Global Deutetrabenazine Market Size and Forecast – 2026 to 2033
The global deutetrabenazine market is estimated to be valued at USD 1.08 Bn in 2026 and is expected to reach USD 1.88 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.24% from 2026 to 2033. The market's steady growth can be attributed to the increasing prevalence of neurological disorders and advancements in drug formulation.
Moreover, patients are becoming more informed about available treatment options, disease management approaches, and the benefits of targeted therapies such as deutetrabenazine. Together, these factors are expected to offer greater market penetration and increased revenue over the coming years.
Key Takeaways of the Global Deutetrabenazine Market
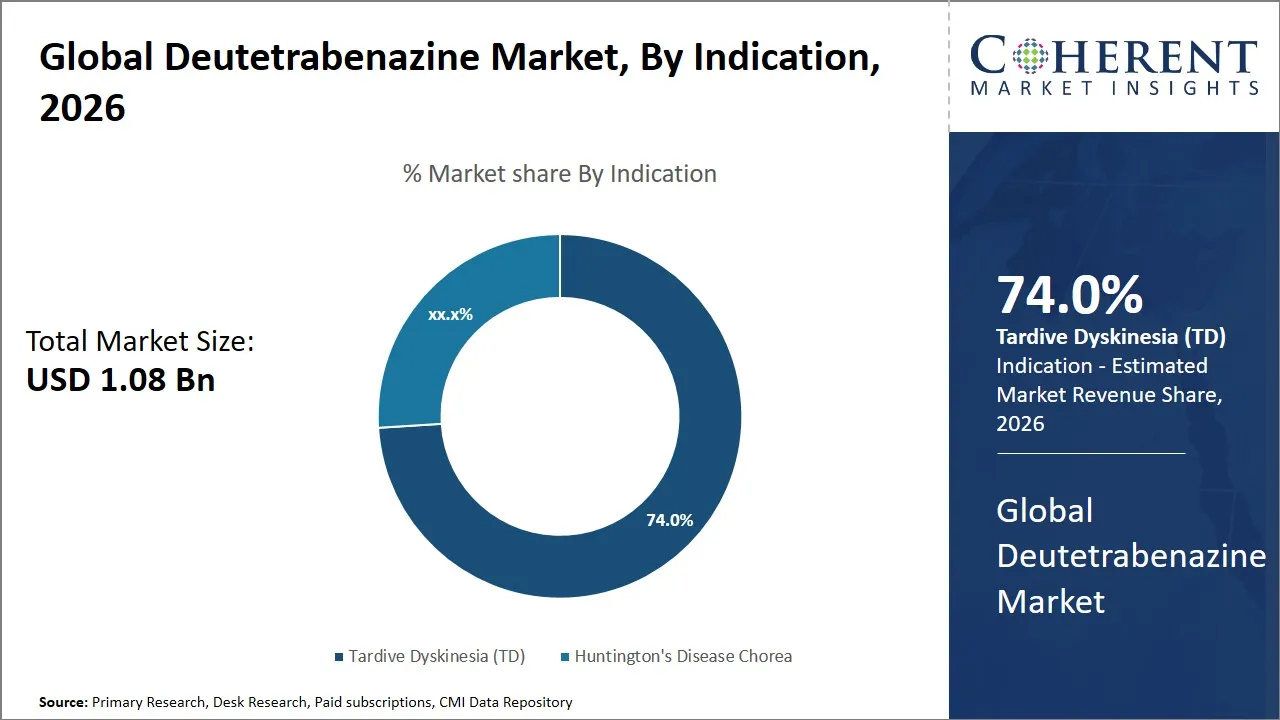
- The Tardive Dyskinesia (TD) segment is expected to dominate the global deutetrabenazine market by indication, accounting for approximately 74.0% of the total market share in 2026.
- The Immediate-Release (IR) segment is expected to dominate the global deutetrabenazine market by formulation type, accounting for approximately 68.0% of the total market share in 2026.
- The hospitals segment is expected to dominate the global deutetrabenazine market by end user, accounting for approximately 57.0% of the total market share in 2026.
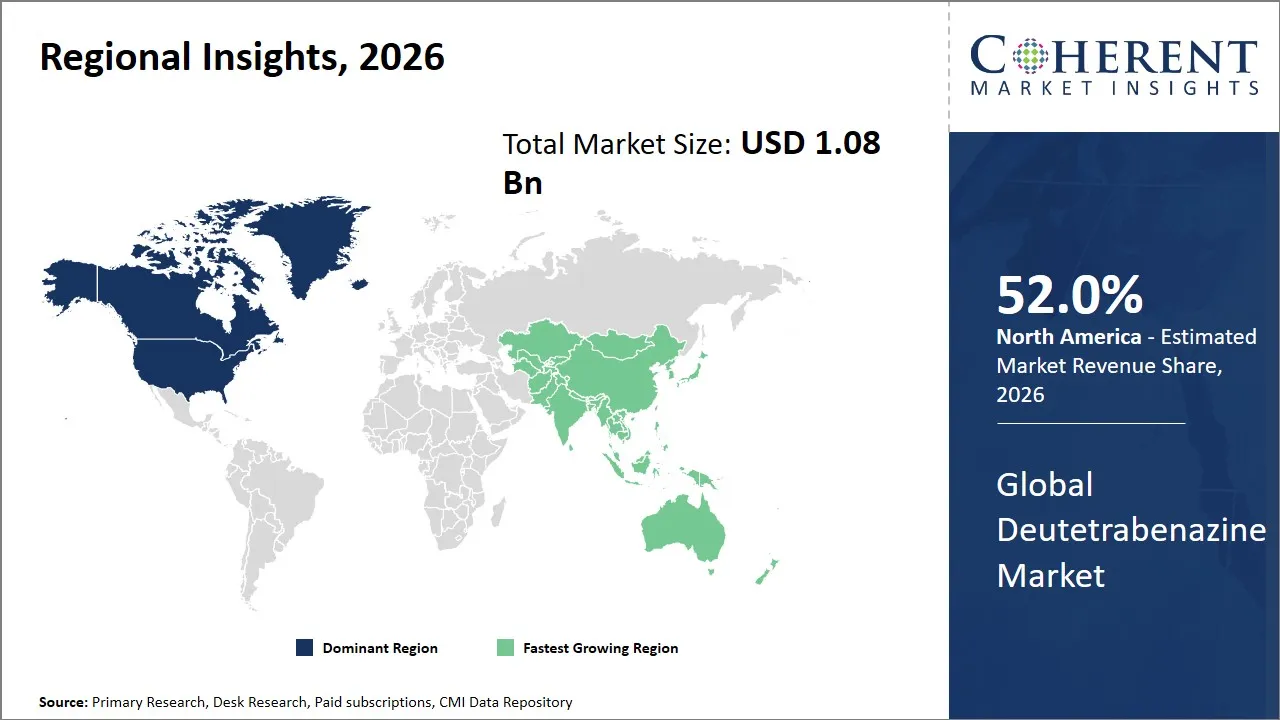
- North America is expected to dominate the global deutetrabenazine market in 2026, accounting for approximately 52.0% of the total market share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 12.0% in 2026.
Market Overview
- Deutetrabenazine is a vesicular monoamine transporter 2 inhibitor and is generally used for the treatment of movement disorders such as chorea associated with Huntington’s disease and tardive dyskinesia. The medication helps control movements and improve the quality of life for patients by regulating the release of dopamine in the brain. The better pharmacokinetics of deutetrabenazine compared to previous treatments have made the drug more tolerable for patients.
- The global deutetrabenazine market is growing steadily due to an increasing number of cases of neurological disorder, movement disorders, and rising awareness of diseases such as tardive dyskinesia. The increasing use of antipsychotic drugs is also a major factor for the growing deutetrabenazine market because these medications are a major cause of tardive dyskinesia.
- Furthermore, positive clinical trial results, a better safety profile, and ongoing research into new uses are also contributing to the growth of the market. Effective commercialization activities, particularly in developed countries, as well as a focus on Central Nervous System (CNS) diseases, are also contributing to the growth of the market. However, high costs of treatment, limited patient pool, and competitive drugs may act as hurdles in the growth of the market.
Currents Events and Their Impact
|
Current Events |
Description and its Impact |
|
Expansion of VMAT2 Inhibitor Labeling in Key Markets |
|
|
Advancements in Digital Monitoring for Movement Disorders |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Tardive Dyskinesia (TD) Segment Dominate the Global Deutetrabenazine Market in 2026?
The Tardive Dyskinesia (TD) segment is anticipated to hold a market share of 74.0% in 2026, primarily due increase in awareness among the population suffering from this condition. Tardive Dyskinesia (TD) is usually caused by the prolonged use of antipsychotic drugs. With the condition becoming more recognized as a serious health condition, more emphasis is being given to treating the condition effectively. Deutetrabenazine has come up as a major drug in treating Tardive Dyskinesia (TD) due to its proven efficacy in managing involuntary movements.
The segment's growth can be further justified by the advancements in clinical knowledge and diagnostic techniques, which have improved the detection of Tardive Dyskinesia (TD). The limitations and adverse effects of other treatment options have helped to strengthen the position of deutetrabenazine in the market, given its safety and tolerability profile. The changing treatment guidelines and the emphasis placed on managing patients with Tardive Dyskinesia (TD) have helped to increase the prescription rate of the drug. The widespread use of antipsychotic medications among different patient groups has helped to increase the patient population at risk, which in turn will help to maintain the market demand for deutetrabenazine.
Why Does the Immediate-Release (IR) Segment Dominate the Global Deutetrabenazine Market in 2026?
The Immediate-Release (IR) segment is projected to hold a market share of 68.0% in 2026, mainly because of the quick symptom-relieving properties that are often required in the management of disorders such as tardive dyskinesia and the chorea associated with Huntington’s disease. The quick action is particularly advantageous in the management of variable or severe movement disorders, as the symptoms can be quickly stabilized during the early stages of treatment.
The other important factor that has led to the dominance of IR formulations in the market is their flexibility in dose titration, which makes it possible for clinicians to adjust their therapeutic regimens based on individual patient requirements while minimizing adverse effects such as sedation and parkinsonism. Finally, patients and clinicians may choose IR formulations due to their flexibility in patient management and control during the stabilization phase of therapy, despite the convenience of using extended-release formulations. The precision, flexibility, and response associated with IR formulations make them crucial in maintaining their dominance in the market despite the convenience of using extended-release formulations.
For instance, according to the prescribing information from the U.S. Food and Drug Administration, deutetrabenazine Immediate-Release (IR) tablets are started at a dose of 6 mg twice a day. The dose is then adjusted weekly, depending on how well the patient responds and how well they tolerate the medication. This highlights the need for flexible and personalized dosing. This structured titration process shows the clinical benefits of IR formulations. It allows for precise dose adjustments and close monitoring, which is why they are often preferred by doctors and are the most common type of formulation used.
The Hospitals Segment Dominates the Global Deutetrabenazine Market
Hospitals segment is projected to hold a market share of 57.0% in 2026, due to their role as a center for specialized neurological care and management of patients with neurological diseases. Patients with neurological diseases such as tardive dyskinesia and Huntington’s disease-associated chorea require proper management under a neurologist’s supervision and proper diagnostic tests along with continuous monitoring of response and side effects of drugs. A hospital setting is best suited for managing patients with neurological diseases as it is a controlled environment for starting a drug like deutetrabenazine, which has a dose-dependent safety profile.
Moreover, modern diagnostic tools are also being used by hospitals to aid in the diagnosis of diseases and develop personalized plans for patients. The availability of skilled and trained health professionals who are able to handle complex drug regimens and manage adverse reactions also increases the likelihood of selecting a hospital for deutetrabenazine use. This comprehensive care environment also increases the likelihood of deutetrabenazine being adopted by a high number of hospitals, thereby maintaining the dominance of the end users.
Regulatory Landscape & Clinical Practice Guidelines Influencing Deutetrabenazine Adoption
|
Region |
Key Regulatory Authority |
Approval & Regulatory Status |
Clinical Practice Guidelines |
Impact on Deutetrabenazine Adoption |
|
North America |
U.S. Food and Drug Administration |
Approved for tardive dyskinesia and Huntington’s disease chorea; continuous label expansions and post-marketing surveillance |
American Psychiatric Association and American Academy of Neurology recommend VMAT2 inhibitors as first-line therapy for TD |
Strong regulatory backing and guideline-driven prescribing significantly boost adoption and market dominance |
|
Europe |
European Medicines Agency |
Limited/gradual approvals and market access variations across EU countries; regulatory pathways evolving |
European clinical updates increasingly recognize VMAT2 inhibitors as effective treatments with high evidence levels |
Slower uptake compared to U.S., but improving with regulatory harmonization and guideline updates |
|
Asia Pacific |
Pharmaceuticals and Medical Devices Agency |
Selective approvals and country-specific regulatory pathways; access varies widely |
Growing adoption of international guidelines; increasing inclusion of VMAT2 inhibitors in psychiatric care protocols |
Moderate adoption driven by improving diagnostics, awareness, and healthcare infrastructure |
|
Latin America |
National Health Surveillance Agency |
Gradual approvals with pricing and reimbursement constraints |
Limited formal TD-specific guidelines; reliance on U.S./European clinical practices |
Adoption remains constrained but improving with expanding access and physician awareness |
|
Middle East & Africa |
Saudi Food and Drug Authority |
Emerging approvals; regulatory frameworks still developing |
Limited region-specific guidelines; dependence on global recommendations (APA/AAN) |
Lower adoption due to access and awareness barriers, but potential growth with healthcare investments |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Deutetrabenazine Market Analysis and Trends
The North America region is projected to lead the market with a 52.0% share in 2026, attributed to the highly developed infrastructure for healthcare and a strong presence of pharmaceutical companies and government support for innovation in the field of neurological disorders and movement disorders. The U.S. is particularly favored due to a number of clinical trials and fast-tracked approval of drugs by government agencies such as the FDA.
The presence of a strong reimbursement system and high levels of awareness regarding diseases such as Huntington’s and tardive dyskinesia also drive the demand for deutetrabenazine. The presence of Teva Pharmaceutical Industries Ltd., which developed deutetrabenazine and markets it by the brand name Austedo, is a significant factor for the dominance of the global deutetrabenazine market in the region due to its active engagement in research and commercialization of the product. The presence of a number of specialist clinics such as Mayo Clinic and Cleveland Clinic, among others, helps in the high adoption of deutetrabenazine-based drugs in the country.
For instance, according to the Centers for Medicare & Medicaid Services, Medicare Part D provides outpatient prescription drug coverage through plan-specific formularies, which determine the inclusion and reimbursement conditions for therapies such as deutetrabenazine. These formularies are developed based on clinical value and cost considerations and may include utilization management tools such as prior authorization. This confirms that reimbursement for neurological drugs like deutetrabenazine in North America is structured through Medicare Part D formulary systems, which play a critical role in determining patient access, pricing, and overall market adoption.
Asia Pacific Deutetrabenazine Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 12.0% in 2026, owing to the increased investment in healthcare, a growing patient population, and improving diagnostic capabilities for diseases. The prevalence of neurological disorders, combined with increased awareness and investment in healthcare in emerging nations such as China and India, has been driving the growth of the deutetrabenazine market. Government initiatives to expand access to advanced treatments, along with improvements to existing infrastructure, have been instrumental in driving growth in this sector.
The expansion of multinational corporations like Teva, alongside collaborations with local entities, has also boosted market penetration, trade liberalization, along with increased regulatory harmonization in countries such as South Korea and Japan, has also helped improve access to breakthrough treatments, making Asia Pacific an emerging market for deutetrabenazine.
Deutetrabenazine Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Deutetrabenazine Market?
The U.S. remains at the forefront in the deutetrabenazine market due to a conducive R&D environment, high-end healthcare facilities, and a smooth regulatory environment. The significant investments made by Teva Pharmaceutical in clinical trials and educational activities help in improving market reach. The strong prescriber base for treating movement disorders and other neurological conditions also contributes to the growth of the deutetrabenazine market. Furthermore, the availability of insurance coverage in the U.S. makes it a market powerhouse.
For instance, in April 2024, Teva Pharmaceutical Industries unveiled real-world evidence at the American Academy of Neurology (AAN) Annual Meeting, which showed the strong efficacy and patient satisfaction with the treatment of Huntington’s disease chorea using the structured 4-week titration approach with deutetrabenazine. This shows the improvement in the treatment optimization and adherence to the treatment in the U.S. clinical setting. This increases the confidence among the doctors to prescribe the drug widely in the U.S.
How is Germany Helping in the Growth of the Deutetrabenazine Market?
The deutetrabenazine market in Germany is positively impacted due to factors such as a strong level of health spending and well-developed infrastructure for addressing neurological disorders. Government-backed initiatives to ensure access to such treatments for patients also contribute to making the market more credible. The stringent norms set by the European Medicines Agency (EMA) for approving new treatments also contribute in making the deutetrabenazine market in Germany more credible. Key players such as Teva are entering into partnerships with healthcare providers to ensure effective utilization of such treatments. Personalized medicine is also an important aspect of Germany, which contribute in making the best utilization of deutetrabenazine treatments.
Key Drivers for the Growth of the China Deutetrabenazine Market
The market for deutetrabenazine in China is constantly changing due to the growing awareness of the condition, the expansion of the healthcare infrastructure, as well as government initiatives to ensure that people have access to innovative medicine. The progressive approach taken by the Chinese National Medical Products Administration in approving medicine has encouraged multinational companies to forge alliances with local companies, hence expediting the process.
The high rate of urbanization in the country, as well as the growth in the population of the middle class, also contribute to the demand. The diagnostic facilities available, as well as the number of neurologists in the country, also help the adoption rate for deutetrabenazine.
Japan Deutetrabenazine Market Trends
The deutetrabenazine market in Japan represents a rising trend, driven by an increasing population and an increasing incidence of neurological diseases. The government's focus on healthcare innovation and accelerating drug approval by the Pharmaceuticals and Medical Devices Agency (PMDA) allows rapid market entry for the drug.
The major companies, both domestic and foreign, are participating in the development of this market through strategic alliances. Additionally, Japan has an established healthcare infrastructure, which help in patient access and compliance to drug therapy.
Epidemiological Burden and Patient Population Analysis of Target Indications Supporting Deutetrabenazine Market Expansion
|
Region |
Huntington’s Disease Prevalence |
Tardive Dyskinesia (TD) Prevalence |
Key Epidemiological Insights |
Impact on Deutetrabenazine Market |
|
North America |
~5–10 per 100,000 population |
~20–30% among patients on long-term antipsychotics |
High diagnosis rates and strong awareness; well-established screening practices |
Large diagnosed patient pool drives strong demand and high treatment adoption |
|
Europe |
~5–8 per 100,000 population |
~15–25% among antipsychotic users |
Increasing recognition of TD and improved psychiatric care infrastructure |
Moderate to high adoption supported by evolving clinical guidelines |
|
Asia Pacific |
~1–4 per 100,000 population |
~10–20% among antipsychotic users |
Underdiagnosis remains a challenge; growing awareness and improving diagnostics |
Emerging growth potential with expanding patient identification |
|
Latin America |
~3–6 per 100,000 population |
~10–20% among antipsychotic users |
Limited epidemiological tracking but rising psychiatric treatment rates |
Gradual market expansion driven by increasing disease recognition |
|
Middle East & Africa |
~1–3 per 100,000 population |
~5–15% among antipsychotic users |
Low awareness and diagnostic gaps persist |
Untapped market with long-term growth opportunities |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In May 2024, Teva Pharmaceutical Industries received U.S. FDA approval for AUSTEDO XR (deutetrabenazine) extended-release tablets, which provide a once-daily dosing schedule at clinically effective levels. This development improves patient adherence to the treatment plan, also solidifies the company's standing in the deutetrabenazine market by making it more convenient for patients and extending the product's lifecycle.
- In February 2024, Teva Pharmaceutical Industries and Jiangsu Nhwa Pharmaceutical announced a collaboration. The goal is to make AUSTEDO (deutetrabenazine) more accessible to patients in China. This partnership is all about boosting sales and making the drug easier to get in a rapidly growing market. It's a move designed to support the long-term growth of the deutetrabenazine market across the region.
Top Strategies Followed by Global Deutetrabenazine Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders focus on continuous R&D and lifecycle management to enhance formulations, safety, and dosing flexibility. They emphasize real-world evidence, label expansions, and strong physician engagement. Further, they leverage global distribution networks and strategic partnerships to expand market reach and sustain dominance. |
In October 2024, Teva Pharmaceutical Industries announced continued investment in lifecycle management of AUSTEDO, focusing on expanding physician outreach and strengthening its global neurology portfolio through ongoing clinical and post-marketing studies |
|
Mid-Level Players |
Mid-level players adopt cost-efficient manufacturing and value-based pricing strategies to compete in price-sensitive markets. They are engaged in licensing and contract manufacturing partnerships to enhance scalability and reduce time-to-market. Further, they focus on expanding regional presence while maintaining regulatory compliance and therapeutic quality. |
In July 2024, Lupin Limited expanded its specialty drug manufacturing capabilities through partnerships to strengthen its CNS portfolio and improve access to affordable neurological therapy |
|
Small-Scale Players |
Small-scale players focus on niche innovation and early-stage research targeting unmet needs in movement disorders. They collaborate with academic institutions and research bodies to develop novel mechanisms and delivery systems. They prioritize regional market entry and differentiation through specialized, technology-driven solutions. |
In May 2024, Addex Therapeutics continued advancing its early-stage pipeline targeting movement disorders through collaborations and research programs focused on novel allosteric modulators for neurological conditions. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Deutetrabenazine Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.08 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.24% | 2033 Value Projection: | USD 1.88 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
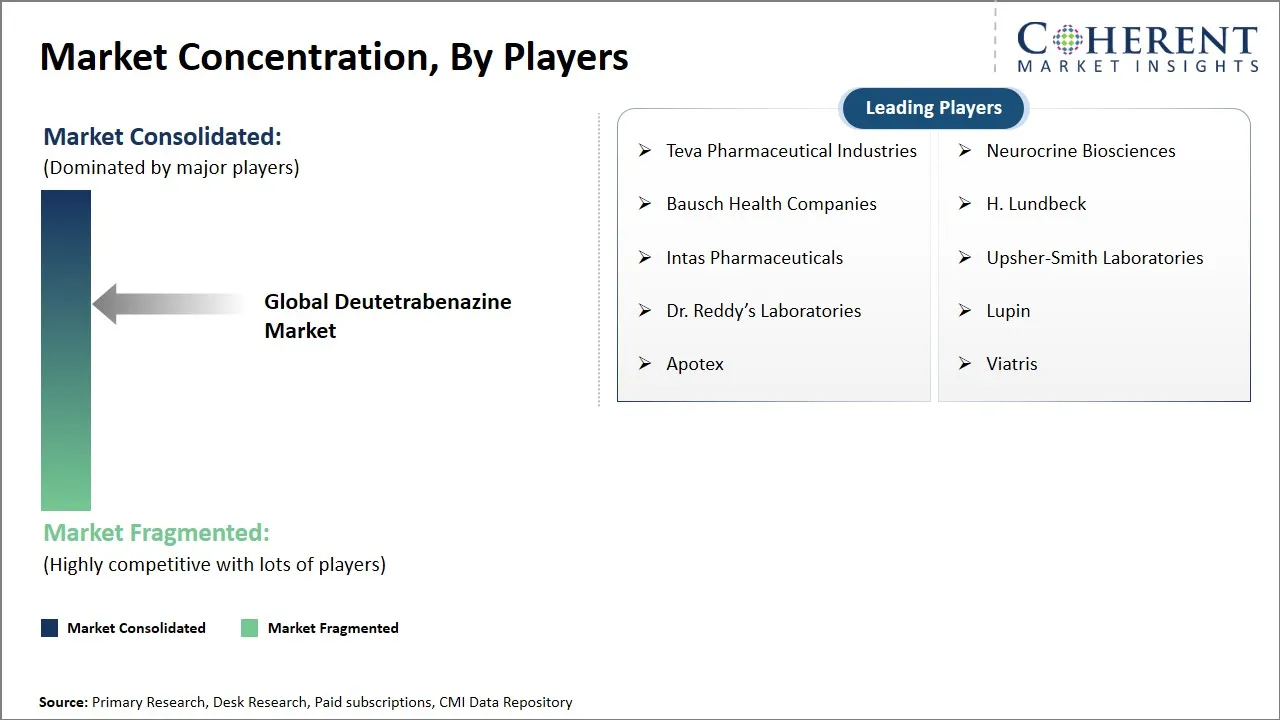
Teva Pharmaceutical Industries, Neurocrine Biosciences, Bausch Health Companies, H. Lundbeck, Intas Pharmaceuticals, Upsher-Smith Laboratories, Dr. Reddy’s Laboratories, Lupin, Apotex, and Viatris |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Deutetrabenazine Market Dynamics
To learn more about this report, Request Free Sample
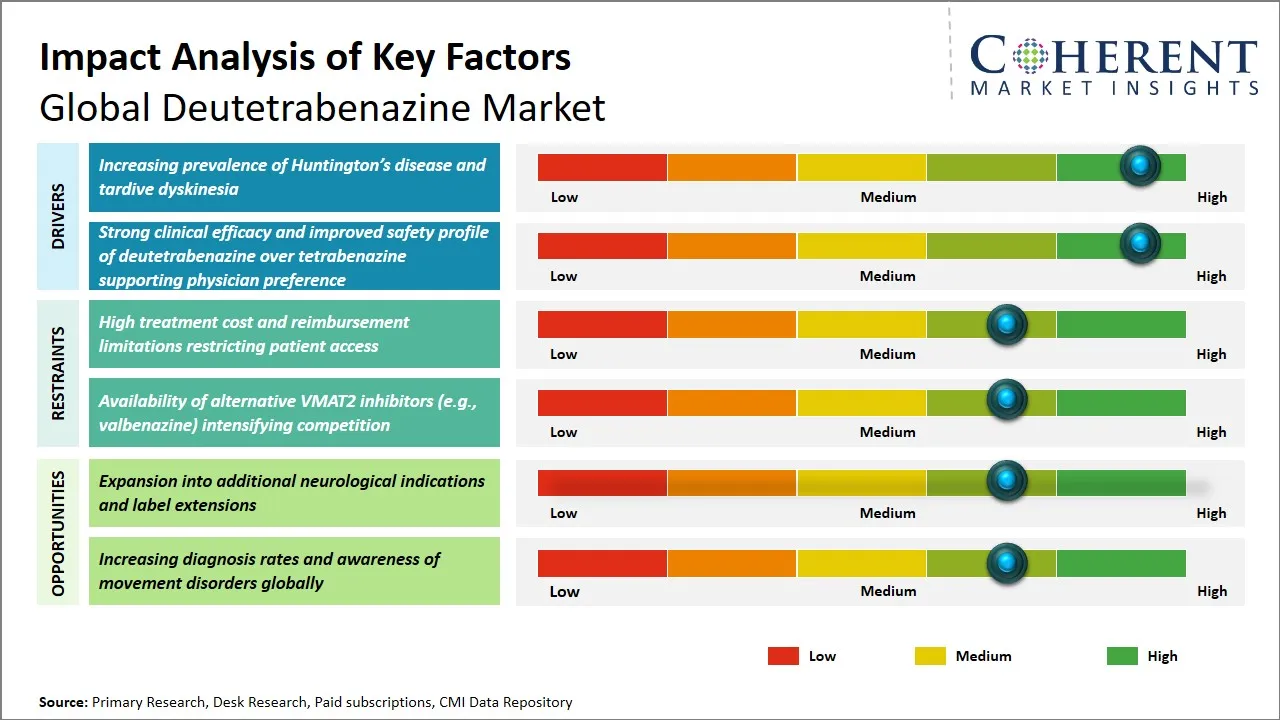
Global Deutetrabenazine Market Driver – Rising Prevalence of Huntington’s Disease and Tardive Dyskinesia
The rising incidence rate of neurodegenerative diseases like Huntington’s disease and movement disorders like tardive dyskinesia has been one of the major contributing factors to the increasing demand for deutetrabenazine. Huntington’s disease, which causes motor, cognitive, and psychiatric problems, needs therapeutic strategies to control involuntary movements. This increased the need for deutetrabenazine. Tardive dyskinesia, a condition often caused by long-term use of antipsychotic drugs, is characterized by involuntary and repetitive movements. This disorder can have a severe impact on the quality of life.
The rising incidence rate of these diseases, which increases the patient pool, has a direct impact on the demand for therapeutic strategies to control these diseases. The ability of deutetrabenazine to modulate the dopamine system increases the favorable safety and efficacy profile compared to other therapeutic strategies, which increases the demand for the drug. Further, increasing focus on personalized medicine and chronic disease management can be related to the increasing demand for deutetrabenazine, which results in the direct impact of the incidence rate of these diseases.
For instance, according to a systematic review and meta-analysis carried out by the National Institutes of Health (NIH), the prevalence of Huntington’s disease worldwide is approximately 4.88 cases per 100,000 population, as indicated by epidemiological studies carried out in different geographical locations.
Likewise, a global meta-analysis published in SAGE Journals reveals that tardive dyskinesia affects 25.3% of persons undergoing antipsychotic treatment globally, thus emphasizing the impact of tardive dyskinesia on persons undergoing such a treatment.
Global Deutetrabenazine Market Opportunity - Expansion into Additional Neurological Indications and Label Extensions
The global deutetrabenazine market offers a significant opportunity for growth in terms of expanding into other neurological disorders and label expansions. Initially approved for use in Huntington’s disease-associated chorea and tardive dyskinesia, the mechanism of action of deutetrabenazine involves the inhibition of the vesicular monoamine transporter 2 (VMAT2), which makes it a potential therapeutic option for a number of other hyperkinetic disorders.
Current research is exploring its potential for use in disorders such as Tourette syndrome and Parkinson’s disease-related dyskinesia, which will significantly broaden the potential population. Another opportunity for growth is expanding its use into the pediatric and adolescent population and throughout the spectrum of disease progression.
The approval for these expanded indications would not only increase the clinical utility of the drug but would also help in increasing the market base of the drug by means of both on-label and off-label usage. This would provide opportunities to pharmaceutical companies to strengthen their market base by strategic clinical development and usage. This would be in line with the increasing incidence of neurological disorders worldwide and the need for long-term, well-tolerated agents, which would eventually help in increasing the market base and revenues.
For instance, emerging clinical research published in National Library of Medicine in November 2016, demonstrated that deutetrabenazine achieved a 37.6% reduction in tic severity scores in patients with Tourette syndrome over an 8-week treatment period, highlighting its potential applicability in additional hyperkinetic movement disorders.
Analyst Opinion (Expert Opinion)
- The market for deutetrabenazine is a niche market with high growth potential due to its clinical efficacy in treating tardive dyskinesia and Huntington’s disease chorea, mainly in developed countries.
- The market has growth prospects due to rising diagnosis rates of tardive dyskinesia and Huntington’s chorea, along with rising rates of antipsychotic use, thus increasing the patient population for deutetrabenazine.
- The market is challenged by the small patient population size and high drug costs, along with competition from other VMAT2 inhibitors. The future growth of the market will come from innovation and access, not from volume growth.
Market Segmentation
- Indication Insights (Revenue, USD Bn, 2021 - 2033)
- Tardive Dyskinesia (TD)
- Huntington’s Disease Chorea
- Formulation Type Insights (Revenue, USD Bn, 2021 - 2033)
- Immediate-Release (IR)
- Extended-Release (XR)
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Specialty Clinics
- Homecare Settings
- Others
- Distribution Channel Insights (Revenue, USD Bn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Specialty Pharmacies
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Teva Pharmaceutical Industries
- Neurocrine Biosciences
- Bausch Health Companies
- Lundbeck
- Intas Pharmaceuticals
- Upsher-Smith Laboratories
- Reddy’s Laboratories
- Lupin
- Apotex
- Viatris
Sources
Primary Research Interviews
Industry Stakeholders
- Neurology Drug Manufacturers (VMAT2 inhibitors)
- Specialty Pharmaceutical Companies (CNS-focused portfolios)
- Contract Manufacturing Organizations (CMOs) – neuro APIs
- Active Pharmaceutical Ingredient (API) Suppliers (dopamine modulators)
- Regulatory Affairs Experts (neurology drug approvals)
- Hospital Procurement Specialists (neurology & psychiatry departments
End Users
- Hospitals & Neurology Centers
- Movement Disorder Clinics
- Psychiatric Care Centers
- Specialty Neurologists & Psychiatrists
Government and International Databases
- World Health Organization – Neurological disorder burden & epidemiology
- U.S. Food and Drug Administration – Drug approvals, safety labels
- European Medicines Agency – Regulatory approvals and orphan drug status
- National Institutes of Health – Clinical and epidemiological studies
- ClinicalTrials.gov – Ongoing and completed deutetrabenazine trials
Trade Publications
- Pharmaceutical Technology
- Fierce Pharma
- BioPharma Dive
- NeurologyLive
- Pharmaceutical Executive
Academic Journals
- Neurology and Therapy
- Movement Disorders Journal
- Journal of Neurology, Neurosurgery & Psychiatry
- The Lancet Neurology
- CNS Drugs
Reputable Newspapers
- The Wall Street Journal – Pharmaceutical industry movements, mergers, and strategic developments
- Financial Times – Global healthcare investments and regulatory landscape
Industry Associations
- International Parkinson and Movement Disorder Society (MDS)
- American Academy of Neurology (AAN)
- Huntington’s Disease Society (HDSA)
- European Academy of Neurology (EAN)
Public Domain Resources
- NIH PMC– open-access clinical research
- WHO Global Health Observatory
- FDA Adverse Event Reporting System (FAERS)
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients