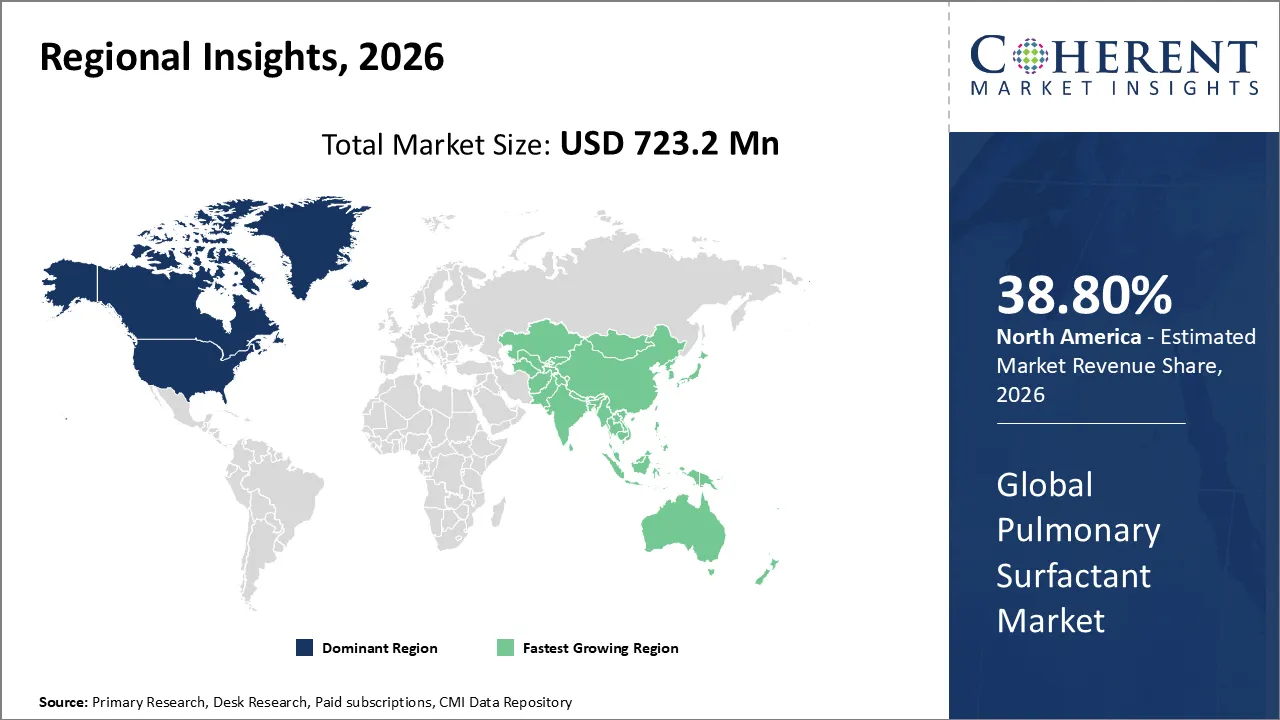
The Pulmonary Surfactant Market is estimated to be valued at USD 723.2 Mn in 2026 and is expected to reach USD 947.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.2% from 2026 to 2033.
The rising incidence of respiratory distress syndrome, particularly among preterm infants, drives the pulmonary surfactant market, while healthcare systems continue to expand neonatal intensive care infrastructure worldwide. Healthcare providers are increasing awareness, enabling early diagnosis, and following strong clinical guidelines to boost surfactant therapy adoption. Clinicians widely use animal-derived products such as Poractant alfa due to their proven efficacy. Additionally, advancements in minimally invasive administration techniques and improving healthcare access in emerging economies further accelerate market growth, while ongoing research enhances treatment outcomes.
|
Current Events |
Description and its impact |
|
Regulatory Approvals and Policy Updates |
|
|
Technological Innovations and Research Advances |
|
|
Epidemiological Trends and Healthcare Demand |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Injectable captures the largest market share of 53.3% in 2026. The pulmonary surfactant market depends on injectable formulations because direct intratracheal administration delivers the drug rapidly and precisely to the lungs. Clinicians use this method to achieve immediate therapeutic effects, which are essential for treating respiratory distress syndrome in preterm infants. Physicians prefer injectables for their reliable clinical performance, alignment with standard NICU protocols, and suitability for minimally invasive techniques. The growth of neonatal intensive care units and the requirement for on-site hospital administration continue to strengthen the widespread use of injectable surfactants globally. For instance, AlveoGene, a company developing inhaled gene therapies for rare respiratory diseases, has launched after securing seed funding from Oxford Science Enterprises, Harrington Discovery Institute at University Hospitals, and Old College Capital, the University of Edinburgh’s venture fund.
Animal Derived Surfactant hold the largest market share of 52.2% in 2026. Animal-derived surfactants drive the pulmonary surfactant market by closely mimicking natural human lung surfactant, which enables effective reduction of alveolar surface tension and supports rapid clinical improvement. Physicians actively prefer these therapies to manage respiratory distress syndrome in preterm infants, relying on strong clinical evidence and established treatment protocols. Clinicians use products such as Poractant alfa for consistent outcomes, while expanding neonatal care facilities and rising institutional deliveries continue to drive demand across global healthcare systems.
Poractant alfa expected to hold largest market share of 42.2% in 2026. Poractant alfa propels the pulmonary surfactant market by rapidly and reliably improving lung function in preterm infants with Respiratory Distress Syndrome. Its higher phospholipid content and optimized dosing enable faster stabilization and reduce the need for repeat treatments. Physicians actively choose it for its robust clinical evidence, compatibility with modern minimally invasive administration techniques, and proven safety. Expanding neonatal care units and rising hospital deliveries continue to drive its widespread adoption across developed and emerging healthcare systems.
Respiratory Distress Syndrome (RDS) acquired the prominent market share of 36.7% in 2026. The prevalence of respiratory distress syndrome drives the pulmonary surfactant market as it primarily affects preterm and low-birth-weight infants who lack sufficient natural surfactant. Healthcare providers actively administer exogenous surfactants to prevent respiratory failure. Increased awareness among clinicians, improvements in prenatal care, and the expansion of neonatal intensive care units enable timely diagnosis and treatment. These combined efforts maintain strong demand for surfactant therapies across both developed and emerging healthcare systems. InnAccel has introduced SAANS, the world’s first portable, infrastructure-independent neonatal CPAP system, designed to support breathing in critically ill neonates with Respiratory Distress Syndrome (RDS) in resource-limited settings and during transport.
Hospital Pharmacies hold the largest market share of 37.7% in 2026. Hospital pharmacies drive the pulmonary surfactant market by providing immediate access to surfactants for critically ill preterm infants. They actively manage controlled storage, maintain cold-chain conditions, and enable safe, timely administration within neonatal intensive care units. Clinicians depend on hospital pharmacies for bulk procurement, efficient distribution, and strict adherence to treatment protocols. The growth of NICUs, rising hospital deliveries, and increased awareness of surfactant therapy continue to strengthen hospital pharmacies’ role in ensuring consistent access and effective management of respiratory distress in newborns.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. Advanced neonatal care infrastructure and widespread adoption of evidence-based treatment protocols drive the North America pulmonary surfactant market. Hospitals increasingly use minimally invasive surfactant administration techniques, enhancing outcomes for preterm infants with respiratory distress. Leading pharmaceutical companies actively ensure product availability, while strong healthcare funding and reimbursement systems support extensive use. Clinicians’ growing awareness, along with expanding NICU capacities and early diagnosis programs, continues to boost demand, establishing North America as a leading region for innovation and adoption in surfactant therapy. For instance, Discovery Laboratories has launched Surfaxin (lucinactant) Intratracheal Suspension to prevent respiratory distress syndrome (RDS) in premature infants at high risk.
Increasing preterm birth rates and growing awareness of neonatal respiratory care drive the expansion of the Asia Pacific pulmonary surfactant market. Governments and healthcare providers actively invest in advanced NICU infrastructure, improving access to surfactant therapy in urban and semi-urban hospitals. Physicians use proven animal-derived and synthetic surfactants to treat respiratory distress in newborns, while minimally invasive administration techniques gain adoption. Rising institutional deliveries, expanding hospital networks, and enhanced training for neonatal care professionals continue to boost demand across the region. For instance, Vygon Group has introduced Surfcath, a specialized catheter for administering surfactant via the LISA method to treat preterm infants with Respiratory Distress Syndrome (RDS).
Hospitals and NICUs drive growth in the United States pulmonary surfactant market by adopting advanced neonatal care practices and evidence-based treatment protocols. Clinicians increasingly implement minimally invasive surfactant administration techniques to enhance outcomes for preterm infants with respiratory distress. Leading pharmaceutical companies actively maintain consistent product availability, while robust healthcare funding and reimbursement policies enable broad access. Expanding NICU capacities, early diagnosis initiatives, and rising awareness among healthcare professionals continue to boost adoption, establishing the U.S. as a leading market for innovation and effective neonatal respiratory care.
Rising preterm births and growing awareness of neonatal respiratory care drive the expansion of the China pulmonary surfactant market. Hospitals and neonatal intensive care units actively invest in advanced infrastructure and staff training, improving access to both animal-derived and synthetic surfactants. Clinicians increasingly apply minimally invasive administration techniques to enhance outcomes for newborns with respiratory distress. Government initiatives, increasing institutional deliveries, and expanding hospital networks continue to boost surfactant adoption, establishing China as a rapidly developing market for neonatal respiratory therapies and advanced neonatal care.
The increasing incidence of preterm and low-birth-weight infants globally fuels demand for pulmonary surfactants. Hospitals are expanding neonatal intensive care units to manage respiratory complications effectively. Early diagnosis and timely surfactant administration are becoming standard practice, prompting hospitals and clinicians to adopt both animal-derived and synthetic surfactants to reduce mortality and improve neonatal outcomes across diverse healthcare settings.
Clinicians are increasingly adopting minimally invasive surfactant delivery techniques such as LISA and MIST. These methods reduce the need for mechanical ventilation, lower complication risks, and enhance patient outcomes. The trend encourages pharmaceutical companies to develop formulations compatible with these techniques, allowing hospitals to integrate surfactant therapy seamlessly into modern NICU protocols while improving safety and treatment efficiency.
There is a strong opportunity to innovate with less invasive administration techniques, such as LISA and MIST, which reduce mechanical ventilation reliance. Companies can design surfactant formulations compatible with these approaches, improving patient safety, clinical outcomes, and adoption rates. Hospitals increasingly prefer these methods for fragile preterm infants, providing a strategic avenue for product differentiation and technological advancement in neonatal care.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 723.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 947.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Boehinger Ingelheim, AbbVie Inc, Chiesi Farmaceutici, ONY Biotech Inc., Lyomark Pharma, Abbott, Aviva Systems Biology Corporation, Windtree Therapeutics, Inc, Tekzima (Noargen), Biomatik, Nanjing Norris Pharm Technology, Reddot Biotech |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients