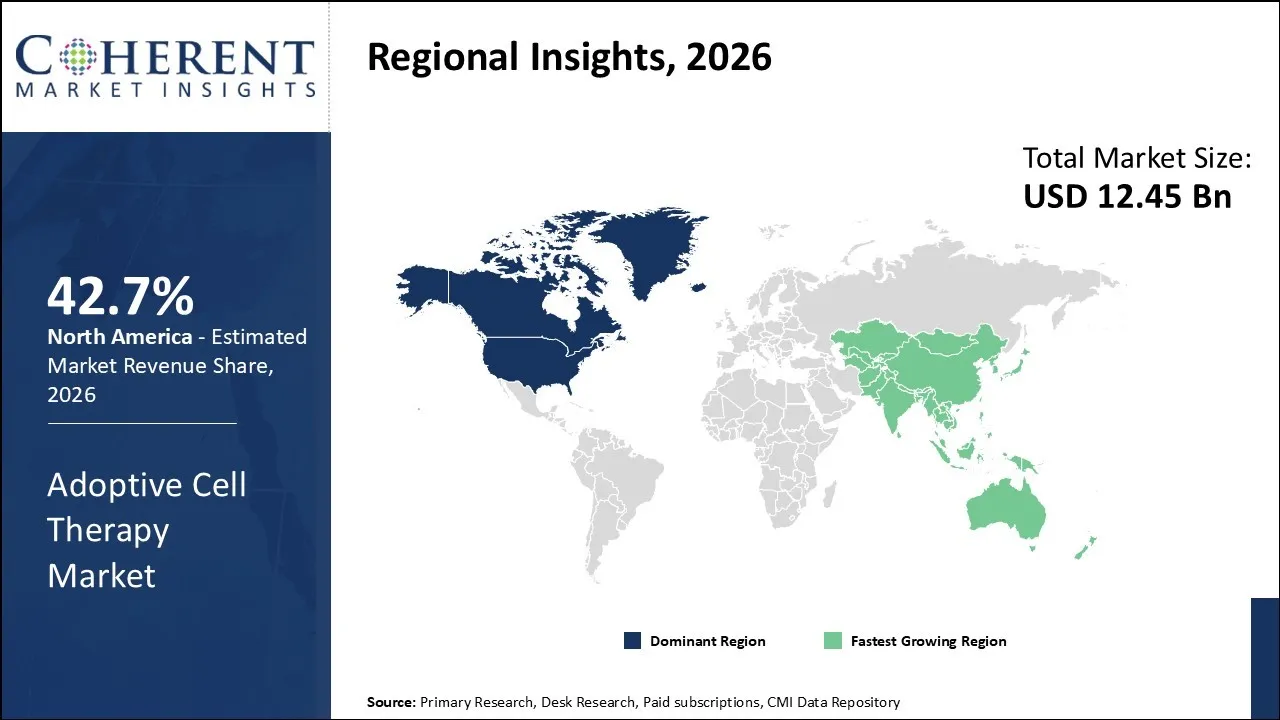
The adoptive cell therapy market was valued at USD 12.45 Bn in 2026 and is forecast to reach a value of USD 22.4 Bn by 2033 at a CAGR of 12.1% between 2026 and 2033.
Adoptive cell therapy (ACT) is a kind of immunotherapy, in which T cells are genetically modified to express a chimeric antigen receptor (CAR) or T cell receptor (TCR). Adoptive cell therapy has made a great difference in treating multiple types of tumors. Some approaches of ACT involve directly isolating body’s immune cells and simply expanding their numbers, whereas other approaches involve genetically engineering body’s immune cells (via gene therapy) to enhance cancer-fighting capabilities
|
Current Event |
Description and its Impact |
|
Technological Advances in Adoptive Cell Therapy |
|
|
Regulatory Approvals and Market Adoption |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of type, the CAR-T segment is expected to lead the market with 40.4% share in 2026, due to several compelling factors rooted in its clinical success, regulatory approvals, and increasing adoption across various hematologic malignancies. CAR-T (Chimeric Antigen Receptor T-cell) therapy has emerged as a groundbreaking innovation, transforming the landscape of cancer treatment, particularly for refractory or relapsed B-cell malignancies such as certain leukemias and lymphomas.
For instance, according to an article published in The American Journal of Managed Care in January 2026, an analysis of open claims data from the HealthVerity Marketplace dataset covering 43 patients treated across four new authorized treatment centers between July 2023 and June 2025 showed a gradual increase in CAR-T therapy adoption. The number of treated patients rose from fewer than 11 in 2023 to 16 in 2024 and 22 in the first half of 2025, reflecting expanding clinical uptake and supporting the segment’s projected market leadership.
In terms of drug class, the lymphoma segment is expected to hold 48.2% share of the market in 2026. Due to its significant clinical prevalence and the established success of adoptive cell therapies in treating various subtypes of this hematologic malignancy. The prominence of lymphoma in the ACT market is underpinned by the fact that non-Hodgkin lymphoma (NHL), particularly diffuse large B-cell lymphoma (DLBCL), represents a substantial portion of lymphoid cancers diagnosed worldwide.
For instance, according to a study published in Nature Communications in May 2025, CD19-directed CAR-T cell therapies approved by the United States Food and Drug Administration for relapsed or refractory DLBCL have demonstrated complete response rates of approximately 40–54%, significantly improving outcomes for patients with previously limited treatment options. These developments reinforce lymphoma’s leading position within the adoptive cell therapy market.
In terms of end user, the hospitals segment is projected to account for 55 % share of the market in 2026. Owing to their extensive infrastructure, established patient base, and multifaceted treatment capabilities that encompass a broad spectrum of diseases beyond cancer. Hospitals generally operate as comprehensive healthcare delivery hubs, offering a wide array of services that include but are not limited to oncology, hematology, immunology, and regenerative medicine, making them ideally positioned to incorporate sophisticated therapies such as adoptive cell therapy.
To learn more about this report, Request Free Sample
North America is expected to dominate the adoptive cell therapy market with 42.7% share in 2026. This leadership is supported by a highly advanced healthcare infrastructure, significant government and private sector investments in cell therapy research, and a strong presence of biotechnology companies pioneering innovative therapies.
For instance, in August 2025, the Food and Drug Administration (FDA) approved afamitresgene autoleucel (afami-cel, brand name Tecelra) to treat certain patients with metastatic synovial sarcoma, marking the first approval of a T-cell receptor (TCR) therapy for cancer. In addition, multiple CAR-T therapies for hematologic malignancies, including relapsed/refractory B-cell leukemias and lymphomas, have also received FDA approval, demonstrating the United States’ role as a global hub for adoptive cell therapy innovation. These regulatory milestones highlight North America’s capacity to both develop and deliver advanced cell therapies, reinforcing its projected market dominance.
Asia Pacific is expected to exhibit the fastest growth, this growth can be attributed to factors such as rapid technological adoption, the presence of large, untapped patient pools, and significant governmental encouragement in including countries such as China, Japan, South Korea, and India
For instance, in November 2025, Marengo CIMS Hospital in Ahmedabad, India, has introduced CAR-T (Chimeric Antigen Receptor T-cell) therapy for the first time. This is an important step forward in advanced cancer care and brings next-generation, personalized treatment to patients at the hospital.
The U.S. leads the region owing to its cutting-edge advancements in cancer immunotherapy, specifically chimeric antigen receptor T-cell (CAR-T) therapies, which have been clinically validated and approved for multiple hematologic malignancies.
For instance, in December 2025, the U.S. Food and Drug Administration (FDA) has approved a new use for Breyanzi (Lisocabtagene maraleucel), making it the first CAR T-cell therapy in the U.S. for adults with marginal zone lymphoma (MZL) who haven't responded to or have relapsed after two or more treatments. Breyanzi works by using genetically modified T-cells from the patient’s own body to target and kill cancer cells.
In China, significant advancements in domestically developed CAR-T therapies have resulted in clinical outcomes that align with global standards, particularly in hematologic malignancies. The country has made substantial strides in developing innovative CAR-T cell therapies, which have shown promising results in treating relapsed/refractory B-cell malignancies like acute lymphoblastic leukemia (ALL) and non-Hodgkin lymphoma (NHL).
For instance, according to a study published in Discover Oncology, CAR-T treatments for B-cell malignancies have achieved overall response rates (ORR) between 79% and 89%, highlighting their high efficacy in relapsed or refractory diseases. In multiple myeloma, long-term data reveals a 64% progression-free survival rate at 12 months, indicating durable responses in patients. While these results demonstrate remarkable success in blood cancers, CAR-T therapies for solid tumors still face significant hurdles, including antigen heterogeneity
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12.45 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.1% | 2033 Value Projection: | USD 22.4 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, Gilead Sciences, Inc., Castle Creek Biosciences, Inc., Lineage Cell Therapeutics, Inc., Transgene SA, Cellectis, ImmunityBio, Inc., Sorrento Therapeutics, bluebird bio, Inc., Arcellx, Sana Biotechnology, Inc., Biodesix, Inc, and Laurus Labs |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising prevalence of cancer globally is a significant factor propelling the growth of the adoptive cell therapy (ACT) market. Cancer, being one of the leading causes of morbidity and mortality worldwide, has seen a steady increase in incidence rates due to aging populations, lifestyle changes, and environmental factors. Adoptive cell therapy, which involves engineering or expanding patient-derived immune cells to target and eradicate cancer cells, represents a promising advancement in personalized medicine.
The increasing number of product approvals for adoptive cell therapy (ACT) has significantly propelled the growth of the Adoptive Cell Therapy Market by validating the clinical efficacy and safety of these innovative treatments. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have steadily expanded their approvals for various adoptive cell therapy products, particularly in the realm of chimeric antigen receptor T-cell (CAR-T) therapies.
The development of allogeneic, off-the-shelf therapies with mass production potential is fundamentally transforming the adoptive cell therapy (ACT) market by addressing several limitations associated with autologous treatments. Traditionally, ACT—such as CAR-T cell therapies—relies on harvesting and modifying a patient’s own cells, a process that is time-consuming, costly, and often yields variable product quality due to patient-specific factors.
The adoptive cell therapy market is transitioning from proof-of-concept to proof-of-practicality, driven by substantial technological innovation and expanding clinical applications. Rising cancer prevalence, coupled with increasing demand for personalized medicine approaches, continues to propel market interest among healthcare systems and pharmaceutical companies.
The emergence of advanced technologies such as CRISPR-Cas9 gene editing and CAR-T cell therapies represents a fundamental shift in treatment efficacy, offering more targeted and durable therapeutic options. Recent FDA approvals and the development of off-the-shelf therapies have accelerated commercialization momentum across the sector.
However, significant challenges warrant careful consideration. Manufacturing scalability and cost constraints remain formidable barriers to widespread adoption, while complex regulatory landscapes and safety concerns, including immune-related adverse events necessitate rigorous clinical validation and long-term efficacy monitoring. Patient eligibility criteria further limit addressable market segments.
North America remains the dominant region, driven by robust FDA approval pathways and substantial healthcare investment. Europe and Asia-Pacific are positioned for accelerated growth, supported by increasing government funding and emerging strategic collaborations between pharmaceutical companies and academic institutions. Notably, the shift toward outpatient CAR-T administration and the rise of autologous cell therapies indicate the market's maturation and growing operational sophistication.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients