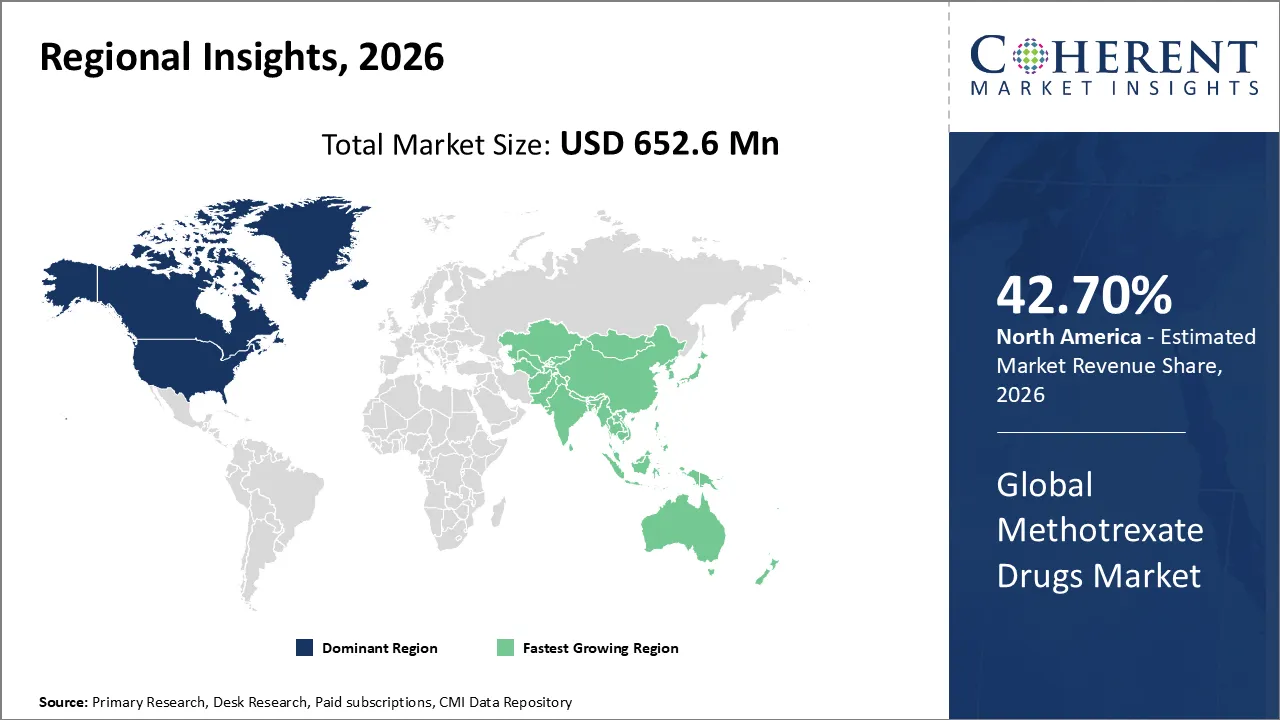
Methotrexate drugs market is estimated to be valued at USD 652.6 Mn in 2026 and is expected to reach USD 743.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 2.8% from 2026 to 2033, driven by oncology and autoimmune disease applications.
Methotrexate is a folate antagonist. Methotrexate is one of the most effective and widely used medications for treating inflammatory types of arthritis. It is also one of the safest arthritis drugs. Methotrexate drugs are indicated for the treatment of Psoriasis, Rheumatoid Arthritis. Methotrexate is also used to treat certain types of cancer including cancers that begin in the tissues that form around a fertilized egg in the uterus, breast cancer, lung cancer, certain cancers of the head and neck, certain types of lymphoma, and leukemia (cancer that begins in the white blood cells).
Methotrexate treats cancer by slowing the growth of cancer cells. Methotrexate treats psoriasis by slowing the growth of skin cells to stop scales from forming. Methotrexate may treat rheumatoid arthritis by decreasing the activity of the immune system.
|
Current Event |
Description and its Impact |
|
Regulatory Changes in Biosimilar and Generic Drug Approvals |
|
|
Rheumatoid Arthritis and Cancer Treatment Landscape Evolution |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Treatment Strategy |
ACR20 Response |
ACR50 Response |
ACR70 Response |
Radiographic Progression |
Notes |
|
Methotrexate Monotherapy |
30–40% |
20–25% |
10–15% |
Moderate progression |
Response seen in 6–12 weeks |
|
Methotrexate + TNF Inhibitors |
60–70% |
40–50% |
25–30% |
↓ 50–60% vs monotherapy |
Common biologics: adalimumab, etanercept, infliximab |
|
Methotrexate + JAK Inhibitors |
65–75% |
45–55% |
30–35% |
↓ 55–65% vs monotherapy |
Common JAK inhibitors: tofacitinib, baricitinib |
|
Predictive Research (AI/ML) |
~70–80% accuracy |
— |
— |
— |
Biomarkers + machine learning improve prediction accuracy to >80% |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of treatment type, the rheumatoid arthritis segment is expected to hold 57% share of the market in 2026, due to it is the best first-line treatment. Clinical guidelines always suggest methotrexate for early and long-term disease control. Because it is cheap, works well for a long time, and high volume of patients, it will always be the biggest treatment area in the world.
For instance, in October 2025, Beta Drugs Limited got approval from the DCGI to sell methotrexate oral solution, which is India's first such product. This formulation is used to treat rheumatoid arthritis, juvenile idiopathic arthritis, psoriasis, psoriatic arthritis, and some types of cancer. This new idea makes it easier for patients to follow their treatment plan by giving them a convenient option to tablets and injections for long-term autoimmune care.
In terms of route of administration, the injection segment is expected to lead the market with 62% share in 2026, because it is more bioavailable and has fewer side effects on the stomach than oral forms. Subcutaneous and intramuscular routes enhance patient compliance and therapeutic results. Doctors are growing increasingly preferring injections for consistent dosing, making it the most common way to give medicine for autoimmune and cancer care.
For instance, in August 2025, The FDA provided Aldeyra Therapeutics the Fast Track Designation for ADX-2191, a sterile intravitreal methotrexate injection. The drug is being made to treat primary vitreoretinal lymphoma and retinitis pigmentosa. This shows how methotrexate injections have changed, making them useful for more than just rheumatoid arthritis and cancer. They can now also be used to treat rare eye diseases.
In terms of distribution channel, the hospital pharmacies segment is projected to account for 53% share of the market in 2026, because of centralized treatment plans for cancer and arthritis. When giving methotrexate, it is often necessary to monitor on toxicity and therapeutic drug levels, which makes hospital-based dispensing even more important. The purchasing power of institutions and the oversight of regulations make hospital pharmacies even stronger as the main way to get medicines around the world.
To learn more about this report, Request Free Sample
North America is expected to dominate the methotrexate drugs market with 42.70% share in 2026, because autoimmune diseases have high prevalence, healthcare is well-developed, and regulatory approvals are strong. The market is even stronger as many doctors are increasing adoption, there are new formulations available, and patients’ awareness. This ensures steady growth and easy access across a wide range of therapeutic uses.
For instance, in September 2025, Researchers found that methotrexate, a common arthritis drug, can lower blood pressure significantly. Clinical trials showed that systolic and diastolic levels went down, which suggests that it has cardiovascular benefits beyond its anti-inflammatory role. This discovery could broaden the therapeutic applications of methotrexate, providing dual benefits in the treatment of arthritis and the mitigation of hypertension risks.
Asia Pacific is expected to exhibit the fastest growing region, due to more people are getting sick in India and China, healthcare infrastructure is getting better, and generics are becoming more affordable. The region is the fastest-growing market segment in the world because of government programs, more people knowing about autoimmune disorders, and big investments in pharmaceutical research and development.
For instance, in October 2025, Hyphens Pharma has started selling Metoject Pen, a methotrexate auto-injector, in Thailand and Cambodia. This change makes it easier for people in Southeast Asia with rheumatoid arthritis and psoriasis to get methotrexate therapy on their own, which helps them stick to their treatment and makes established autoimmune disease management options more widely available in the region.
The U.S. Methotrexate Drugs Market will be in high demand in 2026 due to high incidence rate of autoimmune diseases, the advanced healthcare system, and doctors great utilization. Regulatory approvals, new drug formulations, and patient awareness all help make methotrexate more accessible. This means that the U.S. will continue to be a major player in the growth of the global methotrexate market.
For instance, in May 2025, A study in the United States found that methotrexate works just as well as prednisone for treating pulmonary sarcoidosis. Published in Chest, the trial demonstrated that methotrexate diminished inflammation and preserved lung function, presenting a steroid-sparing alternative. This gives patients fewer long-term side effects and makes methotrexate more useful for treating lung problems.
In 2026, India's methotrexate drugs market will grow because more people are getting autoimmune disorders, growing cases of cancer, and the country's healthcare system is getting bigger. Affordable generics, government programs to make drugs easier to get, and more patients knowing about them all help adoption grow even more, making India a key growth engine in the Asia-Pacific region.
For instance, in October, 2025, AIIMS Bhopal has been given a national research grant to study "metabolomic profiling of methotrexate response in rheumatoid arthritis patients." The goal of the project is to find biomarkers that can predict how well treatment will work, which will make personalized therapy possible. This project makes India a bigger player in precision medicine and could help people with autoimmune diseases manage them better over time.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 652.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.6% | 2033 Value Projection: | USD 743.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
LEO Pharma A/S, Bristol Myers Squibb, Nordic Pharma, Eli Lilly and Company, Horizon Therapeutics plc., Cumberland Pharmaceuticals Inc., Azurity Pharmaceuticals, Inc., Pfizer Inc., Hikma Pharmaceuticals PLC., and Onco Therapies Limited (Strides Arcolab Limited) |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The methotrexate drugs market growth is rapid because a growing number of products are getting approved by regulatory bodies around the world. Regulatory clearance makes sure that they are safe, effective, and of high quality. This builds trust between patients and healthcare providers. As more methotrexate formulations get approved, drug companies can reach more areas and build stronger portfolios. This trend not only speeds up commercialization but also makes it easier for more people to use, which leads to more people using it and more market growth worldwide.
The future of methotrexate therapies is being shaped by ongoing investment in research and development. To improve patient outcomes, drug companies are working on new formulations, better ways to deliver drugs, and combination therapies. The Methotrexate Drugs Market demand is growing because new products are always launching that offer better ways to treat conditions like rheumatoid arthritis, psoriasis, and some cancers. These efforts not only make the therapeutic landscape more diverse, but they also give companies a competitive edge that will help them grow and stay relevant in the global pharmaceutical industry for a long time.
Methotrexate is being studied increasingly for uses other than its usual uses in rheumatoid arthritis and some cancers. Therapeutic applications have now grown to include treating psoriasis, inflammatory bowel disease, and other autoimmune diseases. This diversification makes the drug more useful in a wider range of medical fields, opening up new ways to care for patients and enter new markets. The methotrexate drugs market forecast predicts steady growth as more people use the drug in different therapeutic areas and new formulations are developed.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients