturing directors
- Process development scientists
- Good Manufacturing Practice quality assurance heads
- Regulatory affairs specialists for advanced therapies
- Cold chain and cryopreservation logistics managers
- Contract manufacturing operations leaders
End-users list
- Hospital cell therapy program directors
- Transplant center clinicians
- Academic medical center cell therapy investigators
- Apheresis and cell processing lab managers
- Clinical trial site coordinators in regenerative medicine
- Advanced therapy treatment center procurement heads
Government and International Databases
- U.S. Food and Drug Administration
- European Medicines Agency
- ClinicalTrials.gov
- World Health Organization
- European Commission EudraLex and EudraGMDP databases
Trade Publications
- BioProcess International
- GEN Genetic Engineering and Biotechnology News
- Pharmaceutical Technology
- Pharma Manufacturing
- Outsourced Pharma
- BioPharma Dive
Academic Journals
- Cytotherapy
- Molecular Therapy Methods and Clinical Development
- Nature Biotechnology
- Molecular Therapy
- Cell and Gene Therapy Insights
- Human Gene Therapy
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The New York Times
- The Guardian
- Nikkei Asia
Industry Associations
- International Society for Cell and Gene Therapy
- American Society of Gene and Cell Therapy
- Alliance for Regenerative Medicine
- Foundation for the Accreditation of Cellular Therapy
- International Alliance for Biological Standardization
- Parenteral Drug Association
Public Domain Resources
- FDA guidance documents for cellular and gene therapy
- EMA scientific guidelines on advanced therapy medicinal products
- National Center for Biotechnology Information
- World Health Organization technical resources
- European Commission pharmaceutical legislation resources
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients