Global Paracentesis Kit Market Size and Forecast – 2026 To 2033
The global paracentesis kit market is expected to grow from USD 1.28 Bn in 2026 to USD 2.25 Bn by 2033, registering a compound annual growth rate (CAGR) of 8.39%. The global paracentesis kit market is poised for significant expansion, fueled by increased cases of liver disorders that cause complications such as ascites, which need constant drainage of fluid.
The Centers for Disease Control and Prevention (CDC) reports that approximately 4.5 million adults in the U.S. are diagnosed with liver disease (1.8% of the adult population), indicating a substantial at-risk population. In terms of severity, chronic liver disease and cirrhosis account for over 52,274 deaths annually in the U.S., making it the 9th leading cause of death, with a mortality rate of 15.4 deaths per 100,000 population
(Source: Centers for Disease Control and Prevention)
Key Takeaways of the Global Paracentesis Kit Market
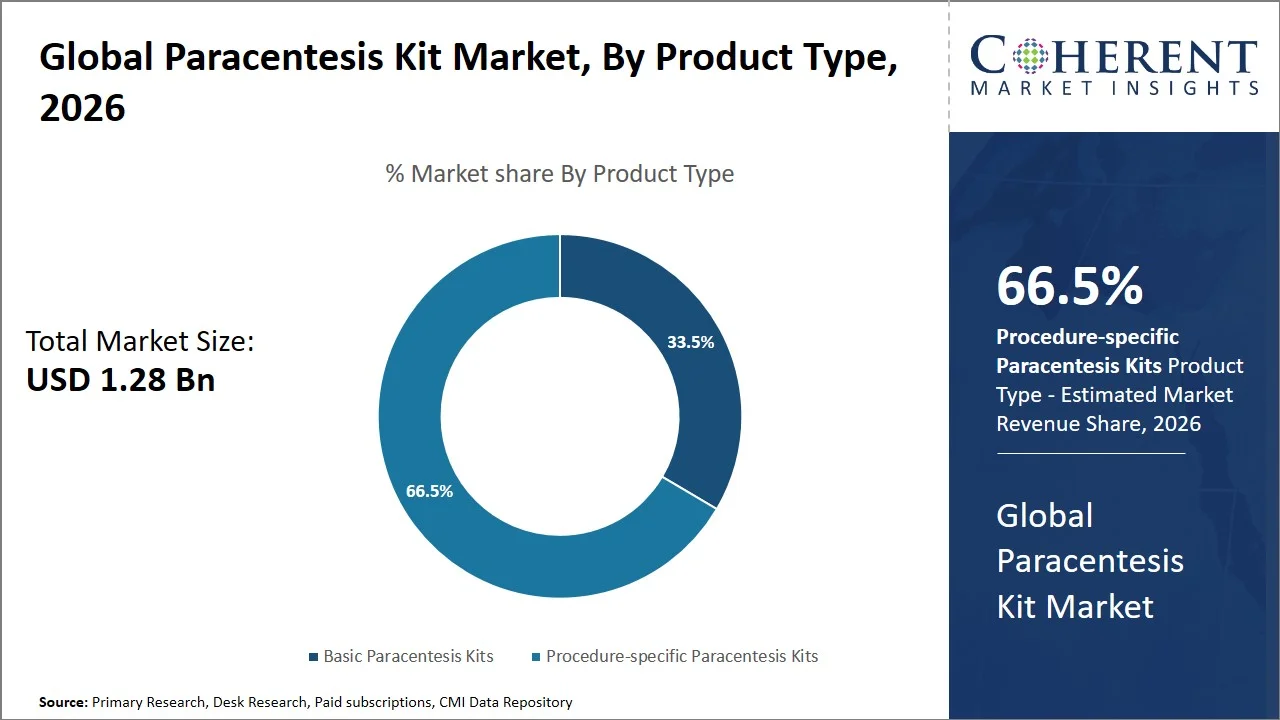
- Procedure-specific paracentesis kits are projected to hold 66.5% of the global paracentesis kit market share in 2026, making it the dominant product type segment, with North America leading the dominance because of its stringent policies on infection control (such as CDC Standard Precautions Guidelines; OSHA Bloodborne Pathogens Standard) and the use of single-use sterile devices. For instance, according to the recommendations of the CDC, single-use medical devices should be used on one individual before being thrown away to avoid infections. (Source: CDC)
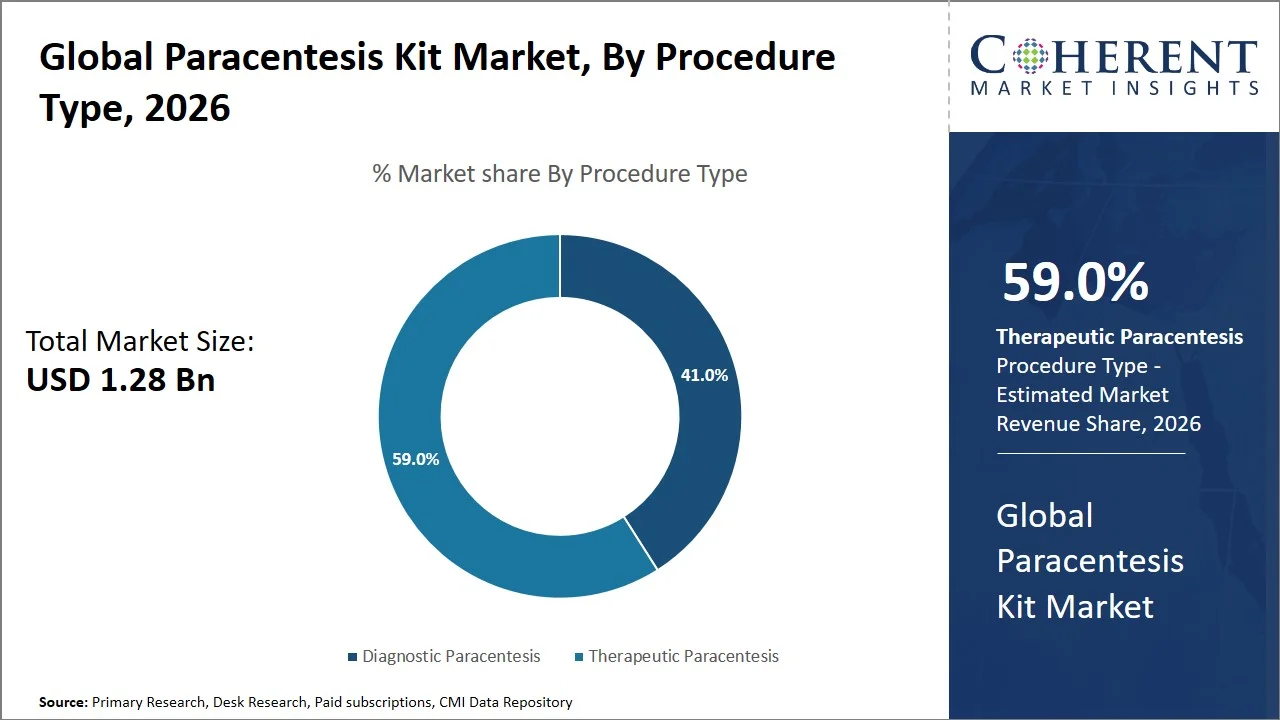
- Therapeutic paracentesis is projected to hold 59.0% of the global paracentesis kit market share in 2026, making it the dominant procedure type segment, with Europe as one of the major contributors due to the existing guidelines on handling ascites among patients with liver disorders. For instance, the European Association for the Study of the Liver suggests large volume paracentesis as a regular management technique for ascites among their patients, reinforcing the high frequency of therapeutic procedures across European healthcare systems. (Source: Journal of Hepatology)
- Hospitals are projected to hold 62.0% of the global paracentesis kit market share in 2026, making it the dominant end user segment, with North America being the leader due to the large number of procedures carried out and the strict observance of infection control measures in the hospital environment. For instance, the CDC mentions that medical and surgical instruments used during procedures that interact with the sterile parts of the body must be properly sterilized in order not to transmit any infections. (Source: CDC)
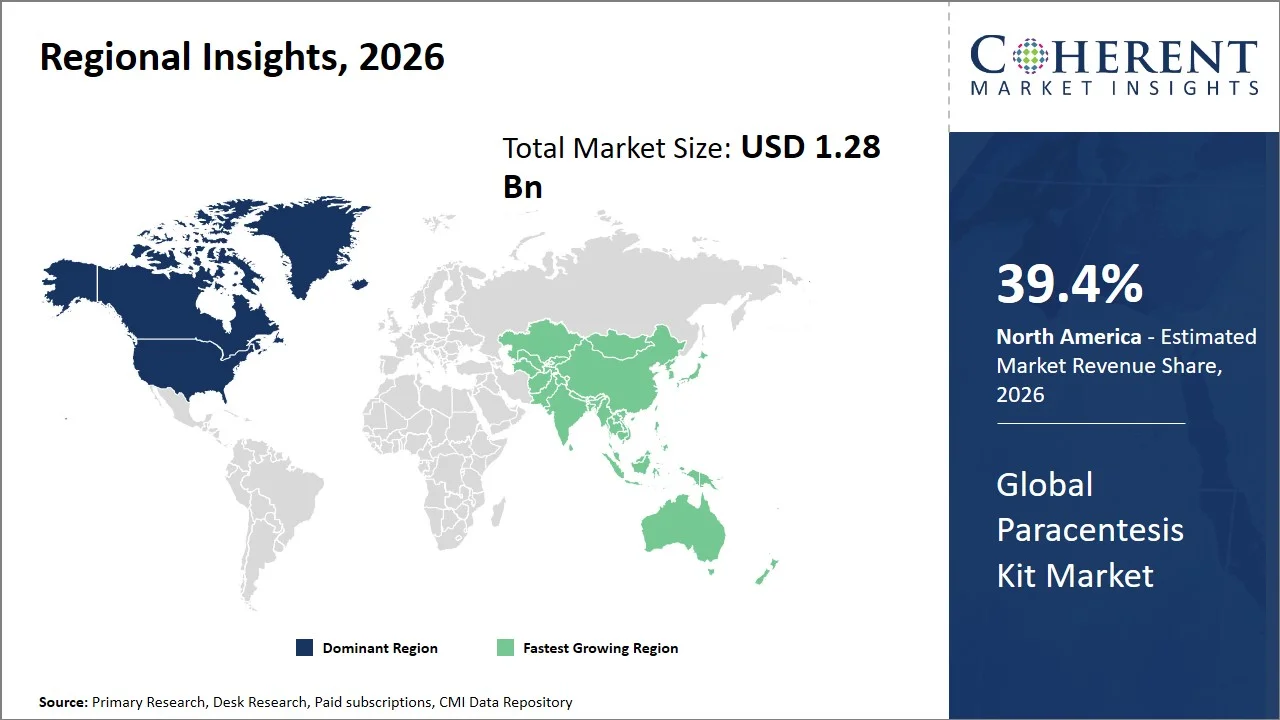
- North America maintains dominance with an expected share of 39.4% in 2026, attributed to the high procedural and surgical volumes that characterize sophisticated healthcare infrastructures and stringent clinical training. For instance, according to the American Hospital Association, the U.S. has more than 6,100 hospitals providing high volumes of hospitalization and procedure-based treatment, thus enabling a high number of intervention procedures, like paracentesis. Such a well-developed healthcare delivery infrastructure ensures that specialized kits are consistently used in different institutions. (Source: American Hospital Association)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 23.5% share in 2026, caused by the significant rise in the incidence of liver diseases and the availability of diagnosis and intervention services. For instance, the World Health Organization (WHO) reports that liver disease, which includes cirrhosis, is one of the main reasons for death in the region, especially in nations such as China and India. (Source: WHO)
- Increasing Adoption of Ultrasound-Guided Paracentesis Procedures: The rising number of portable ultrasound devices is making paracentesis procedures safer and more accurate, minimizing potential risks like puncturing organs during the procedure. This phenomenon is leading to the increased need for paracentesis kits that can support image-guided
- Rising Demand for Home-Based and Outpatient Ascites Management In line with the high incidence of chronic liver conditions and cancer-related ascites, a transition towards minimally invasive repeated drainage methods in outpatient facilities are being observed. The demand for user-friendly paracentesis kits that can be used in outpatient facilities and even at home is growing.
Why Does the Procedure-specific Paracentesis Kits Segment Dominate the Global Paracentesis Kit Market?
Procedure-specific paracentesis kits are projected to hold the market share of 66.5% in 2026, as they offer high standardization, safety, and efficiency compared to individually assembled components. Procedure-based kits include all necessary sterilized medical devices, including drainage catheters, needles, tubing, and collecting devices. This means that one does not need to assemble equipment before use, which minimizes the chances of contamination. Sterilized medical equipment is important in paracentesis procedure because of recommendations provided by organizations such as CDC. Organizations recommend sterilized equipment for procedures carried out under aseptic conditions with single-use equipment. For instance, the WHO’s guidelines on the prevention of surgical site infections state that any procedure carried out invasively should always be done using sterile equipment (Source: WHO). Additionally, manufacturers are focused on developing integrated systems. For example, Merit Medical ReSolve Paracentesis Kit is a new innovative medical device that provides a full set for the fluid drainage procedure. (Source: Merit Medical Systems)
Why is Therapeutic Paracentesis the Most Preferred Procedure Type?
To learn more about this report, Request Free Sample
Therapeutic paracentesis is projected to hold a market share of 59.0% in 2026, because it helps provide quick symptomatic relief to patients who are suffering from ascites that can be either moderate or severe. Patients with liver cirrhosis and fluid accumulation due to their malignancies usually have more severe symptoms. The difference between diagnostic and therapeutic paracentesis is the fact that diagnostic paracentesis is used solely for fluid examination, whereas therapeutic paracentesis involves the withdrawal of large amounts of fluid, hence providing much needed relief to the patients. The American Association for the Study of Liver Diseases has recommended the use of large volume paracentesis as a first line of therapy for patients with tense ascites who cannot be managed with diuretics. (Source: American Association for the Study of Liver Diseases)
Moreover, increased incidence rates of liver disorders and recurring cases of ascites have resulted in multiple treatment sessions, thereby raising procedure counts. Such an evident clinical need along with preference for the procedure backed by guidelines and rising disease burden is propelling the adoption of therapeutic paracentesis.
Hospitals Segment Dominates the Global Paracentesis Kit Market
The hospitals segment is projected to hold a market share of 62.0% in 2026, because hospitals remain the key locus of care for cases of acutely, complicated, and first-time ascites when an all-inclusive approach is needed. The healthcare facility allows for the integration of various treatment approaches like imaging procedures, such as ultrasound guidance; laboratory analysis of the ascitic fluid; and availability of emergency treatment in the event of complications like hypotension or infection.
Furthermore, centralized procurement processes and standard procedural practices ensure the use of kit packages specific to the procedure and sterile. For example, the WHO suggests that invasive procedures should be carried out in healthcare facilities that have sufficient infrastructure, along with sterilized equipment and qualified professionals, for optimal infection prevention. (Source: WHO)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Strengthening Infection Prevention Regulations and Sterile Device Standards (2025) |
|
|
Implementation of EU Medical Device Regulation (MDR) Strengthening Compliance (2024–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: CDC, European Commission)
Global Paracentesis Kit Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
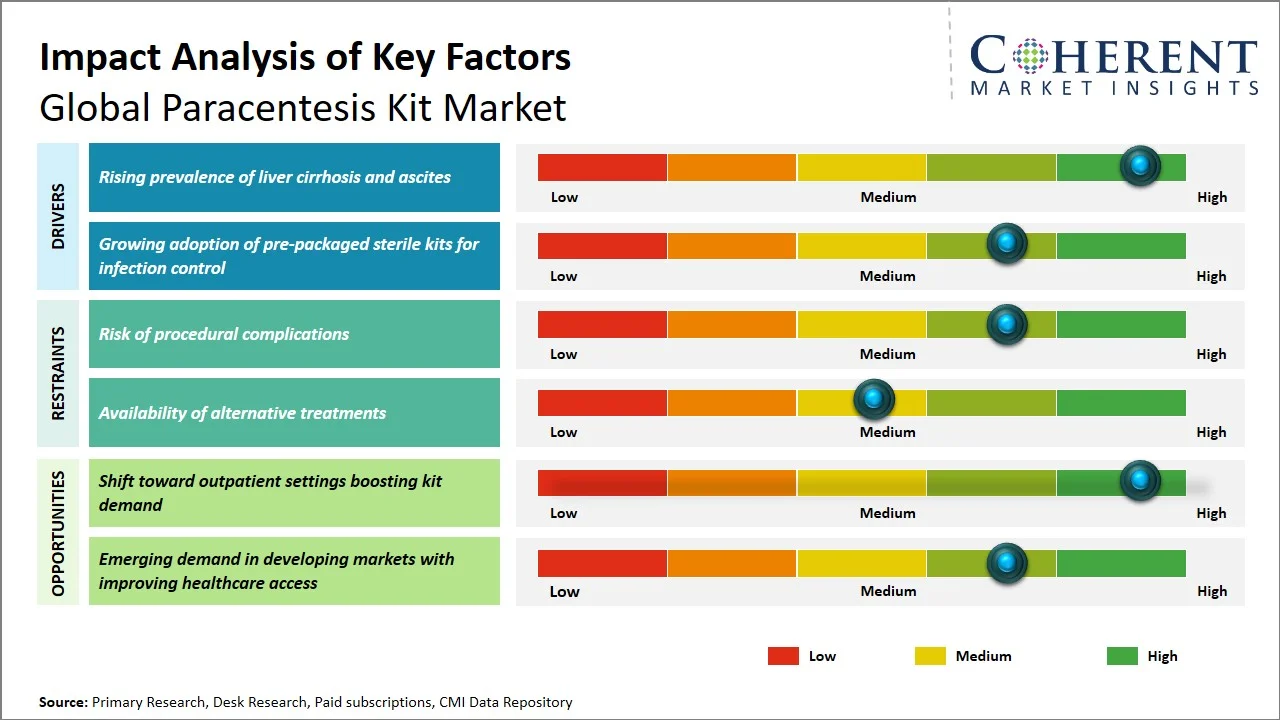
- Rising prevalence of liver cirrhosis and ascites: As per reports, global burden liver cirrhosis incidence rose to almost 58.4 million in 2021, marking an increase from the reported 36.9 million in 1990, with more than 1.4 million deaths yearly, which denotes the rising health issue faced by the population. This trend will continue, with predictions estimating that there will be a significant increase in the incidence and mortality due to cirrhosis in 2060, owing to rising incidences of alcohol-induced liver diseases, hepatitis, and NAFLD, which will further escalate the necessity of procedures such as paracentesis (Source: National Library of Medicine). Additionally, ascites, a serious complication of liver cirrhosis, affects almost 50–70% of liver cirrhosis patients over 10 years with almost all fluid drainage procedures being due to ascites (Source: ScienceDirect). Furthermore, it is believed clinically that 5-10% of liver cirrhosis patients develop ascites on an annual basis, making the need for treatment options like paracentesis very important. (Source: Open Exploration Publishing Inc.)
- Growing adoption of pre-packaged sterile kits for infection control: With an increase in emphasis on infection prevention and patient safety, there has been an increasing trend towards the use of single-use, sterile paracentesis kits. The single-use paracentesis kits contain all the equipment required for performing the procedure in a sterile environment; hence, there is no need for manual assembly, thus minimizing the chances of contamination. Healthcare providers now prefer standardized sterile kits because of rising concerns about Hospital Acquired Infections (HAIs). This has made pre-assembled paracentesis kits a good choice because they are more efficient, and they offer great growth opportunities for manufacturing companies.
Emerging Trends
- Shift Toward Safety-Engineered Paracentesis Kits: The inclusion of additional safety features, including needle guards, leak prevention valves, and closed drainage systems, are becoming more common among manufacturers to minimize the risks of infections and procedural complications. The trend reflects increasing demands for patient and healthcare worker safety at hospitals.
- Growing Adoption of Portable and Bedside Paracentesis Solutions: The need for kits that can be used at bedside and point-of-care level, such as those in emergency rooms, are increasingly gaining popularity since they help in quick response and minimize time in the hospital when dealing with recurrent ascites.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Paracentesis Kit?
North America leads the global paracentesis kit market, accounting for an estimated 39.4% share in 2026, owing to the high number of procedures, advanced health care systems, and high rate of adoption of minimally invasive techniques. There are relatively high incidences of diseases related to the liver owing to the obesity and metabolism problems in the patients. For instance, the National Library of Medicine indicates that prevalence of nonalcoholic fatty liver disease (NAFLD) in the U.S. is 38%, having increased by 50% within the past 3 decades (Source: National Library of Medicine). This results in the formation of a high patient base in need of the procedure. The favorable reimbursement system (such as Centers for Medicare & Medicaid Services provides reimbursement for paracentesis procedures under CPT codes 49082 and 49083, Medicare’s Hospital Outpatient Prospective Payment System (OPPS) supports coverage for ultrasound-guided drainage procedures) and use of ultrasound guided technique further contribute to the acceptance of the procedure.
Why Does Asia Pacific Paracentesis Kit Market Exhibit High Growth?
The Asia Pacific paracentesis kit market is expected to exhibit the fastest growth with an estimated contribution of 23.5% share to the market in 2026, owing to the higher burden of liver diseases, higher number of patients, and improvements in the healthcare system. The region contributes to over 62.6% of all deaths around the world from liver conditions, with almost half of all deaths from cirrhosis happening in Asia Pacific. This highlights the need for procedures that will address the high volume of ascites (Source: National Library of Medicine). Moreover, the region is experiencing an increase in metabolic conditions and non-alcoholic fatty liver disease (NAFLD) which have become a common cause of chronic liver diseases, mainly due to changing lifestyle and urbanization. (Source: National Library of Medicine)
In addition to the large population, there have been many investments in healthcare facilities and availability of procedures that have increased the procedure rate. For instance, Asia’s healthcare sector is projected to reach USD 5 trillion by 2030, contributing nearly 40% of global healthcare growth; total healthcare spending in Asia Pacific has already exceeded USD 4 trillion, with consistent annual growth of 7%. (Source: Express Healthcare)
Further, the National Health Commission of China has been making efforts in developing hospitals’ infrastructure and increasing the accessibility of minimally invasive interventions in line with its health reforms, thus ensuring that interventional procedures become more accessible through urban and regional healthcare settings.
Why are the U.S. Leading Innovation and Adoption in the Paracentesis Kit Market?
The innovation and adaptation of the paracentesis kits in the U.S. are the greatest, owing to their advanced medical care system and their quick adoption of less invasive procedures. Strict regulations and controls in the market have been facilitated through the efforts of the U.S. Food and Drug Administration, which guarantees high-quality products but is still able to fast-track the approval process for new innovations.
Moreover, the country boasts leading manufacturers like BD (Becton, Dickinson and Company) and Merit Medical Systems that facilitate ongoing product developments. Good reimbursement practices supported by the Centers for Medicare & Medicaid Services foster adoption of procedures. In addition, wide usage of ultrasound-based procedures and compliance with standard procedures enhance demand for procedure-based kits.
Is U.K. a Favorable Market for Paracentesis Kit?
U.K. represents an ideal environment for paracentesis kit market facilitated by the presence of a conducive environment, attributed to the efficient healthcare system in the country and the existence of stringent clinical guidelines (such as those from the National Institute for Health and Care Excellence and the British Society of Gastroenterology), which standardize the management of ascites and promote safe, evidence-based procedures. The scientific evidence for managing ascites using paracentesis is backed by the clinical guidelines from NICE (National Institute for Health and Care Excellence). The availability of paracentesis ensures that patients receive standard services. Invasive techniques using ultrasound techniques have played a vital role in increasing the success rate and safety of the surgery.
Is China Emerging as a Key Growth Hub for the Paracentesis Kit Market?
China experiences a high prevalence of chronic liver diseases, especially cirrhosis secondary to hepatitis B infections. Hence, there is a growing number of patients who require interventions that manage the complications associated with ascites. For instance, the WHO estimated 87 million people living with chronic hepatitis B and 7.6 million people living with chronic hepatitis C in China (Source: WHO). In addition, health reforms initiated by the National Health Commission of China have resulted in the expansion of hospital facilities and improved access to minimally invasive procedures.
Also, there are increasing expenditures on the purchase of advanced medical equipment and adherence to standardized procedures. The higher prevalence of indigenous medical device companies contributes to affordable prices. For example, companies like Shandong Weigao Group Medical Polymer Company Limited are expanding their medical device portfolios and distribution networks, enhancing the accessibility of procedure-specific kits across China.
Why Does Germany Top the Europe Paracentesis Kit Market?
Germany is the most dominant nation in the paracentesis kit market for Europe owing to its very advanced healthcare setup and procedural capability. Germany has an extensive setup of hospitals equipped with state-of-the-art equipment and a substantial number of minimally invasive procedures conducted by proficient health practitioners. High standards set by EU MDR 2017/745 regulations make sure that there is usage of sterilized and procedure-specific kits.
Moreover, effective reimbursement policy and early adaptation of medical technology by Germany facilitate extensive use of paracentesis procedures. For example, the German statutory health insurance scheme, managed by the Federal Joint Committee (G-BA), covers hospital procedures, guaranteeing access to patients while promoting standardization of medical devices used (G-BA).
The market for paracentesis kit in India is growing due to the increased incidence of liver diseases and better availability of healthcare facilities in the country. The Indian Council of Medical Research and World Health Organization figures indicate that about 259,749 people died in India due to liver disease (about 2.95% of all deaths) (Source: National Library of Medicine). Additionally, an estimated number of 40 million people have been found to have hepatitis B infection, which is one of the reasons for the development of ascites (Source: The Liver Foundation). Furthermore, the improvement in health care infrastructure through programs like Ayushman Bharat is increasing accessibility to hospital treatment, including minimal invasive treatments.
Regulatory Landscape Governing the Global Paracentesis Kit Market
|
Region |
Key Regulatory Authority |
Framework/Regulation |
Approval Pathway |
Key requirements |
|
North America |
U.S. Food and Drug Administration (U.S.), Health Canada |
21 CFR (U.S.), CMDR (Canada) |
510(k) clearance (Class II), Device Licensing (Canada) |
QSR (21 CFR Part 820), ISO 13485, labeling, MDR reporting |
|
Europe |
European Commission, Notified Bodies |
EU MDR 2017/745 |
CE Marking via conformity assessment |
UDI, EUDAMED registration, clinical evaluation, PMS |
|
Asia Pacific |
National Medical Products Administration, Pharmaceuticals and Medical Devices Agency, Central Drugs Standard Control Organization |
Country-specific regulations (aligned with ISO/GHTF) |
Product registration, pre-market approval |
Local clinical data (if required), QMS compliance, licensing |
|
Latin America |
ANVISA, COFEPRIS |
Country-specific regulations |
Device registration & approval |
GMP certification, technical dossiers, reliance on CE/FDA data |
|
Middle East & Africa |
Saudi Food and Drug Authority, Ministry of Health and Prevention UAE |
National frameworks (often EU/FDA-referenced) |
Registration & import approval |
CE/FDA certification, local agent, conformity assessment |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the shift toward outpatient settings boosting kit demand creating new growth opportunities in the global paracentesis kit market?
The trend towards the use of outpatient services is creating substantial demand for paracentesis kits because it allows fluid drainage procedures to be conducted outside hospitals, thus making it possible for them to be done in outpatient clinics and even homes. For example, indwelling catheter devices such as PleurX by BD (Becton, Dickinson and Company) can be used by individuals suffering from recurrent malignant ascites to conduct fluid drainage procedures at home without necessarily requiring them to visit the hospital each time. These devices can be implanted in an outpatient setting and allow constant drainage procedures, hence the growing use of single-use procedure-specific kits (Source: BD). Consequently, companies are seeing emerging growth prospects owing to the emergence of easy-to-use kits coupled with an increased focus on ambulatory surgery centers and home health care segments.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In September 2025, Bearpac Medical signed a multi-year exclusive distribution agreement with SYNDEO Medical for the Passio Pump Drainage System. This agreement gives SYNDEO the exclusive right to sell and distribute the product around the world, except in the U.S. and the U.K. This partnership is expected to help Bearpac become more well-known around the world by using SYNDEO's distribution network and speed up the adoption of the product in other countries.
- In January 2024, Laborie Medical Technologies introduced the RenovaRP Centesis System, a next-generation fluid drainage device designed for both paracentesis and thoracentesis, featuring design enhancements to the handle and nest to improve overall user experience and procedural efficiency.
Competitive Landscape
The global paracentesis kit market is highly competitive owing to the increasing burden of chronic liver diseases and cancer-induced ascites. The need for quality products that facilitate the safe drainage of fluid as well as the affordability of the product is on the rise. The importance of minimizing invasion during surgeries, infection control measures, and consistency within the kit sets are being realized to achieve better outcomes. Firms that offer innovation within their products and at the same time minimize the costs of their kit sets have been able to outsmart their competitors. Key focus areas include:
- Development of procedure-specific, pre-assembled kits
- Integration of safety-engineered components
- Advancements in catheter design
- Increasing adoption of ultrasound-guided paracentesis
- Expansion into outpatient and home healthcare settings
- Strategic collaborations with hospitals, gastroenterology clinics, and distributors
Market Report Scope
Paracentesis Kit Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.28 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.39% | 2033 Value Projection: | USD 2.25 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BD, Merit Medical Systems, Cardinal Health, Teleflex Incorporated, B. Braun Melsungen AG, Cook Medical, Vygon Group, AngioDynamics, Medline Industries, and Rocket Medical plc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future of the paracentesis kit market in the years ahead can be anticipated in light of the shift towards standardization of procedures, design for safety, and an increase in minimally invasive bedside procedures. The market will see more and more emphasis on ready-to-go, one-time use kits as well as ultrasound-guided procedures which can improve efficiency and reduce the chances of complications. Outpatient facilities will play a role in determining changes in how and where the procedures are done.
- Greatest opportunities lie in the procedure-specific kits in the hospitals in the U.S. paracentesis kit market, which sees a large number of procedures performed every year and uses strict protocols that require the continued use of standardized kits. Therapeutic paracentesis in the China paracentesis kit market provides good growth potential, thanks to an increasing burden of liver disease and improved availability of hospital-based intervention.
- For gaining a competitive edge in the market, it is essential for the participants to focus on developing ergonomic, safety-conscious, and feature-packed kits. Regulatory and infection prevention guidelines, as well as clinical evidence, must be taken into consideration when entering the market. In addition to that, forming alliances with hospitals will become a key element in securing their success in the market.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Basic Paracentesis Kits
- Procedure-specific Paracentesis Kits
- Procedure Type Insights (Revenue, USD Bn, 2021 - 2033)
- Diagnostic Paracentesis
- Therapeutic Paracentesis
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Specialty Clinics
- Others
- Distribution Channel Insights (Revenue, USD Bn, 2021 - 2033)
- Direct Sales
- Distributors and Wholesalers
- Others
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- BD
- Merit Medical Systems
- Cardinal Health
- Teleflex Incorporated
- Braun Melsungen AG
- Cook Medical
- Vygon Group
- AngioDynamics
- Medline Industries
- Rocket Medical plc
Sources
Primary Research Interviews
- Paracentesis kit manufacturers and distributors
- Gastroenterologists, hepatologists, and interventional radiologists
- Hospitals, ambulatory surgical centers, and specialty clinics
- Home healthcare providers and nursing professionals
Stakeholders
- Paracentesis Kit Solution Providers
- End-use Sectors
- Hospitals and Clinics
- Ambulatory Surgical Centers
- Specialty Gastroenterology & Hepatology Clinics
- Home Healthcare Settings
- Regulatory & Health Bodies: U.S. Food and Drug Administration, European Medicines Agency, World Health Organization
Databases
- World Bank – Healthcare expenditure database
- World Health Organization – Global Health Observatory (GHO)
- Organization for Economic Co-operation and Development (OECD) – Health Statistics
- Global Burden of Disease (GBD) Study – Liver disease and cancer prevalence
- International Agency for Research on Cancer (IARC) – Cancer statistics
Magazines
- MedTech Dive – Medical device innovations and procedural trends
- Healthcare Business Today – Hospital and outpatient care trends
- Medical Device Network – Product launches and device advancements
- Gastroenterology & Endoscopy News – Clinical procedure updates
- Hospital Healthcare Europe – Healthcare infrastructure and adoption
Journals
- Hepatology – Liver disease and ascites management studies
- Journal of Hepatology – Clinical research on cirrhosis and ascites
- American Journal of Gastroenterology – Paracentesis procedure outcomes
- BMJ Open Gastroenterology – GI disorder management
- The Lancet Gastroenterology & Hepatology – Advanced clinical insights
Newspapers
- The Wall Street Journal – Healthcare investments and medtech trends
- Financial Times – Global healthcare system developments
- Business Standard – Healthcare infrastructure growth
- The Economic Times – Hospital expansion and medical device demand
- Mint – Healthcare innovation and policy updates
Associations
- American Association for the Study of Liver Diseases (AASLD)
- European Association for the Study of the Liver (EASL)
- American College of Gastroenterology (ACG)
- Society of Interventional Radiology (SIR)
- World Gastroenterology Organization (WGO)
Public Domain Sources
- U.S. FDA – Medical device approvals and safety guidelines
- European Commission – Medical Device Regulation (MDR 2017/745)
- National Institutes of Health (NIH) – Liver disease and ascites research
- Centers for Disease Control and Prevention (CDC) – Chronic liver disease data
- National Health Service (NHS) – Clinical guidelines for ascites management
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients