Global Cryogenic Vial Market Size and Forecast – 2026 to 2033
The global cryogenic vial market is estimated to be valued at USD 632 Mn in 2026 and is expected to reach USD 982 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.5% from 2026 to 2033. This steady rise in the market can be attributed to the increasing needs for the product in various industries, such as biobanking, pharmaceutical, and clinical research, as a result of the advancements in life sciences and the need for efficient sample preservation technologies.
Key Takeaways of the Global Cryogenic Vial Market
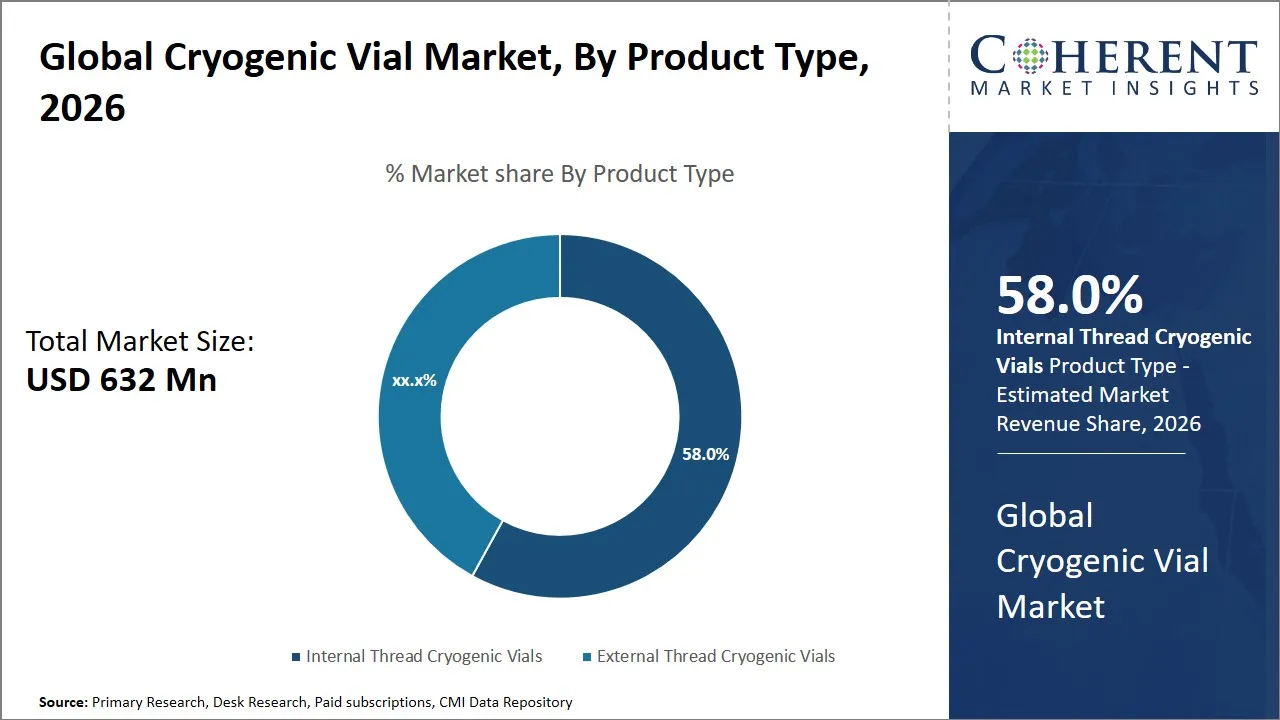
- The internal thread cryogenic vials segment is expected to dominate the global cryogenic vial market, accounting 58.0% share in 2026.
- The flat bottom vials segment is estimated to represent 55.0% of the global cryogenic vial market share in 2026.
- The polypropylene/plastic segment is projected to dominate with 72.0% of the global cryogenic vial market share in 2026.
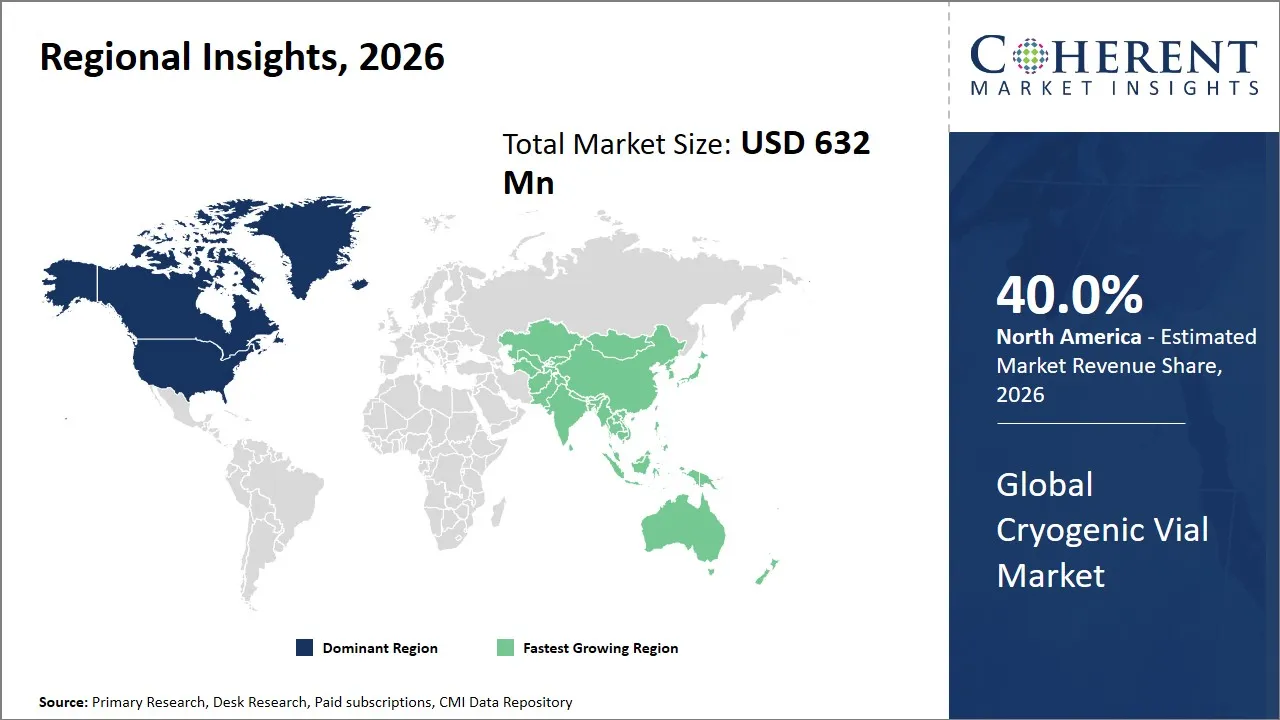
- North America is expected to dominate the global cryogenic vial market, holding a share of 40.0% in 2026.
- Asia Pacific is anticipated to be the fastest-growing region, holding a market share of 27.0% in 2026.
Market Overview
- Cryogenic vials are specialized storage containers that are specifically designed for the storage of biological samples such as cells, tissues, blood, DNA, and other biological samples at extremely low temperatures in the form of liquid nitrogen or mechanical freezers. Cryogenic vials are specifically designed for the storage of samples so that the maximum level of durability, leakage, and contamination can be achieved.
- The cryogenic vial market is expanding at a consistent rate due to the increasing need for biobank, clinical, and pharmaceutical R&D studies. The increasing trend towards cell culture and vaccine development is also adding to the growth of the market.
- Additionally, the increasing trend towards the biotech and life sciences industry and increasing investment in precision medicine and therapy are also contributing to the growth of the market. Cryogenic vials play a key role in the maintenance of samples, thereby facilitating various studies and drug developments at a large scale.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Strengthening Regulatory Frameworks for Sample Storage and Biobanking |
|
|
Growth of Biobanking and Clinical Research Activities |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Internal Thread Cryogenic Vials Segment Dominate the Global Cryogenic Vial Market in 2026?
The internal thread cryogenic vials segment is anticipated to hold a market share of 58.0% in 2026, due to the fact that the internal threading mechanism provides the best locking system, which ensures the best sample security. This locking mechanism ensures that there is no leakage of the sample from the vial to the external environment. This is particularly critical in applications such as biobanking, pharmaceutical research and development, and clinical diagnostics, where sample integrity is of utmost importance for preserving biological materials such as DNA, RNA, proteins, and cell cultures.
Moreover, internal thread cryogenic vials provide a high level of reliability and efficiency in use. This has made internal thread cryogenic vials a highly sought product type in today’s laboratory settings. This is primarily because of the design, which provides a low risk of accidental loosening. This ensures a long-term sample stability. A majority of internal thread cryogenic vials are available in pre-sterilized and DNase/RNase-free formats. This provides a solution to a wide range of applications where such factors are of primary concern. Additionally, internal thread cryogenic vials are suitable for use in automated storage instruments. This provides a further impetus to the use of internal thread cryogenic vials.
Why Does the Flat Bottom Vials Segment Dominate the Global Cryogenic Vial Market in 2026?
The flat bottom vials segment is projected to hold the highest market share of 55.0% in 2026, attributed to their high stability and ease of usage in laboratory conditions. Flat bottom cryogenic vials differ from other cryogenic vials, especially round bottom ones, in that they can be placed upright on laboratory benches, trays, and even in ultra-low-temperature freezers, thus ensuring a high level of safety against accidents, spills, and contaminations during sample handling and processing.
Flat bottom cryogenic vials also have a high level of compatibility with automated systems used in storing and retrieving materials, particularly in today’s highly advanced laboratory conditions where such automated systems play a vital role in ensuring a high level of efficiency along with minimum human error during laboratory procedures and protocols. Flat bottom cryogenic vials have a high level of efficiency in optimizing freezer usage, which is a major criterion in today’s highly demanding laboratory conditions.
For instance, the International Society for Biological and Environmental Repositories emphasizes the significance of standardized and stable sample containers in the management and storage of biological samples for an efficient system of biobanking. The best practices for biorepositories encourage the use of flat bottom cryogenic vials in the storage and management of biological samples in biobanking systems due to the stability in an upright position.
(Source: International Society for Biological and Environmental Repositories)
The Polypropylene/Plastic Segment Dominates the Global Cryogenic Vial Market
The polypropylene/plastic segment is expected to dominate the global cryogenic vial market, accounting for 72.0% share in 2026, attributed to the excellent properties that make these vials highly durable and resistant to chemicals. Moreover, these vials are highly suitable for applications involving extreme temperature conditions. The material, i.e., polypropylene, exhibits excellent stability in extreme conditions and can withstand ultra-low temperatures without deforming. It also remains inert to all chemicals and can be used for preserving biological samples such as DNA, RNA, and cell cultures without any adverse interactions.
In addition, the polypropylene material-based cryogenic vials also provide significant benefits in terms of economics. The material-based vials are highly cost-effective compared to glass material-based vials. This makes these vials highly suitable for applications involving large volumes. The material-based vials are also highly safe to use, as they are light in weight and are highly resistant to shattering. These advantages make polypropylene the most widely used material in the global market for cryogenic vials.
Regulatory and Compliance Standards Influencing Biospecimen Storage, Handling Practices, and Cryogenic Vial Demand
|
Regulatory Aspect |
Description |
Impact on Cryogenic Vial Market |
|
Good Laboratory Practices (GLP) & Good Manufacturing Practices (GMP) |
Regulatory frameworks mandate standardized procedures for handling, storage, and documentation of biological samples in research and clinical environments. |
Drives demand for high-quality, certified cryogenic vials that ensure durability, sterility, and compliance with laboratory standards. |
|
Biobanking Guidelines & Standardization Protocols |
Global biobanking standards emphasize sample traceability, labeling, and long-term preservation under controlled conditions. |
Increases adoption of barcoded, automation-compatible cryogenic vials to support accurate tracking and standardized storage. |
|
Cold Chain & Cryogenic Storage Regulations |
Regulations require strict maintenance of ultra-low temperature conditions (−80°C to −196°C) during storage and transportation of biospecimens. |
Boosts demand for leak-proof, temperature-resistant vials capable of maintaining sample integrity under extreme conditions. |
|
Contamination Control & Sterility Requirements |
Regulatory bodies emphasize DNase/RNase-free environments and contamination prevention for sensitive biological samples. |
Encourages use of sterile, airtight cryogenic vials with advanced sealing mechanisms, supporting reliable long-term preservation. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Cryogenic Vial Market Analysis and Trends
The North America region is projected to lead the market with a 40.0% share in 2026, attributed to the well-established healthcare infrastructure and the presence of a well-developed biotechnology and pharmaceutical industry. The U.S. and Canada have a number of well-developed research institutes that make heavy use of cryogenic vials for sample storage.
Moreover, the regulatory environment in the region focuses on the quality and safety of products, thus encouraging the use of high-quality cryogenic vials for sample storage. Some of the key players in the North America have a well-established presence and R&D facilities in the region, introducing innovative products that cater to the needs of the market. The well-developed infrastructure and logistics in the region enable the key players to gain a strong foothold in the market by ensuring timely delivery of the product to the customers.
For instance, the National Cancer Institute emphasizes the significance of standardized storage practices in biobanks to ensure the quality of cancer research, further emphasizing the requirement for quality cryogenic storage consumables.
(Source: National Cancer Institute)
Asia Pacific Cryogenic Vial Market Analysis and Trends
The Asia Pacific region is expected to exhibit the fastest growth in the cryogenic vial market, contributing 27.0% share in 2026, attributed to increased investments in biomedical research and growing healthcare expenditures in regions such as China, India, Japan, and South Korea. The governments in these regions have recognized the potential for life sciences and biotechnology through various national initiatives. This has led to increased investments in the development of infrastructure and growing clinical research activities. The growing pharmaceutical industry and the increasing number of biobanks also contribute to the growth in the market.
In addition to this, the growing industry for cryogenic vials in the Asia Pacific region can be attributed to the increasing number of local players in the market, such as Bio-Rad Laboratories, Greiner Bio-One Asia Pacific, and others. The favorable import and export policies also contribute to the growth in the market.
For instance, in June 2023, Corning Incorporated and SGD Pharma formed a joint venture with the aim of increasing access to the company’s Velocity vial technology in India. The move was aimed at increasing the level of pharmaceutical vial manufacturing in the Asia Pacific region. This move was a clear indication of the increased focus on the development of high-quality vial technologies in the region and, by extension, the need for storage containers such as cryogenic vials in the region.
(Source: Corning Incorporated)
Cryogenic Vial Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Cryogenic Vial Market?
The U.S. cryogenic vial market has a major role to play, considering the dominance of the region in medical research and developments in biotechnologies. Industry majors like Thermo Fisher Scientific have a major foothold in the region, providing high-end cryogenic vials with better material quality to ensure better durability and efficiency in sealing. The presence of research hospitals, academic centers, and biopharmaceutical organizations ensures a consistent market base, along with support from organizations like the FDA to ensure conformance to the quality of the products, thereby generating a sense of trust to provide high-quality products to the biobanking and cell therapy industry.
For instance, the National Institutes of Health (NIH) sponsors large-scale biobanking projects such as the "All of Us Research Program," which includes the collection and long-term storage of biological samples such as blood and DNA to facilitate precision medicine. This creates a high demand for cryogenic vials to support the storage facilities, thereby increasing the market for cryogenic vial in the U.S.
(Source: National Institutes of Health (NIH))
How is Germany Helping in the Growth of the Cryogenic Vial Market?
Germany cryogenic vial market is driven by the well-developed life sciences industry and the robust pharmaceutical manufacturing sector. The local players like Merck KGaA use their history and expertise to provide innovative cryovials for clinical and industrial applications. The government’s encouragement of scientific research and the quality demands contribute to the growth of the cryogenic vial market in Germany. The geographical position of the Germany allows it to act as a bridgehead for the whole of Europe.
Key Drivers for the Growth of the China Cryogenic Vial Market
The growing biomedical research industry in China is a key factor for the increasing market for cryogenic vial in China. The government is taking many steps to improve the biotechnology industry in China through initiatives such as “Made in China 2025.” This has led to an increased demand for life science products, such as cryogenic vials. Companies such as Bio-Rad are putting in a lot of efforts to improve capacities and transfer technologies to cater to the increasing need for clinical trial and regenerative medicine applications in China. The regulatory scenario in China is also changing, helping to improve the quality of cryogenic vials.
India Cryogenic Vial Market Trends
The India market for cryogenic vial is growing at a fast rate due to the increasing pharmaceutical industry and clinical research activities. The government's efforts to improve the research infrastructure and the need for biobanks in hospitals and academic institutes have also contributed to the growth of the cryogenic vial market in India. Multinational companies are partnering with local players to increase the accessibility and affordability of the product. The cost advantage and the emergence of biopharma clusters make India an important part of the global map for the manufacture of cryogenic vials.
Funding & Reimbursement Support for Genomics and Biobanking Programs
|
Funding/ Policy Area |
Description |
Impact on Cryogenic Vial Market |
|
Government Funding for Genomics & Precision Medicine |
Governments are increasingly investing in large-scale genomics programs and national precision medicine initiatives to advance disease understanding and personalized treatments. |
Drives large-scale biospecimen collection and long-term storage, increasing demand for cryogenic vials used in research and clinical applications. |
|
Public Biobank Infrastructure Investments |
National and regional authorities are funding the establishment and expansion of biobanks to support clinical trials and translational research. |
Boosts demand for high-volume, reliable cryogenic storage consumables, including vials for organized and long-term sample preservation. |
|
Reimbursement Support for Advanced Diagnostics |
Expanding reimbursement coverage for genomic testing and molecular diagnostics is encouraging adoption of advanced diagnostic procedures. |
Leads to increased sample collection and processing, thereby supporting consistent demand for cryogenic vials in diagnostic workflows. |
|
Grants & Research Funding Programs |
Academic institutions and research organizations receive funding through grants for biomedical and life sciences research projects. |
Sustains continuous demand for cost-effective and high-quality cryogenic vials across research laboratories and universities. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- On March 4, 2026, Azenta Life Sciences completed the acquisition of UK Biocentre Limited. The acquisition was aimed at enhancing the biorepository capabilities to fulfill the need for enhanced infrastructure in the storage of biological samples at a large scale, especially in the life sciences industry, including the need for cryogenic storage solutions such as vials.
- In December 2025, Thermo Fisher Scientific expanded its bioprocess design centers throughout Asia. This included a new facility in India, a strategic step to strengthen biopharmaceutical research and manufacturing capabilities. The intention was to better address the increasing demand for sample storage and handling solutions, including cryogenic vials, within biobanking and life science research.
- In July 2025, Alfa Laval, a company at the forefront in the manufacturing of quality products, completed the acquisition of the cryogenic business from the French industrial group, Fives. This acquisition has helped the company in enhancing the offerings in the field of cryogenic processing and low-temperature technology, thus contributing to the demand for cryogenic vials.
Top Strategies Followed by Global Cryogenic Vial Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders emphasize product innovation, materials, and automation compatibility during the establishment of global manufacturing and distribution infrastructures through strategic partnerships. |
In March 2024, Merck KGaA intends to pour over US$ 348 million (300 million Euros) into establishing a new bioprocessing production facility in Daejeon, South Korea. This center bolstered their biologics development and sample processing capabilities. Consequently, this expansion indirectly boosts the need for cryogenic storage solutions, such as vials. |
|
Mid-Level Players |
Mid-level players focus on effective and cost-conscious manufacturing strategies, strategic partnerships, and local growth with a focus on product quality and scalability for research institutions. |
In April 2024, DWK Life Sciences broadened its selection of laboratory consumables. They also bolstered their distribution partnerships, aiming to make top-tier sample storage solutions more readily available worldwide. |
|
Small-Scale Players |
Small-scale players emphasize product development for specific applications through the process of customizing and accelerating product innovation with the aid of advanced materials and manufacturing flexibility. |
In March 2024, Advanced sample storage tubes have been introduced in by Micronic, offering enhanced traceability features such as laser-etched barcodes. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Cryogenic Vial Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 632 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.5% | 2033 Value Projection: | USD 982 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
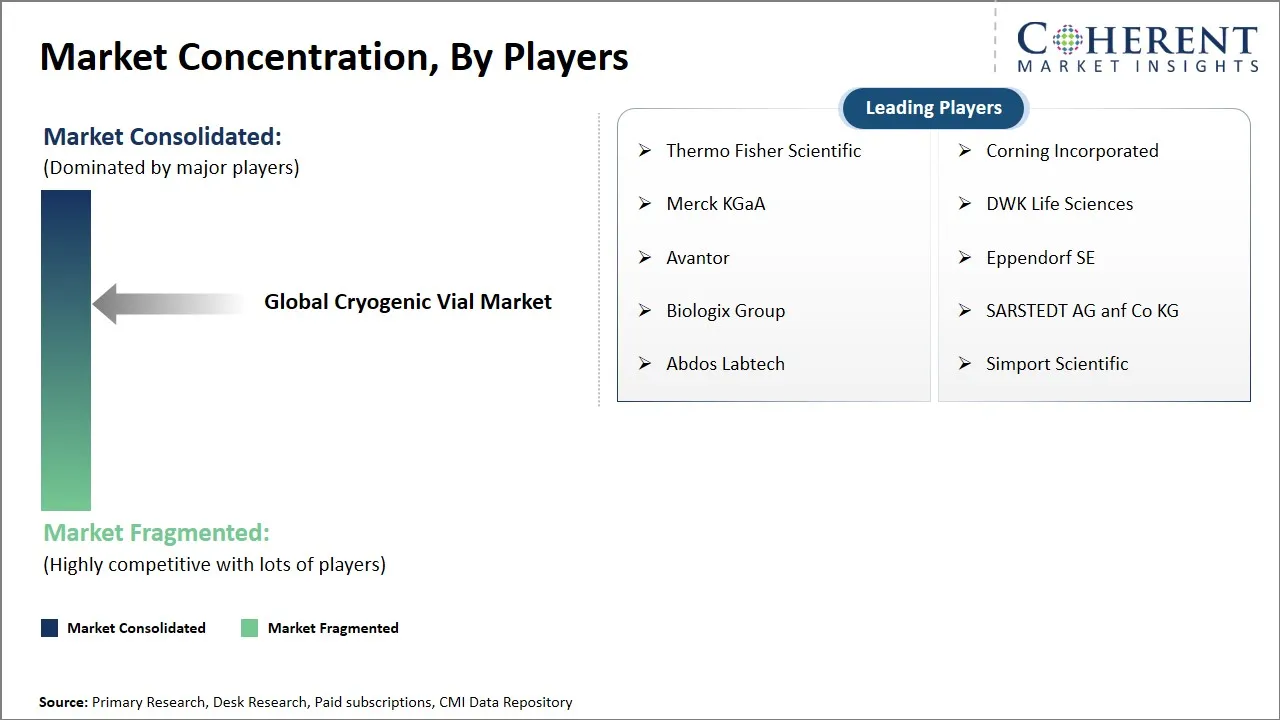
Thermo Fisher Scientific, Corning Incorporated, Merck KGaA, DWK Life Sciences, Avantor, Eppendorf SE, Biologix Group, SARSTEDT AG anf Co KG, Abdos Labtech, and Simport Scientific |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Cryogenic Vial Market Dynamics
To learn more about this report, Request Free Sample
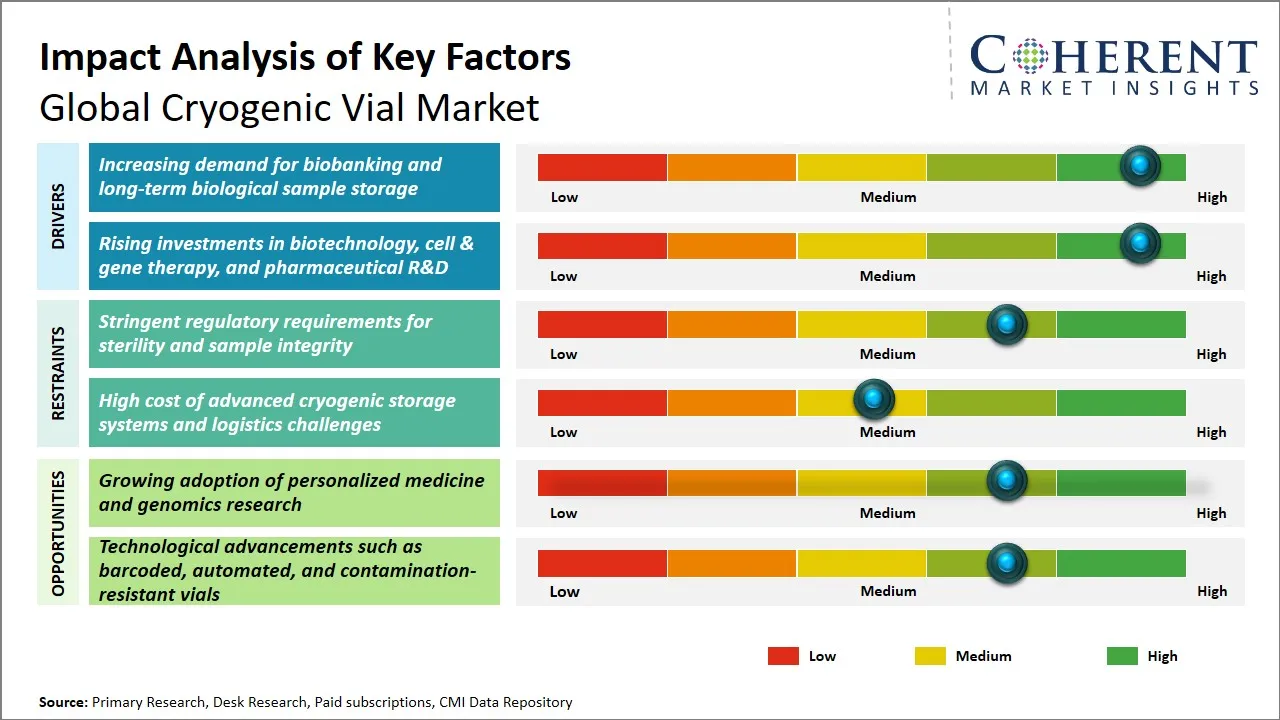
Global Cryogenic Vial Market Driver – Increasing Demand for Biobanking and Long-Term Biological Sample Storage
The growing scope related to biobanking projects worldwide has increased the market for cryogenic vial. These vials are required for the storage of biological samples for long periods. The growing activities related to biomedical research, personalized medicine, and drug development have increased the market for these cryogenic vials. The ability to protect the samples from contamination and degradation at ultra-low temperatures makes the cryogenic vials highly suitable for storage.
The growing activities related to genetic research and regenerative medicine have increased the market for these vials. This is due to the fact that many organizations are investing in biobanking facilities. The ability to protect biological samples for long periods and analyze them in the future for drug development and disease diagnosis also plays an important role in increasing the market for these cryogenic vials. This increases the significance of these vials in modern life sciences and healthcare sectors.
For instance, the UK Biobank holds around 17 million biological sample containers, with millions being stored in automated freezers kept at -80 degrees Celsius, and backup samples stored in liquid nitrogen and kept at -196 degrees Celsius.
(Source: UK Biobank Limited)
Global Cryogenic Vial Market Opportunity - Growing Adoption of Personalized Medicine and Genomics Research
The global cryogenic vial market is poised to gain from the increasing trend of personalized medicine and advancements in genomics research that are revolutionizing the field of medicine and the life sciences. Personalized medicine requires the collection and storage of samples such as DNA, RNA, and proteins in a manner that maintains the integrity of the sample and prevents degradation and contamination. Cryogenic vials find a special place in the collection and storage of such samples by maintaining the integrity of the sample at ultra-low temperature and preventing degradation and contamination.
The increasing trend of next-generation sequencing (NGS) and the use of technologies such as CRISPR and the identification of biomarkers for various diseases also contribute to the increasing trend of the use of cryogenic vials for the collection and storage of samples for genomics research and personalized medicine. The increasing trend of biobanks and genomics research in clinical trials is also contributing to the increasing trend of the use of cryogenic vials for the collection and storage of samples for genomics research and personalized medicine
For instance, in March 2025, a research paper published by SAGE Publications highlighted the importance of storage containers in ensuring the integrity and analysis of the results in cryopreserved biospecimens. This shows that the efficacy of storage containers has a direct impact on the results of the research, hence the need for advanced cryogenic vials.
(Source: SAGE Publications)
Analyst Opinion (Expert Opinion)
- The market for cryogenic vial is positioned for continuous growth in the future, mainly driven by the tremendous growth prospects of the biobanking, cell and gene therapy, and regenerative medicine markets.
- Regarding the regulatory requirements, the increasing importance of sample traceability, quality assurance, and standardization in storage practices is pushing research and biopharmaceutical organizations to use high-quality and high-performance cryogenic vials. This is more evident in clinical trial and cell and gene therapy procedures, as the quality of the sample is more crucial to the success of the research.
- The increasing importance of the integration of automated storage systems and digital laboratory concepts in the market is expected to give a significant edge to the players.
- Furthermore, the increasing global footprint of the pharmaceutical industry in the R&D sector and the increasing investment in precision medicine acta as a market growth drivers, although to a limited extent.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Internal Thread Cryogenic Vials
- External Thread Cryogenic Vials
- Design Insights (Revenue, USD Mn, 2021 - 2033)
- Flat Bottom Vials
- Round Bottom Vials
- Material Insights (Revenue, USD Mn, 2021 - 2033)
- Polypropylene/Plastic
- Glass
- Capacity Insights (Revenue, USD Mn, 2021 - 2033)
- Up to 1 mL
- 1-2 mL
- 2-5 mL
- Above 5 mL
- Sterility Insights (Revenue, USD Mn, 2021 - 2033)
- Sterile Cryogenic Vials
- Non-Sterile Cryogenic Vials
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Cryopreservation
- Sample Storage
- Drug Discovery and Development
- Cell and Gene Therapy
- Transport of Biological Samples
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Biobanks
- Pharmaceutical Companies
- Biopharmaceutical Companies
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Hospitals and Diagnostic Laboratories
- Academic and Research Institutes
- Cell Therapy/Gene Therapy Laboratories
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Thermo Fisher Scientific
- Corning Incorporated
- Merck KGaA
- DWK Life Sciences
- Avantor
- Eppendorf SE
- Biologix Group
- SARSTEDT AG anf Co KG
- Abdos Labtech
- Simport Scientific
Sources
Primary Research Interviews
Industry Stakeholders
- Cryogenic Vial Manufacturers
- Laboratory Consumables and Life Science Plasticware Companies
- Biobanking Solution Providers
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Regulatory and Quality Assurance Experts
End Users
- Biobanks and Biorepositories
- Pharmaceutical and Biotechnology Companies
- Academic & Research Institutes
- Clinical and Diagnostic Laboratories
- Hospitals and Research-driven Healthcare Facilities
Government and International Databases
- U.S. Food and Drug Administration (FDA) – Guidelines on biological sample storage and handling
- European Medicines Agency (EMA) – Clinical research and biologics regulations
- World Health Organization (WHO) – Biobanking and laboratory best practices
- National Institutes of Health (NIH) – Biomedical research and sample storage standards
- OECD – Guidelines for biological resource centers and biorepositories
Trade Publications
- Laboratory Equipment Magazine
- Genetic Engineering & Biotechnology News (GEN)
- Lab Manager Magazine
- BioPharma International
- Pharmaceutical Technology
Academic Journals
- Cryobiology
- Journal of Biopreservation and Biobanking
- Nature Biotechnology
- Journal of Pharmaceutical Sciences
- Biologicals
Reputable Newspapers
- Financial Times – Global pharmaceutical and life sciences trends
- The Wall Street Journal – Biotech investments and R&D developments
Industry Associations
- International Society for Biological and Environmental Repositories (ISBER)
- Biotechnology Innovation Organization (BIO)
- American Association for Clinical Chemistry (AACC)
- Society for Cryobiology
Public Domain Resources
- WHO Technical Reports on Laboratory Biosafety
- NIH Research Publications
- Government-funded biobanking project reports
- Open-access scientific repositories and databases
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients