Global Propranolol Market Size and Forecast – 2026 to 2033
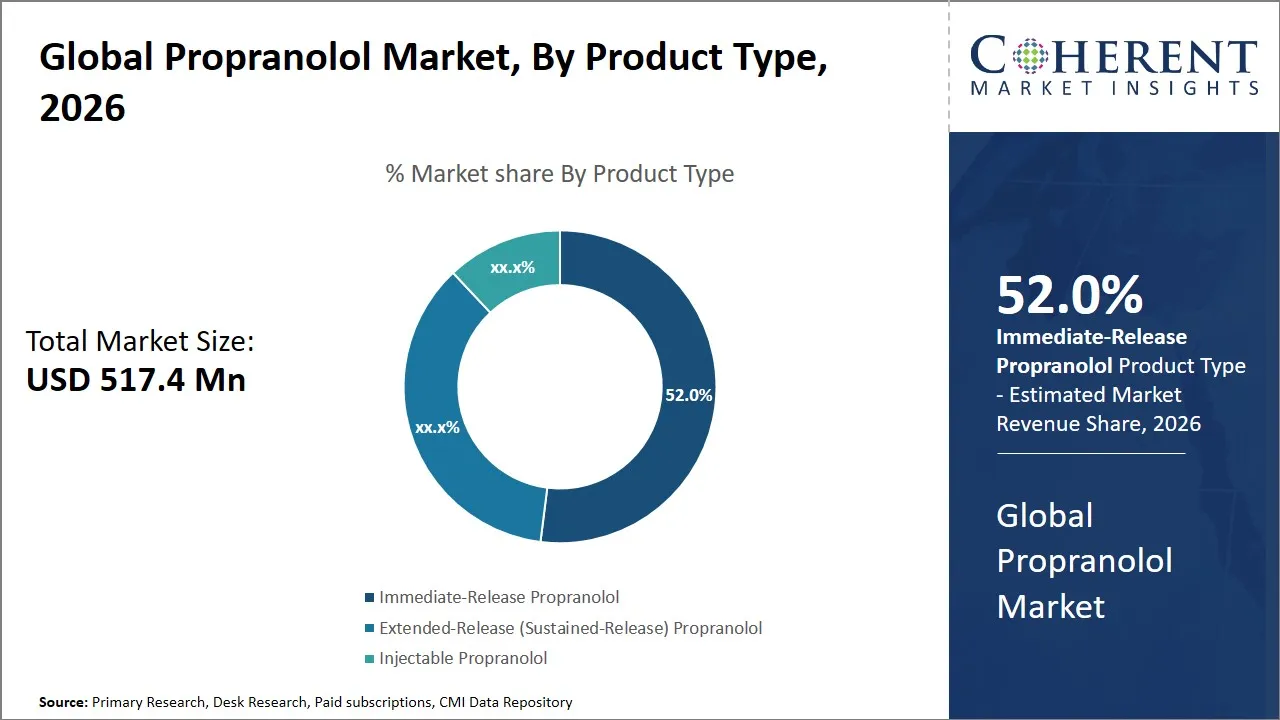
According to Coherent Market Insights, the global propranolol market is estimated to be valued at USD 517.4 Mn in 2026 and is expected to reach USD 894.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 3.8% from 2026 to 2033. This steady growth reflects increasing awareness and adoption of propranolol for various cardiovascular and neurological conditions, along with expanding healthcare infrastructure in emerging economies.
Key Takeaways of the Global Propranolol Market
- Immediate-Release propranolol segment is expected to lead the global propranolol market, capturing 52% share in 2026.
- 20 to 40 mg segment is estimated to represent 45% of the global propranolol market share in 2026.
- Hypertension segment is projected to dominate with 30% of the propranolol market share in 2026.
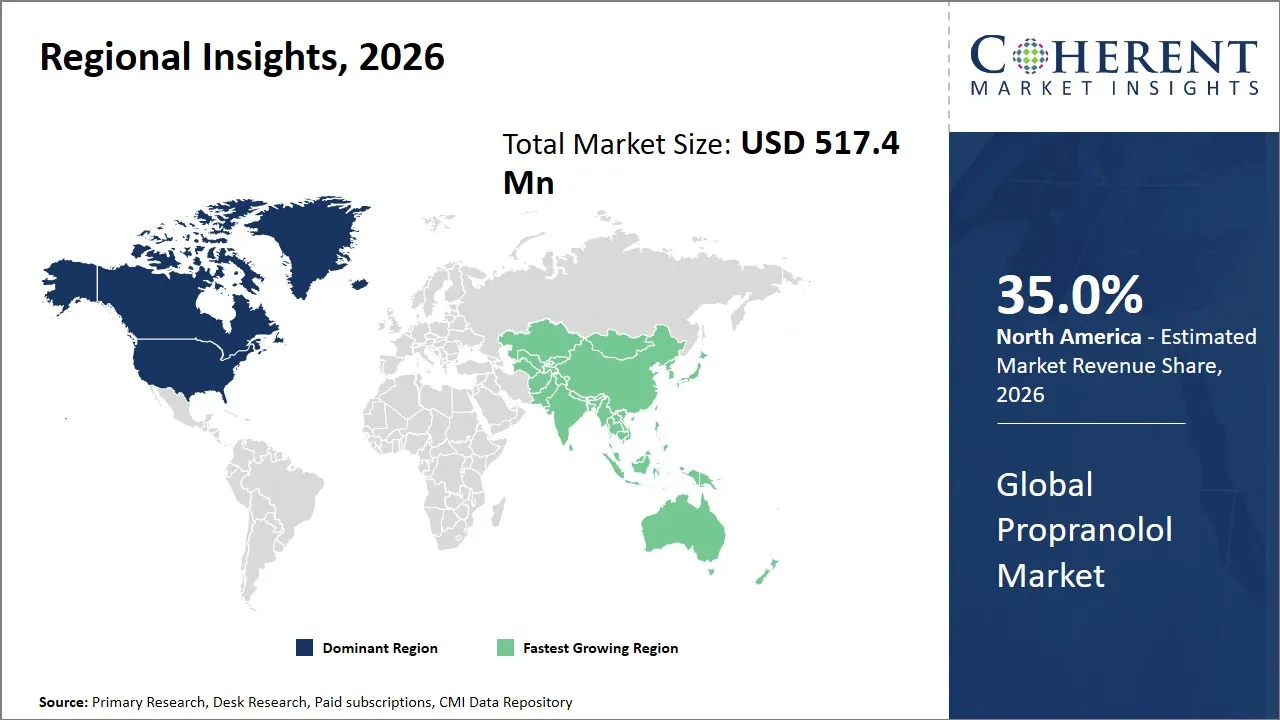
- North America is expected to lead the market, holding a share of 35% in 2026. Asia Pacific is anticipated to be the fastest-growing region, with 30% share in 2026.
Market Overview
- Propranolol is a first-generation non-selective beta-blocker with decades of clinical usage. It is positioned as a cost-effective, well-established therapy for hypertension, arrhythmia, migraine prevention, essential tremor, and anxiety-related conditions. The product is in a mature lifecycle phase with limited patent-driven innovation.
- Cardiovascular indications account for the majority of global revenue, while neurological and anxiety-related applications contribute incremental growth. Off-label prescribing trends, particularly in anxiety management, are gradually increasing retail pharmacy sales in developed markets.
- Telemedicine platforms and e-prescription services are gradually increasing propranolol accessibility, particularly for hypertension and anxiety management in remote and underserved regions. This also facilitates adherence tracking and repeat prescription fulfillment.
- The aging global population and rising prevalence of cardiovascular and neurological disorders are key demand drivers. Older adults represent a high-prescription segment, particularly for chronic propranolol therapy.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Increased Global Prevalence of Cardiovascular Diseases |
|
|
Regulatory Safety Updates |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Immediate-Release Propranolol Segment Dominate the Global Propranolol Market in 2026?
The immediate-release propranolol segment is expected to hold the largest share of 52% in 2026, due to its ability to deliver a swift therapeutic effect, making it the predominant product type within the global propranolol market. The fast absorption of this form helps in the quick management of acute symptoms in diseases like anxiety attacks, arrhythmias, some emergencies involving the heart where time is vital. The immediate-release formulations are sometimes favored by healthcare practitioners when they need to adjust doses accurately and change their schedules at the individual patient level.
For instance, in November 2025, the UK Department of Health and Scottish Government reported shortages of 80 mg and 160 mg modified-release propranolol capsules, prompting clinicians to switch patients to immediate-release tablets to maintain treatment continuity.
Why Does the 20 to 40 mg Segment Dominate the Global Propranolol Market in 2026?
The 20 to 40 mg segment is expected to hold 45% share in 2026, due to its effective therapeutic window and manageable side effect profile. It is specifically the dosage range that would be most appropriate in the treatment of chronic diseases like hypertension and angina, where it is important to maintain the blood pressure level without unnecessary exposure to hypotension or bradycardia.
It is in this middle dose range that physicians seek to find a perfect balance between clinical effects and patient tolerance during initiation or continuing treatment. In some cases, lower doses, up to 10mg, are not suitable to combat most cardiovascular signs whereas higher doses, 60mg and more are fraught with risk of adverse effects, like fatigue, dizziness, and aggravation of respiratory problems. Consequently, the range between 20 and 40 mg provides clinicians with a safer and a preferred therapeutic starting point or a maintenance dose.
For instance, in Europe, national health systems such as the UK NHS prioritize 20 mg and 40 mg doses in hospital formularies for both immediate- and extended-release propranolol, making them the preferred choice for chronic cardiovascular management due to flexibility in titration and patient compliance.
The Hypertension Segment Dominates the Global Propranolol Market
The hypertension segment is expected to hold the largest share of 30% in 2026, driven by its high prevalence and the established role of propranolol as a foundational antihypertensive therapy. The prevalence of hypertension among adults is one of the most critical health-related issues in the world, which contributes to the popularity of this segment of the target population in the propranolol market. The effectiveness of propranolol in blood pressure reduction and cardiovascular risk factors has entrenched its position as a dependable beta-blocker in antihypertensive management especially in those patients who might require further regulation of cardiac rate and cardiac load.
Impact of API Supply Chain Volatility on the Global Propranolol Market
- Concentration of API Production in Asia Pacific - Most of the manufacturing of propranolol active pharmaceutical ingredient (API) is focused on China and India, and it makes the global supply chain very sensitive to the disruption of the region. The temporary downfall can be caused by regulatory checks, environmental inspections, exportation controls, or logistical setbacks in these nations and this will result in the delayed delivery and price variations to the final companies all over the world.
- Raw Material and Cost Sensitivity- The production of Propranolol API depends on the chemical intermediates like epichlorohydrin among others. Raw material volatility, price or tariff on importation of raw materials directly influence production costs and this may be reflected in variable prices of finished goods and market stability especially where the markets largely rely on imports.
Regional Insights
To learn more about this report, Request Free Sample
North America Propranolol Market Analysis and Trends
The North America region is projected to lead the market with a 35% share in 2026, driven by a well-established healthcare infrastructure combined with significant pharmaceutical industry presence. The region has advantageous regulatory measures and massive government spending on healthcare, which guarantees large-scale supply and prescript of propranolol in cardiovascular and other associated indicators. The existence of major pharmaceutical firms like Pfizer, Mylan (since acquired by Viatris) and Novartis, contributes to keeping a healthy supply chain and constant innovation in the formulations of the drugs including the extended-release and the generic drugs. Also North America is the leader in both production and consumption of propranolol because of the favorable patent laws and the strong intellectual property protection to enable continued investment in R&D. The region is also in favor of trade dynamics, and it has efficient distribution networks to export to other markets.
For instance, propranolol was one of the most dispensed non‑cardioselective beta blockers in the U.S. with approximately 9.7 million prescriptions in 2023, accounting for about 34.9 % of all non‑cardioselective beta‑blocker prescriptions in that category.
(Source- https://pmc.ncbi.nlm.nih.gov/articles/PMC9456064/#:~)
Asia Pacific Propranolol Market Analysis and Trends
The Asia Pacific region is expected to exhibit the fastest growth in the propranolol market contributing 30% share in 2026, fueled by expanding healthcare access, improving regulatory environments, and increasing awareness about cardiovascular diseases. Other countries such as India and China are spending a lot on health infrastructure and encouraging the production of pharmaceuticals within the country. The market penetration has been expedited by government efforts to advance health results and increase the number of insured people. Domestic generic drug manufacturers like Lupin, Sun Pharma and Cipla have been very instrumental in making propranolol more affordable and accessible. Moreover, increasing numbers of middle-class and the increased number of cases of hypertension, arrhythmia, and migraine in this area has increased demand.
Propranolol Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Propranolol Market?
The propranolol market in the U.S. still has a lead because of the high expenditure on healthcare, modernization of medical facilities and the usage of evidence-based interventions. Large companies like Pfizer and Mylan have built strong penetration in the market, both as brand and generic versions propranolol. The approval process of the U.S. Food and Drug Administration (FDA) guarantees the safety and effectiveness of drugs and increases the trust of the physicians and the consumers. Moreover, the propensity towards customized medication and on-going sophistication in drug delivery is an added advantage of enduring demand.
How is Germany Helping in the Growth of the Propranolol Market?
Germany market of propranolol is advantaged by a strong healthcare system, universal coverage and huge state financial support. There are many pharmaceutical firms in the country such as Bayer and Stada Arzneimittel that focus on the high standards of drug manufacturing. The regulatory environment in Germany allows access to branded and generic drugs, which allow it to manage costs without affecting the availability of treatment. German pharmaceutical exports also have an effect on the regional trade, putting Europe in a more advantageous position in the propranolol market.
Key Drivers for the Growth of the India Propranolol Market
India plays an important role in the Asia Pacific region and the propranolol market is growing on the basis of increasing access to healthcare services and a large population of patients with heart diseases. Lupin, Sun Pharma and Cipla, the Indian pharmaceutical giants have developed a position as the world suppliers of cheap generic propranolol to meet the needs of the domestic and international markets. This growth is supported by government programs that encourage low-cost medicines and expansion of healthcare. Moreover, the country has a well-trained pharmaceutical labor force and economically competitive production facilities that keep on attracting investments.
China Propranolol Market Trends
The growing healthcare system and the growing rates of lifestyle related diseases in China are factors that have led to the growth in the demand of propranolol. Domestic firms such as Sino Biopharmaceutical and Harbin Pharmaceutical Group have intensified their output and released competent generic alternatives of the drug. Market dynamics have been enhanced by regulatory reforms that were intended to simplify drug approvals as well as increase quality control. The attention of the government to the modernization of healthcare and the management of chronic diseases also contributes to the growth of the market.
Geography-Wise Prescribing Patterns and Treatment Guidelines for Propranolol
|
Region |
Prescribing Patterns |
Treatment Guidelines |
|
North America (U.S. & Canada) |
Propranolol is widely prescribed for hypertension, angina, arrhythmia, migraine prophylaxis, and performance anxiety; both immediate-release and extended-release formulations are used |
American Heart Association (AHA) and Canadian Cardiovascular Society (CCS) recommend propranolol for chronic hypertension and certain arrhythmias; off-label use for migraine and anxiety is common under physician discretion |
|
Europe (U.K., Germany, France) |
High generic usage in hospital and retail settings; extended-release preferred for patient adherence; migraine prophylaxis also common |
European Society of Cardiology (ESC) guidelines recommend propranolol as first-line beta-blocker for hypertension, arrhythmia, and migraine prevention; prescription influenced by national health system formularies |
|
Asia Pacific (India, China, Japan, Australia) |
Mix of branded and generic propranolol; urban centers see more ER formulation use; rising off-label use for anxiety in younger adults |
National guidelines (e.g., Indian Council of Medical Research, Japanese Circulation Society) prioritize propranolol for cardiovascular diseases; migraine prophylaxis included in Japan; off-label use monitored regionally |
|
Latin America (Brazil, Mexico, Argentina) |
Generic propranolol dominates; dosing often aligned with cost-effectiveness; immediate-release more common than ER due to affordability |
Regional cardiovascular treatment guidelines align with WHO and ESC recommendations; national formularies emphasize generic substitution to improve accessibility |
|
Middle East & Africa |
Increasing prescriptions in urban hospitals; use for hypertension and arrhythmia rising; limited ER adoption due to cost |
Guidelines generally follow ESC or AHA recommendations; local health ministry’s monitor safety and adjust reimbursement lists accordingly |
|
Global Off-Label Use |
Growing prescriptions for performance anxiety, essential tremor, and migraine prophylaxis across multiple regions |
No unified global guideline; physician discretion and local ethical committees guide off-label propranolol use; regulatory agencies advise monitoring for safety and contraindications |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On February 13, 2026, SSY Group Limited announced that it had obtained approval from China’s National Medical Products Administration to register Propranolol Hydrochloride as a bulk drug for cardiovascular indications. The approval strengthens its cardiovascular portfolio and supports expanded production for the formulations market in China, bolstering its competitive position in high-demand chronic disease segments.
- In January 2025, Exicure, Inc. entered into a share purchase agreement with GPCR Therapeutics, Inc., acquiring all issued and outstanding equity of GPCR Therapeutics USA Inc. and integrating its proprietary technology into Exicure’s development portfolio. The transaction included a license and collaboration agreement designed to support the advancement and commercialization of GPCR Therapeutics’ CXCR4 inhibitor technology currently in Phase 2 clinical trials with the U.S. Food and Drug Administration.
- In August 2023, Lupin Limited announced that it had received regulatory approval from Health Canada for its Propranolol Hydrochloride Extended-Release (ER) Capsules in strengths of 60 mg, 80 mg, 120 mg, and 160 mg and has launched the product in the Canadian market.
Top Strategies Followed by Global Propranolol Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players dominate the propranolol market through strong R&D investments, innovation in formulations, strategic partnerships with OEMs, and global expansion into emerging markets to capture growing demand. |
Lupin Limited strengthened its global presence by launching extended-release propranolol capsules in Canada to replace discontinued Inderal LA, combining innovation with market expansion to boost regional adoption and revenue. |
|
Mid-Level Players |
Mid-level propranolol market players compete by offering cost-effective, high-quality products, leveraging strategic collaborations to scale production and expand market reach, and targeting both generic and niche therapeutic segments. |
Zydus Cadila partners with regional manufacturers in India to produce affordable propranolol formulations, balancing quality and cost while expanding distribution in urban and semi-urban markets. |
|
Small-Scale Players |
Small-scale propranolol players focus on niche markets and innovative products, using advanced delivery technologies and digital integration, while forming local partnerships to target specific therapeutic applications and patient segments. |
Hetero Labs’ specialized propranolol formulations for migraine and anxiety in select Indian regions demonstrate how small firms leverage niche focus and strategic alliances to gain market presence. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Propranolol Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 517.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.8% | 2033 Value Projection: | USD 894.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd., AstraZeneca PLC, Apotex Inc., Viatris (Mylan legacy), Novartis AG, Sanofi S.A., Zydus Cadila (Zydus Pharma), Sawai Seiyaku Co. Ltd., AbZ-Pharma GmbH, Towa Yakuhin K.K., Yabang Pharma, Nichi-Iko Pharmaceutical Co. Ltd., Intas Pharmaceuticals Ltd., Lupin Limited, and Sun Pharmaceutical Industries Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Propranolol Market Dynamics
To learn more about this report, Request Free Sample
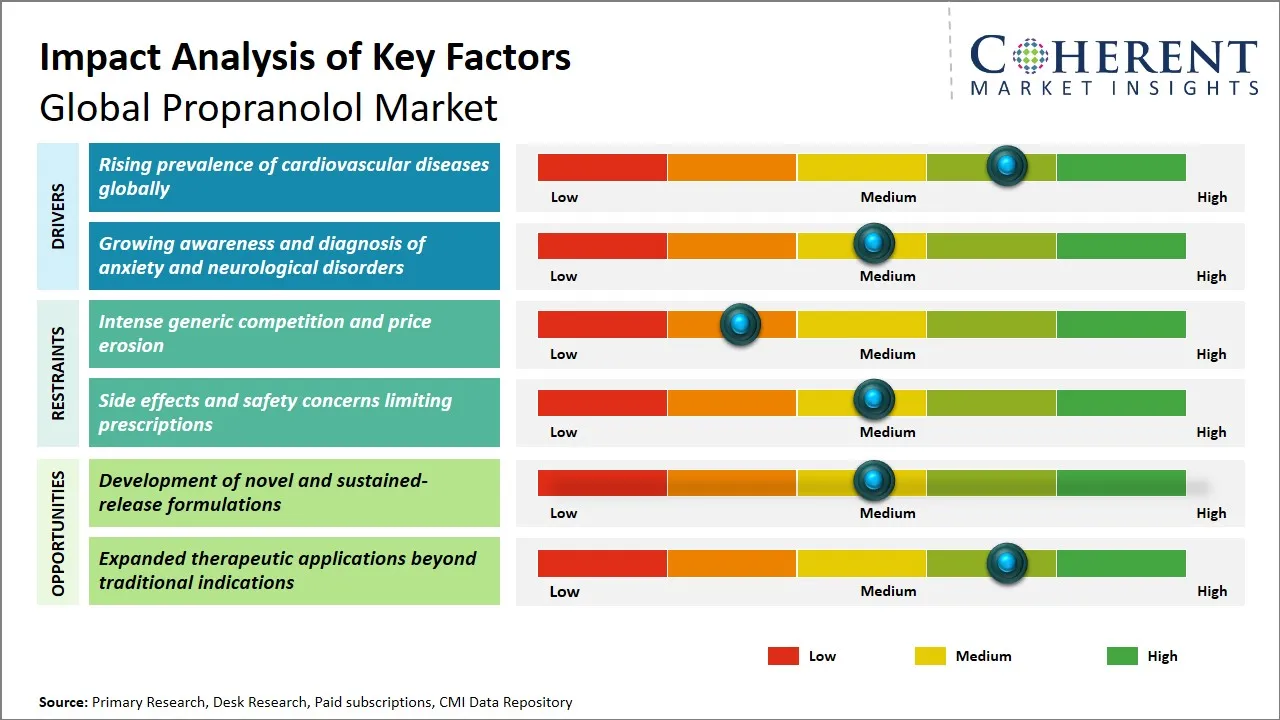
Propranolol Market Driver - Rising Prevalence of Cardiovascular Diseases Globally
The increasingly growing cases of cardiovascular diseases (CVDs) in the world is a major driver of the propranolol drug which is a common prescription beta-blocker with an excellent reputation in treating different heart related diseases. As the concept of urbanization, sedentary lifestyle, poor dietary patterns and higher and higher stress levels are leading to an upsurge of hypertension, arrhythmias, angina and other heart related disorders, the use of beta-blockers such as propranolol are becoming therapeutic options by healthcare providers. The effect of this drug to slow down the heart rate, manage the blood pressure and prevent the complications caused by unusual heart rhythms is what makes this medication an essential factor in the course of cardiovascular treatment.
For instance, according to the World Health Organization (WHO), cardiovascular diseases (CVDs) remain the leading cause of death globally, accounting for approximately 18 million deaths in 2023, with hypertension affecting over 1.28 billion adults worldwide. This high disease burden directly increases demand for beta-blockers like propranolol for both treatment and long-term management, particularly in high-population regions such as China, India, and the U.S.
(Source-https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds))
Propranolol Market Opportunity - Development of Novel and Sustained-Release Formulations
A large opportunity in the global market of propranolol has been offered with the formulation of new and sustained-release preparations, with the aim of increasing the therapeutic efficacy and patient compliance. Conventional propranolol preparations typically have to be administered in several doses a day and this may represent a problem of compliance particularly in older patients or those taking numerous medications. Advancements in technologies of drug delivery (e.g., extended-release pills, transdermal patches, new oral delivery systems) have the potential to deliver more stable plasma drug levels, fewer side effects and better symptom management of diseases like hypertension, angina and anxiety disorders. In addition, sustained-release preparations may reduce the effect of the peaks and troughs, which would result in improved safety profiles due to the reduction in the risks of adverse cardiovascular events linked to the rapid absorption.
For instance, Sun Pharmaceutical Industries Ltd. launched a propranolol sustained‑release tablet in India with details such as once‑daily dosing for hypertension and migraine prevention. Sun Pharma’s official India product listing shows standard propranolol immediate‑release tablets and timed‑release capsules marketed under brand names like BETACAP and BETACAP TR with different strengths.
Analyst Opinion (Expert Opinion)
- The propranolol market is experiencing a steady increase because of the increasing incidence of cardiovascular diseases, the augmentation of its use in the management of migraine and anxiety, and recently the use of long-release formulations which enhance compliance. The opportunities are in the off-label usage and in the growing markets in Asia Pacific and Latin America, where the challenges are the variability of API supply, generics pressure on the prices and regulatory compliance.
- The protocols of treatment and formulations have been emphasized by recent conferences such as the World Congress on Cardiology, International Conference on Cardiovascular Medicine, and Asia Pacific Pharma Summit, etc. Other strategic projects (like an extended-release launch of Lupin in Canada and API approval in China by SSY Group) and government-based programs on cardiovascular care still influence market forces and market expansion potential in a region.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Immediate-Release Propranolol
- Extended-Release (Sustained-Release) Propranolol
- Injectable Propranolol
- Dosage Strength Insights (Revenue, USD Mn, 2021 - 2033)
- 20 to 40 mg
- 60 to 80 mg
- Up to 10 mg
- 100 mg and above
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Hypertension
- Angina Pectoris
- Arrhythmia
- Migraine Prophylaxis
- Anxiety Disorders (Performance/Social)
- Hyperthyroidism / Thyrotoxicosis
- Essential Tremor
- Infantile Hemangioma
- Post-Myocardial Infarction Management
- Other Cardiovascular Indications
- Patient Demographics Insights (Revenue, USD Mn, 2021 - 2033)
- Pediatric
- Adult
- Geriatric
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Government & Institutional Supply
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Specialty Clinics (Cardiology, Neurology, Endocrinology)
- Ambulatory Surgical Centers
- Homecare Settings
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Teva Pharmaceutical Industries Ltd.
- AstraZeneca PLC
- Apotex Inc.
- Viatris (Mylan legacy)
- Novartis AG
- Sanofi S.A.
- Zydus Cadila (Zydus Pharma)
- Sawai Seiyaku Co. Ltd.
- AbZ-Pharma GmbH
- Towa Yakuhin K.K.
- Yabang Pharma
- Nichi-Iko Pharmaceutical Co. Ltd.
- Intas Pharmaceuticals Ltd.
- Lupin Limited
- Sun Pharmaceutical Industries Ltd.
Sources
Primary Research Interviews
Industry Stakeholders
- Cardiologists
- Pharmacists
- Hospital formulary managers
- Pharmaceutical R&D heads
- Clinical trial investigators
- Regulatory affairs specialists
End Users
- Patients with hypertension
- Patients with migraine disorders
- Patients with arrhythmia
- Patients with essential tremor
- Patients using propranolol for anxiety
- Caregivers of chronic cardiovascular patients
Government and International Databases
- World Health Organization (WHO)
- U.S. Food and Drug Administration (FDA)
- European Medicines Agency (EMA)
- Centers for Disease Control and Prevention (CDC)
- National Health Service (NHS) UK
- Indian Council of Medical Research (ICMR)
Trade Publications
- Pharmaceutical Journal
- The Lancet – Pharmacy Section
- Drug Development & Delivery
- BioPharm International
- PharmaTimes
- MedPage Today
Academic Journals
- Journal of Cardiovascular Pharmacology
- American Journal of Hypertension
- British Journal of Clinical Pharmacology
- European Heart Journal
- Journal of Clinical Pharmacy and Therapeutics
- Pharmacological Research
Reputable Newspapers
- The New York Times – Health Section
- The Guardian – Health & Science
- The Washington Post – Health Section
- Financial Times – Healthcare Coverage
- BBC News – Health
- Reuters Health
Industry Associations
- American Heart Association (AHA)
- European Society of Cardiology (ESC)
- International Society of Hypertension (ISH)
- World Federation of Societies of Anaesthesiologists (WFSA)
- British Pharmacological Society (BPS)
- International Pharmaceutical Federation (FIP)
Public Domain Resources
- ClinicalTrials.gov
- WHO Global Health Observatory
- OpenFDA
- NIH National Library of Medicine
- OECD Health Data
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients