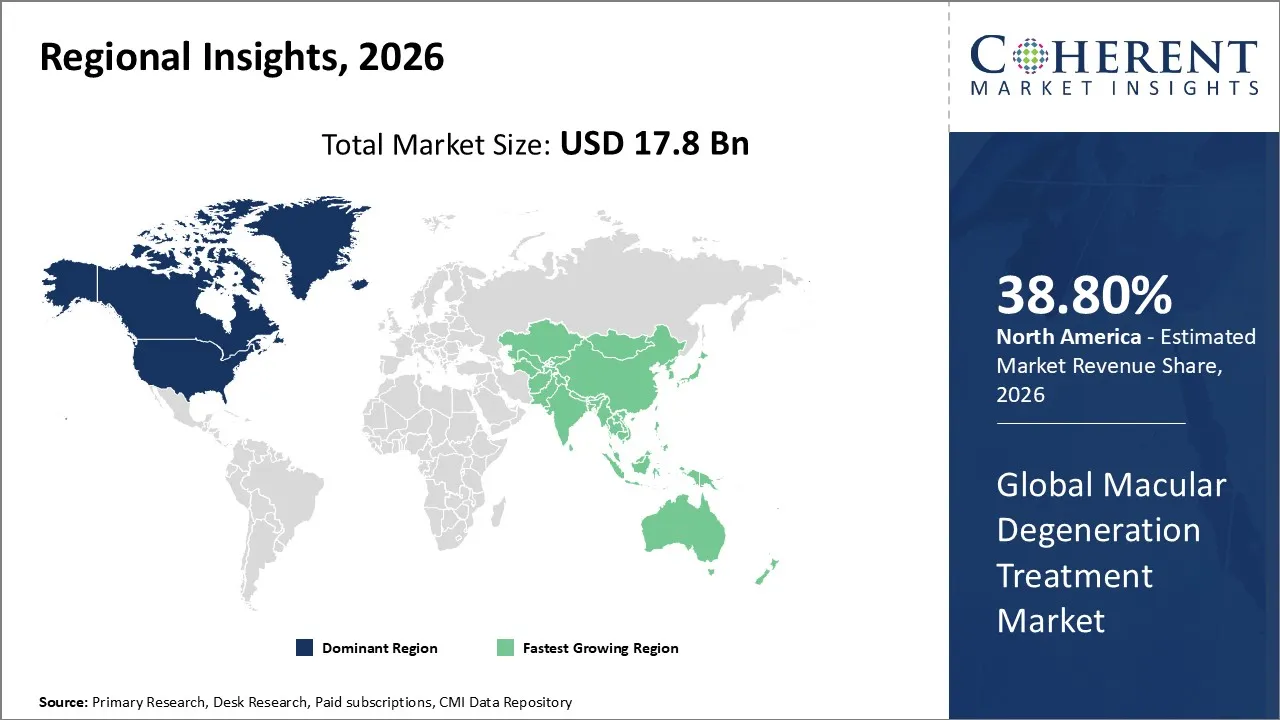
The Macular Degeneration Treatment Market is estimated to be valued at USD 17.8 Bn in 2026 and is expected to reach USD 28.6 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.7% from 2026 to 2033.
Researchers, healthcare providers, and pharmaceutical companies are driving the expansion of the Macular Degeneration Treatment Market as the prevalence of age-related macular degeneration rises and the global elderly population grows. Healthcare organizations are increasing awareness about early diagnosis and adopting advanced retinal imaging and therapeutic technologies. Clinicians use treatment approaches such as anti-VEGF therapies, nutritional supplements, and emerging regenerative and gene-based treatments. Providers emphasize early intervention to prevent vision loss, while industry players continue to strengthen research efforts, expand ophthalmology infrastructure, and introduce innovative drug delivery systems to advance the market.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments |
|
|
Technological Innovations and Clinical Advancements |
|
|
Demographic and Epidemiological Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Intravenous Route acquired the prominent market share of 52.7% in 2026. Healthcare providers increasingly use the intravenous (IV) route because it delivers medications and fluids directly into the bloodstream, producing a rapid effect and precise dosing. Clinicians administer IV therapies for critical care, chemotherapy, and hydration to achieve therapeutic levels quickly and effectively. Innovations in catheter design, infusion pumps, and sterile techniques enhance patient safety and comfort. Expanding hospital infrastructure, the growing prevalence of acute and chronic conditions, and ongoing clinician training actively promote wider adoption of intravenous treatments across medical settings.
Wet Age-related Macular Degeneration hold the largest market share of 53.4% in 2026. The growing aging population drives the incidence of Wet Age-related Macular Degeneration, while the increasing prevalence of retinal disorders and greater awareness of early diagnosis further support its detection and management. Healthcare professionals use advanced imaging technologies such as Optical Coherence Tomography to detect the condition earlier and with greater accuracy, enabling timely treatment. Physicians widely prescribe targeted therapies like Ranibizumab and Aflibercept to improve patient outcomes and encourage treatment adoption. Additionally, expanding healthcare infrastructure, supportive reimbursement policies, and ongoing research into innovative biologics and gene-based therapies continue to advance clinical management of this vision-threatening disease.
For instance, in February 2026, Sandoz, a global leader in affordable medicines, announced that the FDA has approved an expanded label for Enzeevu (aflibercept-abzv) to cover multiple retinal conditions. The drug was initially approved for treating neovascular (wet) age-related macular degeneration (nAMD).
Early-stage AMD expected to hold largest market share of 42.3% in 2026. Aging populations and increased awareness of routine eye exams drive the onset of early-stage Age-related Macular Degeneration. Clinicians use advanced diagnostic tools such as Optical Coherence Tomography and Fundus Photography to identify retinal changes at the earliest stages. Lifestyle-related risk factors, including smoking, poor diet, and extended screen time, actively contribute to the disease’s development. Expanding ophthalmic services, public health screening initiatives, and ongoing research into preventive treatments and nutritional strategies further empower healthcare providers to detect and manage the condition effectively. For instance, Biogen Inc. and Samsung Bioepis announced the U.S. launch of BYOOVIZ (ranibizumab-nuna), a biosimilar to LUCENTIS.
Hospitals capture the largest market share of 37.7% in 2026. Hospitals expand as rising populations and growing demand for healthcare services increase patient access to medical care. Medical professionals use advanced diagnostic tools, treatment methods, and surgical procedures to improve patient outcomes and attract more patients. Governments and private investors actively support hospital growth by funding infrastructure and specialized services. The increasing prevalence of chronic and acute diseases, along with greater health awareness and skilled healthcare staff, drives hospital development, enhances service delivery, and strengthens the overall healthcare system.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.8% in 2026. In North America, healthcare providers shape the Age-related Macular Degeneration treatment market through a robust healthcare infrastructure and the high prevalence of AMD among older adults. Clinicians actively adopt advanced diagnostic tools and administer intravitreal therapies, while favorable reimbursement policies enhance patient access to anti‑VEGF drugs and other biologics. Pharmaceutical companies and ongoing clinical research introduce innovative treatments, and routine eye screenings combined with increasing patient awareness enable early diagnosis and prompt intervention. For instance, in September 2025, Canadian company MacuMira Medical Devices launched the first Health Canada-approved treatment to improve vision in dry age-related macular degeneration. The non-invasive MacuMira system, now in over 150 clinics, delivers low-dose microcurrent stimulation through closed eyelids in 32-minute sessions.
In the Asia Pacific region, healthcare providers and governments drive the Macular Degeneration Treatment market by expanding healthcare infrastructure and addressing the needs of a growing elderly population. Clinics and hospitals actively increase access to ophthalmic diagnostics and specialized treatments in countries like China, Japan, India, and South Korea. Public health initiatives and rising patient awareness promote early diagnosis, while demand for affordable therapies, wider adoption of tele‑ophthalmology, and investments from global pharmaceutical companies actively expand the market across the region.
In the United States, healthcare providers drive the Age-related Macular Degeneration treatment market by addressing the high prevalence of AMD among older adults and utilizing advanced diagnostics and specialized care. Ophthalmologists actively administer anti‑VEGF therapies and other biologics, supported by strong reimbursement systems and extensive retina specialist networks. Researchers and pharmaceutical companies continue to develop longer-acting treatments and improved delivery methods, while routine screenings and growing patient awareness enable earlier diagnosis and prompt treatment adoption across the country. For instance, in June 2025, Outlook Therapeutics unveiled LYTENAVA (bevacizumab gamma) in Germany and the UK for treating wet age-related macular degeneration (wet AMD).
In India, healthcare providers drive the Age-related Macular Degeneration treatment market by addressing rising AMD incidence and the needs of an aging population. Clinics and hospitals actively expand access to diagnostics and therapies, while local manufacturers develop cost‑effective anti‑VEGF treatments to improve affordability and reach. Public awareness campaigns and screening programs enable clinicians to diagnose AMD earlier, and investments in ophthalmic infrastructure, specialist training, and supportive healthcare initiatives actively promote treatment adoption across both urban areas and emerging regional centers. For instance, in February 2026, Zydus Lifesciences launched Anyra, India’s first indigenously developed Aflibercept 2 mg biosimilar, and partnered with Regeneron Pharmaceuticals and Bayer. Anyra treats wet age-related macular degeneration, macular edema from retinal vein occlusion, diabetic macular edema, diabetic retinopathy, and myopic choroidal neovascularization.
Healthcare providers increasingly adopt cutting‑edge therapies to manage Macular Degeneration, focusing on agents that improve visual outcomes and reduce treatment burdens. Anti‑VEGF therapies remain foundational, while newer biologics, sustained‑release implants, and combination regimens expand clinical options. Ophthalmologists weigh efficacy, safety, and durability when selecting treatment plans. This trend supports personalized care pathways and encourages ongoing research into novel molecules designed to extend treatment intervals and optimize patient convenience.
Clinics and hospitals invest heavily in advanced diagnostic tools to detect Macular Degeneration earlier and more precisely. Optical Coherence Tomography (OCT), wide‑field imaging, and retinal biomarkers help clinicians monitor disease progression and tailor treatment decisions. Improved imaging capabilities support differential diagnosis and enable earlier intervention, which enhances long‑term visual outcomes. Greater accessibility to these technologies across urban and regional centers helps broaden patient reach and strengthens standard of care practices.
Advancements in imaging and diagnostic tools present opportunities for companies to expand offerings that improve early detection and disease monitoring. Technologies like enhanced optical coherence tomography, AI‑driven analysis, and portable screening devices can help clinicians identify macular degeneration sooner and more accurately. This supports tailored treatment regimens, better patient outcomes, and integration into standard eye care workflows. Partnerships with clinics and tele‑ophthalmology platforms further broaden market reach and usability.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 17.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.7% | 2033 Value Projection: | USD 28.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bayer AG, REGENXBIO Inc., Aerie Pharmaceutical Inc., Panoptica, Pfizer Inc., F. Hoffmann-La Roche Ltd., Regeneron Pharmaceutical Inc., Novartis AG, and Bausch Health Companies Inc., among others. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients