Middle East and Saudi Arabia Pharmaceutical Drugs Market Size and Forecast – 2026 To 2033
The Middle East and Saudi Arabia pharmaceutical drugs market is expected to grow from USD 37,475.0 Mn in 2026 to USD 59,776.8 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.9%. The Middle East and Saudi Arabia pharmaceutical drugs market is experiencing a significant growth attributed to rising government healthcare spending.
According to Ministry of Finance Saudi Arabia, USD 57 billion (SAR 214 billion) were allocated to health and social development in 2024, representing one of the largest budget allocations, significantly improving healthcare access and pharmaceutical demand.
(Source: Ministry of Finance Saudi Arabia)
Key Takeaways of the Middle East and Saudi Arabia Pharmaceutical Drugs Market
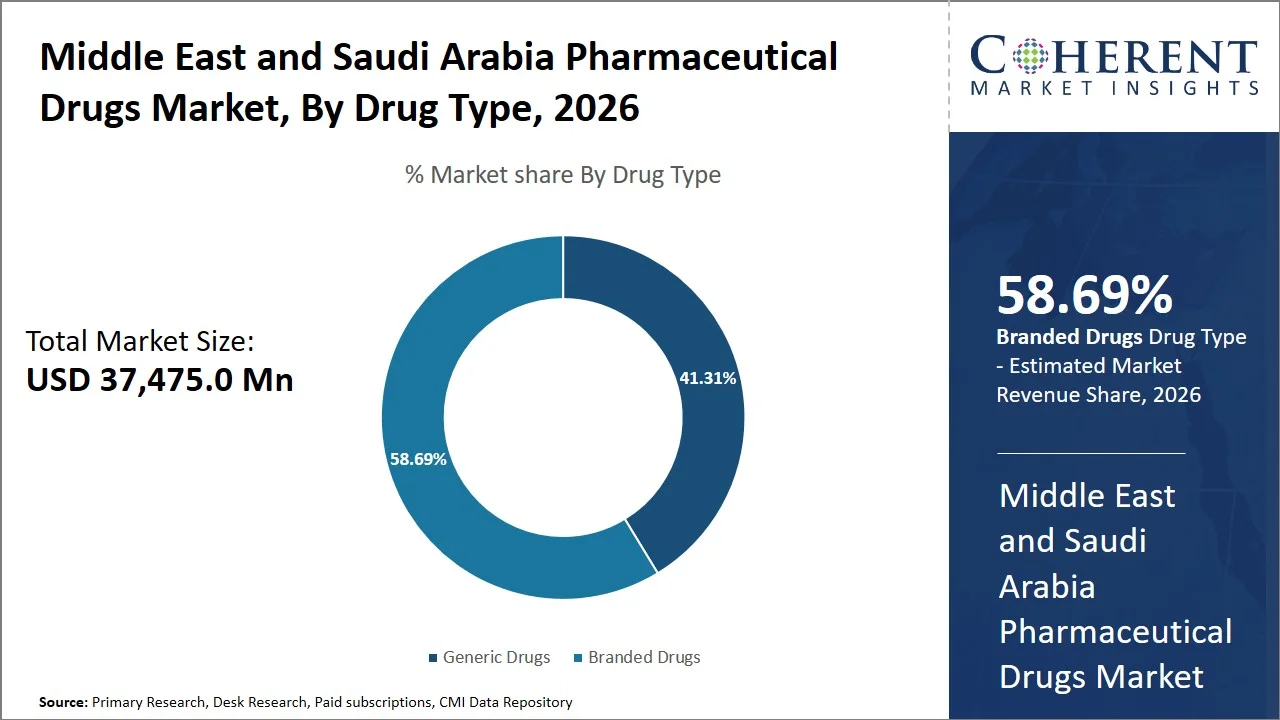
- Branded drugs are projected to hold a market share of 58.69% of the Middle East and Saudi Arabia pharmaceutical drugs in 2026, making it the dominant drug type segment, influenced by high physician preference for patented medicines and quality-focused regulations. In Saudi Arabia, brand medicines have supremacy because of stringent regulations on bioequivalence and substitutability enforced by the Saudi Food and Drug Authority (e.g., SFDA Bioequivalence Requirements for Generic Drug Products and restrictions on automatic generic substitution without proven therapeutic equivalence).
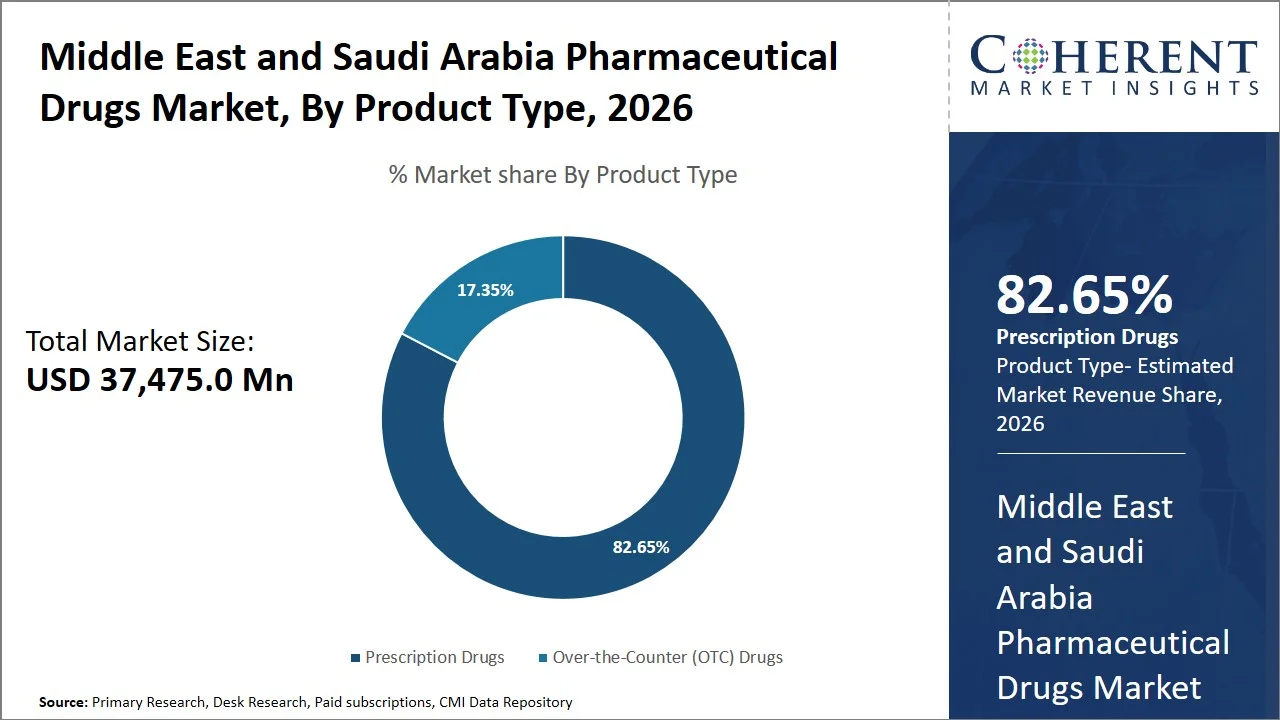
- Prescription drugs are projected to hold a market share of 82.65% of the Middle East and Saudi Arabia pharmaceutical drugs market in 2026, making it the dominant product type segment, supported by tightly regulated dispensing systems across GCC countries. Governments such as the Ministry of Health Saudi Arabia mandate prescription-only access for most therapeutic classes, especially chronic and specialty drugs.
- The antidiabetics segment is projected to hold a market share of 19.34% of the Middle East and Saudi Arabia pharmaceutical drugs market in 2026, making it the dominant drug class segment, due to the region’s high diabetes burden. According to the International Diabetes Federation, the Middle East region has a diabetes prevalence of ~16.2%, among the highest globally, significantly driving demand for antidiabetic drugs. (Source: International Diabetes Federation)
- Saudi Arabia market maintains dominance with an expected share of 29.26%in 2026, supported by high healthcare expenditure and government-led initiatives like Vision 2030, which prioritizes healthcare infrastructure and pharmaceutical localization.
- The UAE pharmaceutical drugs market is expected to exhibit the fastest growth with an estimated contribution of 08% share in 2026, owing to the advanced healthcare infrastructure and strong medical tourism inflows. The Dubai Health Authority reports consistent growth in healthcare utilization and pharmaceutical demand due to rising expatriate population and chronic disease prevalence. (Source: Dubai Health Authority)
- Rising Adoption of GLP-1 Receptor Agonists: The use of advanced medical practices such as the administration of Glucagon-Like Peptide-1 (GLP-1) receptor agonists has improved glycemic control as well as promoted weight loss, leading to higher consumption of high-quality products by city residents of Saudi Arabia and the other Gulf Cooperation Council states.
- Expansion of Digital Diabetes Management Solutions: The successful merging of glucose monitoring systems with mobile applications has made it easier to track and provide treatment to patients, thereby paving the way for pharmaceutical companies to offer additional services along with their diabetes drugs.
Why Do Branded Drugs Dominate the Middle East and Saudi Arabia Pharmaceutical Drugs Market?
Branded drugs are projected to hold a market share of 58.69% in 2026, primarily attributed to effective prescribing practices by doctors, patient preference, and perceived advantages with regards to quality. In spite of government efforts (e.g., implementation of generic substitution policies under the Ministry of Health, Council of Health Insurance (CHI) Circular No. 289 promoting generic utilization) to encourage the use of generics, patient preference is skewed towards branded drugs. For instance, research published in the Journal of Applied Medical Sciences indicated that about 58% of the respondents involved in study prefer branded drugs over generics due to the high efficacy levels and safety measures. (Source: Journal of Applied Medical Sciences)
Why are Prescription Drugs the Most Preferred Product Type in Middle East and Saudi Arabia Pharmaceutical Drugs Market?
To learn more about this report, Request Free Sample
Prescription drugs are projected to hold a market share of 82.65% in 2026, owing to their dominant role in regulated, hospital-centric treatment protocols and the region's stringent drug classification regulations (e.g., Saudi Food and Drug Authority prescription-only medicine classification guidelines, Ministry of Health Saudi Arabia hospital formulary and prescribing regulations, and Dubai Health Authority controlled medication dispensing policies). The Saudi Ministry of Health (MOH) indicates that public hospitals and primary healthcare centers account for more than 60% of healthcare service delivery where treatment is largely physician-directed and reliant on prescriptions.
(Source: National Library of Medicine)
Furthermore, the Saudi Food and Drug Authority (SFDA) designates most therapeutic drugs—antibiotics, cardiovascular medications, and specialty treatments, as prescription drugs. This limits over-the-counter access, a measure designed to safeguard patients. The combination of robust regulatory oversight and a healthcare model focused on hospitals further cements the market's dependence on prescription medications, solidifying their leading position.
Antidiabetics Dominates the Middle East and Saudi Arabia Pharmaceutical Drugs Market
The antidiabetics segment is projected to hold a market share of 19.34% in 2026, owing to the very high prevalence of diabetes in the region. According to International Diabetes Federation (IDF), 589 million people have diabetes in the world and 85 million people in the MENA Region; by 2050, this will rise to 163 million (Source: International Diabetes Federation (IDF)). On a wider scale, the prevalence rate of diabetes in the MENA region was one of the highest worldwide, which was around 18.1%, with most of the GCC countries showing prevalence levels above 20% (Source: Saudi Journal of Medicine & Medical Sciences).
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Shift – Strengthening Biosimilar Approval Framework |
|
|
Industry + Policy Shift – Localization of Pharmaceutical Manufacturing |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Saudi Food and Drug Authority (SFDA), Saudi Vision 2030)
Middle East and Saudi Arabia Pharmaceutical Drugs Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
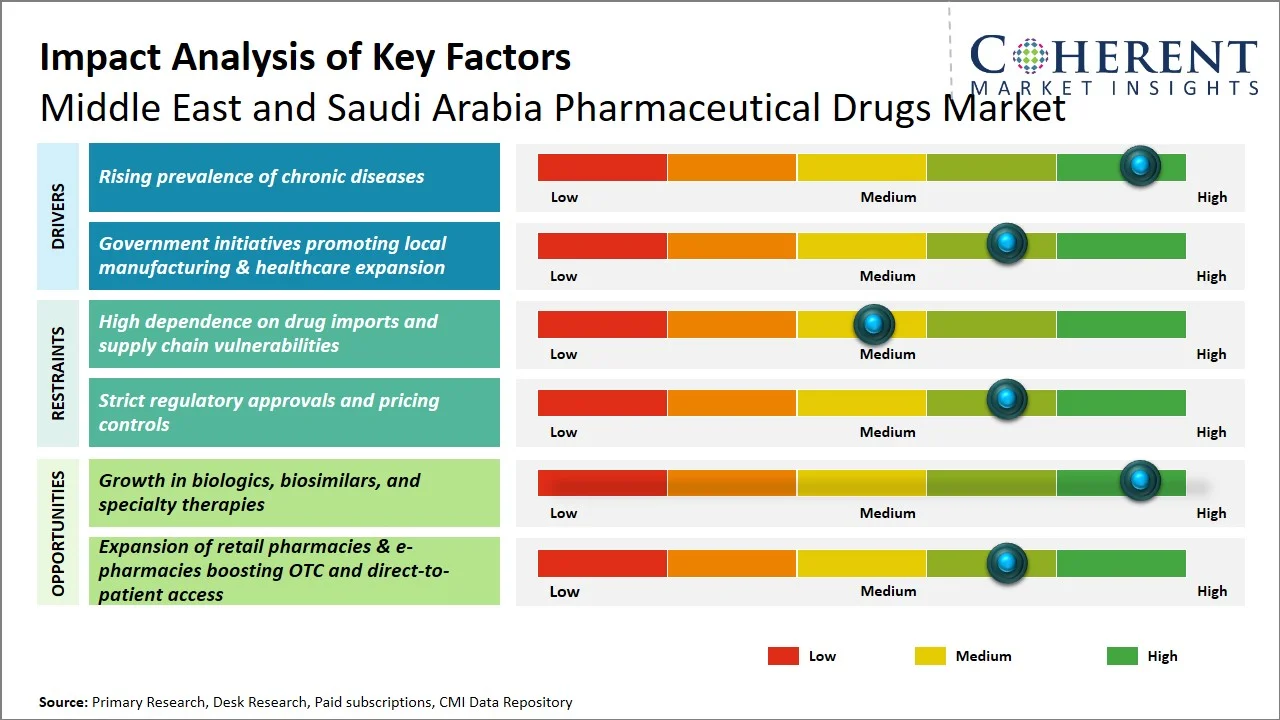
- Rising prevalence of chronic diseases: The rising prevalence of chronic diseases is one of the major factors that fuel the pharmaceutical drugs market growth in the Middle East countries, such as Saudi Arabia. The non-communicable diseases (NCDs) cause nearly 73% of all deaths in Saudi Arabia, where cardiovascular disorders contribute about 37% to all fatalities, while cancers accounts for 10%, and diabetes mellitus accounts for 3% of all deaths, which results in a steady increase in the demand for high-level medication, including biopharmaceuticals and specialty drugs (Source: Journal of Pharmaceutical Research International). Moreover, the research published by ResearchGate in December 2023 estimated that the cost associated with managing priority NCDs in Saudi Arabia will increase from USD 19.8 billion in 2020 to USD 32.4 billion in 2030 (an increase of USD 12.6 billion or 63%). (Source: ResearchGate)
- Government initiatives promoting local manufacturing & healthcare expansion: Government efforts, especially in Saudi Arabia, are notably boosting the pharmaceutical drugs market through healthcare expansion and localization. Saudi Vision 2030 places a strong emphasis on healthcare reform, aiming to improve access, service quality, and digital integration, while also bolstering domestic pharmaceutical production (Source: Saudi Vision 2030). According to the Ministry of Health Saudi Arabia, National Health Accounts (NHA) Saudi Arabia’s total health expenditure reached approximately USD ~44.1 billion (SAR 169.5 billion), underscoring substantial public investment in healthcare infrastructure and services (Source: National Health Accounts (NHA)). Furthermore, reforms like the introduction of new care models, the expansion of hospital facilities, and the encouragement of private sector involvement are improving drug accessibility and fostering local manufacturing. This, in turn, is lessening reliance on imports and building a more robust pharmaceutical industry.
Emerging Trends
- Shift Toward Biologics and Specialty Therapies: The pharmaceutical landscape is shifting, with a clear tilt toward biologics, biosimilars, and specialty drugs. This shift is largely a response to the escalating prevalence of chronic and intricate illnesses. The momentum is further fueled by streamlined regulatory processes and a broader embrace of sophisticated treatment modalities within hospitals and specialized care facilities.
- Localization of Pharmaceutical Manufacturing: Saudi Arabia, and other governments, are focusing on producing drugs domestically. The goal is to lessen their reliance on imports and to make their supply chains more robust. Vision 2030 is driving this, fostering collaborations, attracting investments, and pushing for the creation of local manufacturing plants for valuable pharmaceuticals.
Country Insights
Why is Saudi Arabia Strong Market for Pharmaceutical Drugs?
Saudi Arabia leads the Middle East and Saudi Arabia pharmaceutical drugs market, accounting for an estimated 29.26% share in 2026, buoyed by a healthcare system and infrastructure that the government is steadily expanding.
The Ministry of Health Saudi Arabia reports a consistent rise in healthcare funding. For instance, the Ministry's budget is slated to grow from USD 23 billion (SAR 86.3 billion) in 2024 to USD 26.5 billion (SAR 99.3 billion) in 2025. This increase signals ongoing expansion in healthcare services and capacity. Furthermore, the government's Vision 2030 initiative is actively improving healthcare access, hospital infrastructure, and the quality of services. This, in turn, is driving up the demand for pharmaceutical drugs throughout the Kingdom.
Why Does UAE Pharmaceutical Drugs Market Exhibit High Growth?
The UAE pharmaceutical drugs market is expected to exhibit the fastest growth with an estimated contribution of 13.08% share in 2026, driven by the swift development of its infrastructure and increasing expenditures.
The government, via the Ministry of Health and Prevention, is actively backing advanced medical services and fostering innovation, with the goal of positioning the country as a premier healthcare hub within the region. Additionally, the Dubai Health Authority has indicated that healthcare spending in Dubai reached USD 6.68 billion (AED 24.55 billion) in 2024 representing 10% increase from 2023. This surge is largely due to the widespread implementation of mandatory insurance, which is broadening access to healthcare and boosting pharmaceutical consumption. (Source: Dubai Health Authority)
Regulatory Framework Governing the Middle East and Saudi Arabia Pharmaceutical Market
|
Regulatory Authority |
Region |
Key Role |
Impact on Market |
|
Saudi Food and Drug Authority |
Saudi Arabia |
Drug approval, licensing, GMP compliance, pharmacovigilance |
Mandatory registration ensures safety, efficacy, and quality of all marketed drugs |
|
Pharmaceutical and Health Products Establishments Law |
Saudi Arabia |
Governs product registration and regulatory compliance |
Establishes legal framework for drug approval and market entry |
|
Gulf Health Council (GCC-DR) |
GCC Region |
Centralized drug registration system across GCC countries |
Harmonizes approvals and enables multi-country market access |
|
Ministry of Health (MOH) |
Saudi Arabia |
Procurement, pricing policies, healthcare regulations |
Drives pharmaceutical demand via public healthcare spending |
|
International Council for Harmonisation |
Global / Reference |
Standardizes technical requirements for drug approval |
Alignment improves regulatory efficiency and global market access |
|
Electronic Common Technical Document |
Saudi Arabia / GCC |
Digital submission format for drug dossiers |
Streamlines approval process and improves transparency |
|
Good Manufacturing Practice |
Global / Middle East |
Ensures quality manufacturing standards |
Mandatory for product approval and import authorization |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the growth in biologics, biosimilars, and specialty therapies creating new growth opportunities in the Middle East and Saudi Arabia pharmaceutical drugs market?
The pharmaceutical landscape in the Middle East, particularly in Saudi Arabia, is undergoing a significant shift, fueled by the rise of biologics, biosimilars, and specialty therapies. This shift is reshaping treatment approaches, moving toward more advanced and valuable options. This transformation is bolstered by government initiatives (e.g., Saudi Vision 2030 healthcare transformation program, National Biotechnology Strategy, Saudi Food and Drug Authority (SFDA) biosimilar regulatory guidelines, and localization programs under the National Industrial Development and Logistics Program (NIDLP), and regulatory frameworks.
In Saudi Arabia, the burden of non-communicable diseases accounts for around 73% of total deaths. This statistic highlights the growing demand for biologics in areas such as cancer treatment, cardiovascular disease prevention, and diabetic management (Source: SDI Article). To address this, the Saudi Food and Drug Authority (SFDA) introduced certain regulations on biosimilar development and approval. According to these regulations, not only will the quality and safety of medicines be preserved but also their delivery speed will be accelerated.
Market Players, Key Development, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In March 2026, Strides Pharma Science Limited made a move, snapping up several generic brands from Sandoz. This deal substantially broadened their reach throughout Africa. It also solidified their position in the regional generics market, making affordable treatments more readily available. This, in turn, should help the pharmaceutical market in the wider MEA region flourish.
- In February 2026, Stada Arzneimittel AG revealed plans for an USD 100 million (€85 million) investment to build a pharmaceutical production facility in Saudi Arabia. This move aligns with the nation's Vision 2030, which aims to boost domestic drug manufacturing. The new hub is expected to lessen the country's dependence on imports and strengthen the supply chain, effectively transforming Saudi Arabia into a regional hub for pharmaceutical production.
- In December 2024, Bio-Thera Solutions and Tabuk Pharmaceuticals joined forces to bring Bio-Thera's ustekinumab biosimilar, BAT2206, to market in Saudi Arabia. This collaboration aims to improve access to cutting-edge biologic treatments. The deal gives Tabuk exclusive control over the drug's manufacturing and distribution within the country, aligning with Saudi Arabia's goals for a more self-sufficient pharmaceutical industry.
Competitive Landscape
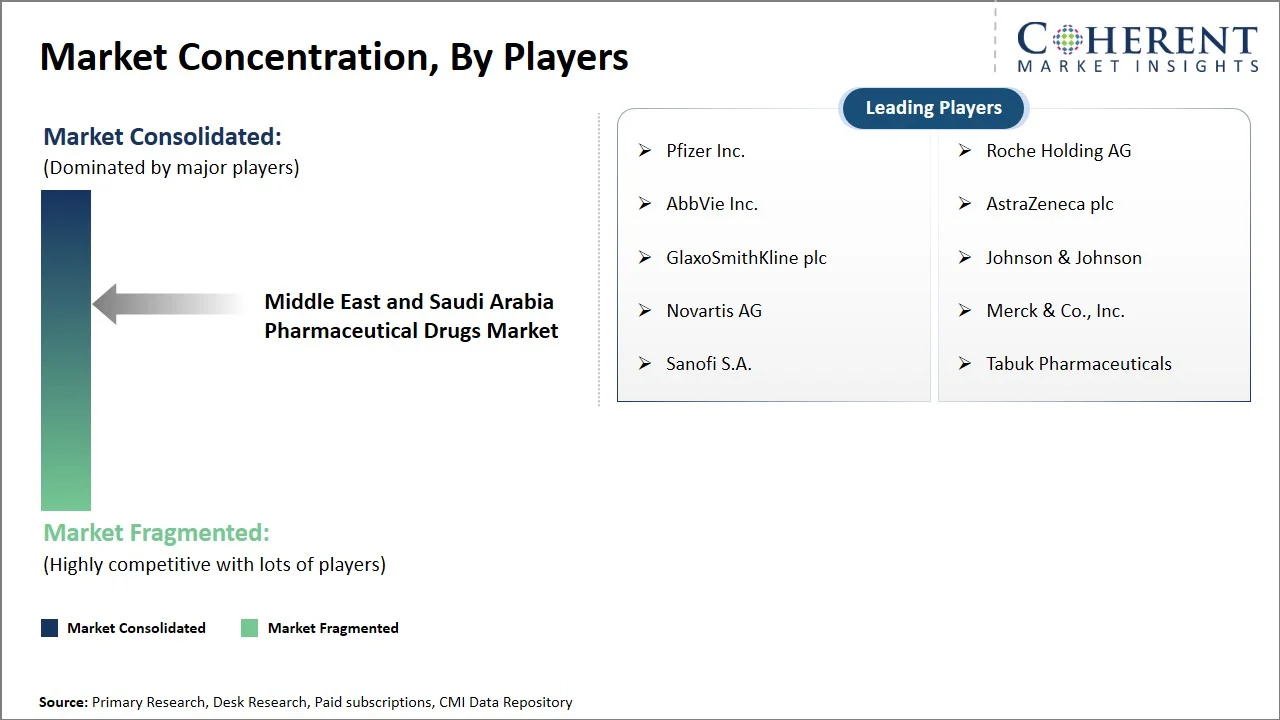
The Middle East and Saudi Arabia pharmaceutical drugs market is moderate to highly competitive due to the presence of large pharmaceutical companies around the world along with good competition from local firms that have been supported by the government through localization initiatives. Competitiveness will continue to increase as specialization, biosimilar production, and partnerships are key trends, whereas prices continue to be low in the generics sector. Key focus areas include:
- Expansion of local manufacturing capabilities aligned with government localization initiatives
- Increasing focus on biologics, biosimilars, and specialty drug portfolios
- Strengthening strategic partnerships with healthcare providers and public procurement bodies
Market Report Scope
Middle East and Saudi Arabia Pharmaceutical Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 37,475.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.9% | 2033 Value Projection: | USD 59,776.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., AbbVie Inc., GlaxoSmithKline plc, Novartis AG, Sanofi S.A., Roche Holding AG, AstraZeneca plc, Johnson & Johnson, Merck & Co., Inc., and Tabuk Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future for the pharmaceutical drugs within the Middle East would materialize in the form of an industry characterized by localization, specialization, and innovations, with Saudi Arabia being its focal point. The future industry will move away from import and towards innovation in medicines such as biologics and biosimilars due to the increased prevalence of chronic diseases and health expenditures, as well as strategies such as Vision 2030. With stronger regulation in tandem with increased prominence of technology medicine, innovation in medicines will be prioritized over generic drugs.
- This opportunity is maximized with regard to biologics and biosimilars in the oncology, cardiovascular, and diabetes therapy areas due to the Saudi Arabia’s strong dominance in the region, improved infrastructure for hospitals, and the prevalence of chronic illnesses in the region. There is ample pricing strength and demand visibility, especially through national procurement of the healthcare sector.
- In order to have an advantage over their competitors, manufacturers would need to localize manufacturing efforts, develop partnerships with the government and healthcare institutions in the country, and expand specialty drugs. They will need to make investments in regulatory affairs, supply chain management, and digitization including artificial intelligence solutions for healthcare. The companies that align themselves earlier with the localization and formulary approaches will be more successful because those who enter the market early will get the volume and sustained demand in the region
Market Segmentation
- Drug Type Insights (Revenue, USD Mn, 2021 - 2033)
- Generic Drugs
- Branded Drugs
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Prescription Drugs
- Over-the-Counter (OTC) Drugs
- Drug Class Insights (Revenue, USD Mn, 2021 - 2033)
- Analgesics
- Antibiotics
- Anti-infectives
- Antidepressants
- Antihypertensives
- Anticoagulants
- Antidiabetics
- Antihistamines
- Bronchodilators
- Antineoplastics
- Diuretics
- Others
- Therapeutic Area Insights (Revenue, USD Mn, 2021 - 2033)
- Cardiovascular Diseases
- Oncology
- Central Nervous System (CNS) Diseases
- Metabolic Disorders
- Respiratory Diseases
- Gynecology
- Gastrointestinal Diseases
- Infectious Diseases
- Dermatological Diseases
- Ophthalmological Diseases
- Musculoskeletal Disorders
- Others
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Oral
- Parenteral
- Topical
- Others
- Dosage Form Insights (Revenue, USD Mn, 2021 - 2033)
- Tablets
- Capsules
- Solutions
- Powders
- Patches
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Clinics
- Homecare Settings
- Specialty Centers
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Country Insights (Revenue, USD Mn, 2021 - 2033)
- Saudi Arabia
- UAE
- Oman
- Bahrain
- Kuwait
- Qatar
- Israel
- Rest of Middle East
- Key Players Insights
- Pfizer Inc.
- AbbVie Inc.
- GlaxoSmithKline plc
- Novartis AG
- Sanofi S.A.
- Roche Holding AG
- AstraZeneca plc
- Johnson & Johnson
- Merck & Co., Inc.
- Tabuk Pharmaceuticals
Sources
Primary Research Interviews
- Pharmaceutical Manufacturing Head (Saudi-based drug manufacturer)
- Supply Chain Director of pharmaceutical distributor (GCC region)
- Tabuk Pharmaceuticals (regional drug manufacturer and commercialization partner)
- Regulatory Affairs Specialist from Saudi Food and Drug Authority
- Hospital Procurement Head (public/private hospitals in Saudi Arabia)
- Clinical Specialist (Endocrinologist / Cardiologist treating chronic diseases)
Stakeholders
- Pharmaceutical Manufacturers (Middle East & Saudi-based + MNC regional offices)
- Biotechnology Companies (GCC-focused biologics & specialty therapies)
- Contract Manufacturing Organizations (Saudi localization initiatives – Vision 2030)
- API & Raw Material Suppliers (import-dependent supply chain in GCC)
- Regulatory & Compliance Experts (Saudi Food and Drug Authority – SFDA, GCC authorities)
- Distributors & Healthcare Supply Chain Providers (regional pharma distributors in KSA & UA
Databases
- World Health Organization (WHO) – Middle East NCD and diabetes prevalence data
- Saudi Food and Drug Authority (SFDA) – Drug approvals, pricing, and localization policies
- Ministry of Health (Saudi Arabia) – Healthcare infrastructure and disease burden data
- Gulf Health Council – Regional health statistics across GCC countries
- Centers for Disease Control and Prevention (CDC) – Comparative disease trends
- National Institutes of Health (NIH) – Clinical research and endocrine-related studies
Magazines
- Pharma Middle East
- MEED (healthcare & pharma investments in GCC)
- Arab Health Magazine
- Pharmaceutical Technology
- Fierce Pharma
- BioPharma Dive
- Contract Pharma
Journals
- Journal of Clinical Endocrinology & Metabolism
- Endocrine Reviews
- Diabetes Care
- The Lancet Diabetes & Endocrinology
- Frontiers in Endocrinology
Newspapers
- Arab News (Saudi healthcare & pharma sector developments)
- Gulf News (UAE & regional pharma updates)
- CNBC (Middle East healthcare investments coverage)
- Asharq Al-Awsat (regional policy & healthcare developments)
- The National (UAE-based coverage of pharma and healthcare sector)
Associations
- Gulf Pharmaceutical Industries Association (GPIA)
- Saudi Pharmaceutical Society
- International Diabetes Federation (Middle East data)
- Endocrine Society (regional disease insights)
- Pharmaceutical Research and Manufacturers of America (for global benchmarking)
Public Domain Sources
- WHO Middle East Regional Reports
- SFDA Drug Database & Pricing Lists
- Saudi MOH Statistical Yearbooks
- Gulf Health Council Reports
- World Bank Middle East Healthcare Data
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients