U.S. Cardiac Implantable Electronic Device Market Size and Forecast – 2026-2033
The U.S. cardiac implantable electronic device market is expected to grow from USD 12,294.4 Mn in 2026 to USD 20,643.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.7%. The U.S. cardiac implantable electronic device market is poised for significant expansion, fueled by the high and growing burden of cardiovascular diseases and arrhythmias.
According to the Centers for Disease Control and Prevention (CDC), cardiovascular disease remains the leading cause of death in the U.S., accounting for 919,032 deaths in 2023, or approximately 1 in every 3 deaths. Additionally, the economic burden is substantial, with healthcare services and medications for heart disease costing over USD 168 billion between 2021 and 2022.
(Source: Centers for Disease Control and Prevention)
Key Takeaways of the U.S. Cardiac Implantable Electronic Device Market
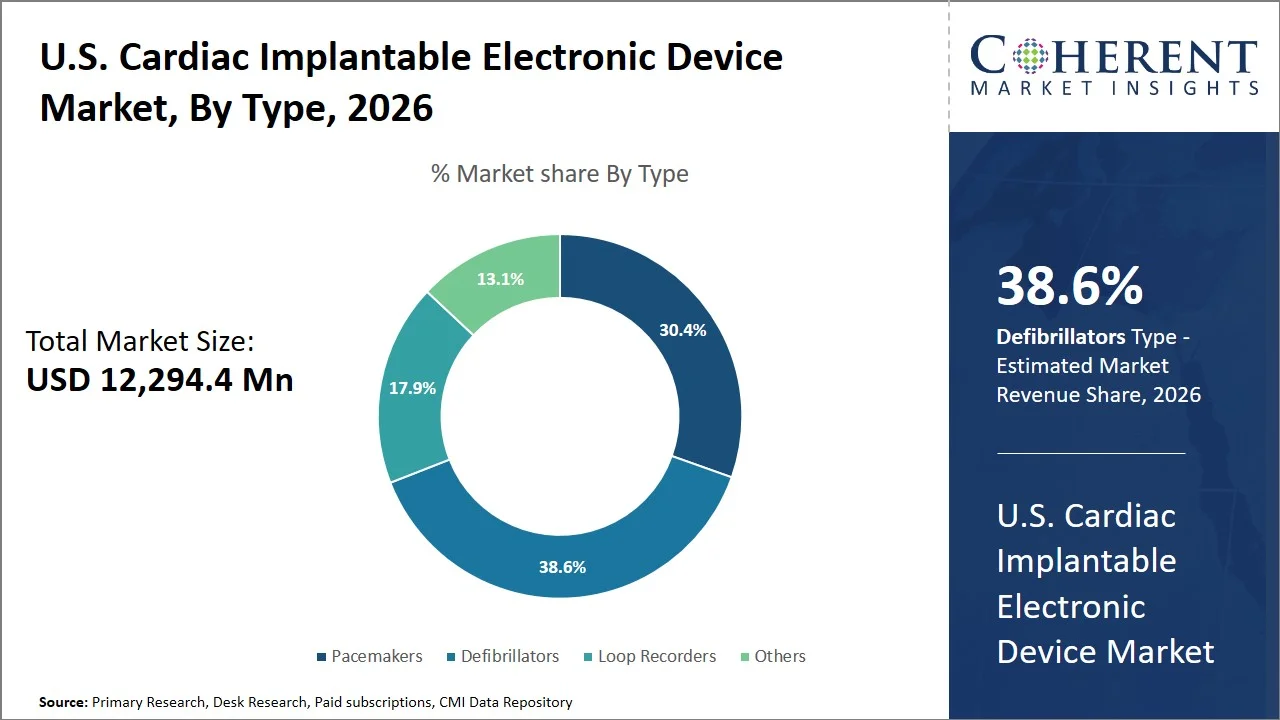
- Defibrillators are projected to hold 38.6% of the U.S. cardiac implantable electronic device market share in 2026, making it the dominant type segment, attributed to the high prevalence of sudden cardiac arrest (SCA) cases in the country. For instance, according to the CDC, more than 356,000 people have an out-of-hospital cardiac arrest in the U.S. every year, and about 60% to 80% of them die before reaching the hospital. (Source: CDC)
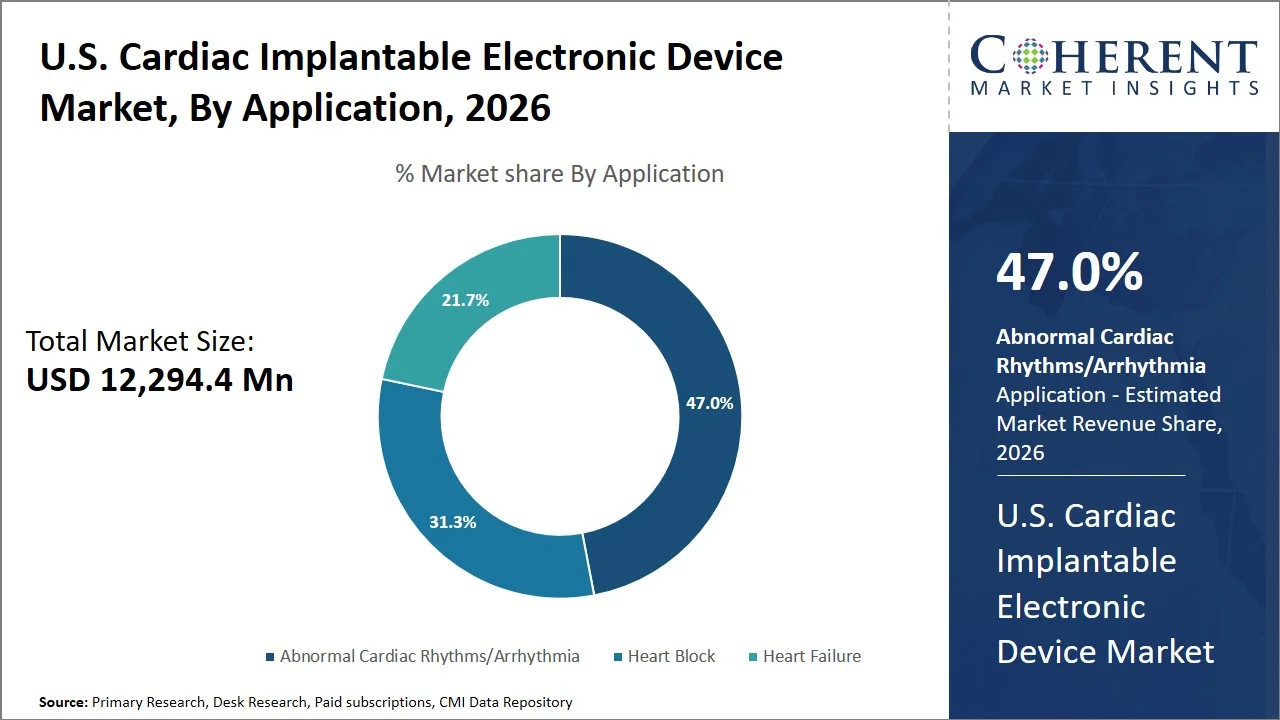
- Abnormal Cardiac Rhythms/Arrhythmia is projected to hold 47.0% of the U.S. cardiac implantable electronic device market share in 2026, making it the dominant application segment, facilitated by the increased incidence of atrial fibrillation and other associated diseases. For instance, according to the CDC, in 2021, atrial fibrillation (AFib) was mentioned on 232,030 death certificates and was the underlying cause of death in 28,037 of those deaths. Furthermore, it is estimated that 12.1 million people in the U.S. will have atrial fibrillation (AFib) by 2050. (Source: CDC)
- Hospitals are projected to hold 41.9% of the U.S. cardiac implantable electronic device market share in 2026, making it the dominant end user segment, owing to the availability of high-quality cardiac care infrastructure. For instance, according to the American Hospital Association, there are more than 6,100 hospitals in the U.S. that have electrophysiology laboratories and cardiac surgical units that can carry out complicated operations for device implantation. The extensive network of hospitals remains fundamental to the application of cardiac implantable electronic device in the U.S. (Source: American Hospital Association)
- Expansion of Leadless and Extravascular Device Technologies: The rising trend of adopting leadless cardiac pacemakers and extravascular cardiac implantable electronic device (EV-ICDs) is providing an opportunity for the U.S. cardiac implantable electronic device market growth. This technology has been designed without the use of transvenous leads, thereby drastically lowering risks of infections, lead displacement, and vessel damage. Increasing inclination towards these advanced devices due to their safety and minimal invasiveness, particularly for elderly and high-risk individuals, is driving their demand.
- Growth of Remote Monitoring and AI-Enabled Cardiac Management: The combination of remote monitoring technologies and AI analytics is becoming an important area in the U.S. cardiac implantable electronic device market. Modern devices have made it possible for patients to transmit continuous streams of data, which allows timely detection of potential issues, such as arrhythmias and problems with the devices themselves, thus avoiding unnecessary hospitalizations. This development is bolstered by the growing presence of digital health technologies and their use within the context of remote cardiac care, including reimbursement opportunities in the U.S.
Why Do Defibrillators Dominate the U.S. Cardiac Implantable Electronic Device Market?
Defibrillators are projected to hold a market share of 38.6% in 2026, because of the proven clinical effectiveness of the devices in lowering mortality among high-risk patients and the strong alignment between U.S. clinical practices and the high volume of procedures performed. According to the National Heart, Lung, and Blood Institute, implantable cardioverter defibrillators have been shown to reduce the probability of sudden cardiac death among patients suffering from heart failure and ejection fraction reduction. Supporting this, a National Heart, Lung, and Blood Institute-funded analysis of the SCD-HeFT trial reported that patients receiving implantable defibrillators experienced a 13% reduction in overall mortality compared to standard therapy (Source: National Heart, Lung, and Blood Institute). The devices have, therefore, been widely adopted in medical practice.
Additionally, innovations, such as the creation of defibrillator systems without intravascular leads that reduce the likelihood of complications from conventional leads, have reinforced physicians and patients’ preference for this device. For example, EMBLEM subcutaneous implantable defibrillator system provides therapy by being implanted beneath the skin, not requiring leads that enter the heart or blood vessels, thus reducing the risk of infection and failure associated with leads while ensuring defibrillation therapy remains effective. (Source: Boston Scientific)
Why is Abnormal Cardiac Rhythms/Arrhythmia a Key Application of Cardiac Implantable Electronic Device?
To learn more about this report, Request Free Sample
Abnormal Cardiac Rhythms/Arrhythmia is projected to hold 47.0% of the market share in 2026, due to the considerable clinical emphasis on its use and related risks that require continuous management. For instance, conditions such as ventricular tachycardia and atrial fibrillation can endanger patients' health due to the increased risks of suffering from heart-related complications, including stroke, heart failure, or even sudden death. As a result, rhythm management through the use of implantable devices should be considered an ongoing procedure. In line with recommendations from the American College of Cardiology, the use of implantable devices, including defibrillators or pacemakers, is considered crucial in the treatment of high-risk arrhythmias along with poor ejection fractions or conductive defects. (Source: American College of Cardiology)
Moreover, technological development for devices, such as continuous monitoring and therapy, has improved the efficacy of treating arrhythmias. For example, gallant implantable cardioverter defibrillator incorporates the integration of Bluetooth technology with remote monitoring systems, facilitating the continuous relay of heart information and detection of arrhythmias to facilitate prompt clinical action. (Source: Abbott)
Hospitals Segment Dominates the U.S. Cardiac Implantable Electronic Device Market
The hospitals segment is expected to hold 41.9% of the U.S. cardiac implantable electronic device market share in 2026, attributed to their central role in conducting complex cardiac procedures and managing high-risk patients requiring continuous monitoring and intervention. The implantation of pacemakers and cardiac implantable electronic device requires special equipment like electrophysiology lab, surgery facility, and intensive care unit, which can only be provided by hospitals.
The U.S. Food and Drug Administration categorizes implantable heart devices like pacemakers and implantable cardioverter defibrillators as Class III medical devices, necessitating Premarket Approval (PMA), the most rigorous U.S. FDA process for regulatory approval involving clinical trials and hospital assessments prior to marketing (Source: FDA). This requirement ensures that hospitals will continue to be the key venues where medical devices will be implanted and evaluated, since only hospitals have all the resources needed to ensure regulatory compliance.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Shift: U.S. FDA Support for Mobile-Based Remote Monitoring in Cardiac Devices |
|
|
Regulatory Shift: Centers for Medicare & Medicaid Services (CMS) Transitional Coverage for Emerging Cardiac Technologies (TCET Pathway) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Cardiac Implantable Electronic Device Market Dynamics
To learn more about this report, Request Free Sample
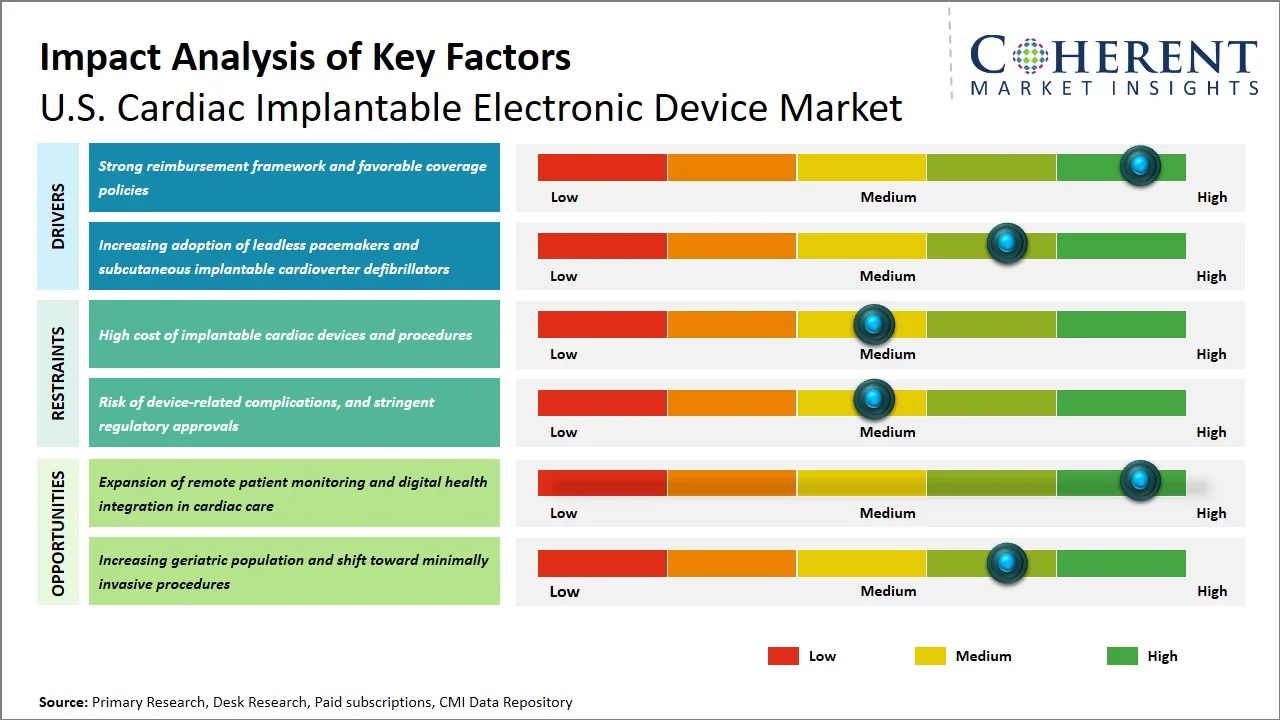
Market Drivers
- Strong reimbursement framework and favorable coverage policies: Reimbursement system in the U.S. plays a significant role in making the market for cardiac implantable electronic device successful, since it ensures that access to cardiac implantable electronic device will be available to patients who need these life-saving procedures. Implantable cardioverter defibrillator falls into the category of items included in the National Coverage Determination (NCD) of the Centers for Medicare and Medicaid Services, which confirms that this type of equipment is necessary for certain people. Therefore, reimbursement can be ensured in the population of Medicare beneficiaries, which should be considered as a key target group when promoting cardiac implantable electronic device (Source: gov). Moreover, the Centers for Medicare and Medicaid Services promotes the development of innovative devices by implementing a coverage with evidence development program, which means that new devices are covered conditionally till their efficiency is proved such as leadless pacemakers. (Source: CMS.gov)
- Increasing adoption of leadless pacemakers and subcutaneous implantable cardioverter defibrillators: The use of innovative technologies such as leadless pacemakers and subcutaneous implantable cardioverter defibrillators in the U.S. market has seen an upsurge as there are minimal risks associated with infection, dislodgment, and vasculature damage when compared with transvenous leads. The approval of technologies such as the Micra leadless pacemakers and subcutaneous implantable cardioverter defibrillator by the U.S. FDA has also contributed towards the increasing trend (Source: FDA). Cardiac devices prove to be beneficial for high-risk patients as there are enhanced patient safety measures along with minimal recovery time, making it easier for patients to adhere to the treatment plans. With the continual accumulation of data indicating benefits linked with the technologies, it is expected that there will be a widespread implementation of medical technologies within the U.S. healthcare industry.
Emerging Trends
- Expansion of Outpatient & Ambulatory Implantation Settings: The implantation process for cardiac implantable electronic device is being slowly shifted out of the hospitals and to outpatient facilities through new payment methods by the Centers for Medicare and Medicaid Services. This will reduce costs and hospital stay duration, increase efficiency, and improve the accessibility of the devices across the country.
- Increasing Focus on Device Longevity and Battery Innovation: Companies are developing devices with high battery life and energy savings to reduce the frequency of replacement. This will help reduce healthcare costs, and the U.S. FDA approval will encourage the adoption of such devices among the populace. For instance, the AVEIR VR Leadless Pacemaker that has received approval from the U.S. FDA is specifically engineered to last longer while also being easily retrievable, thus making fewer replacements necessary. (Source: Abbott)
Regulatory & Reimbursement Landscape Shaping the U.S. Cardiac Implantable Electronic Device Market
|
Category |
Policy/Framework |
Description |
Impact on Market |
|
Regulatory Approval |
U.S. Food and Drug Administration Premarket Approval (PMA) Pathway |
High-risk cardiac devices such as implantable defibrillators and pacemakers require stringent PMA approval to ensure safety and efficacy before commercialization. |
Ensures high-quality standards but increases time-to-market for new devices |
|
Reimbursement Coverage |
Centers for Medicare & Medicaid Services National Coverage Determination (NCD) for Implantable Cardioverter Defibrillators |
CMS recognizes implantable defibrillators as “reasonable and necessary” for treating life-threatening arrhythmias, enabling reimbursement under Medicare. |
Strong reimbursement support drives widespread adoption across eligible patient populations |
|
Coverage for Advanced Devices |
CMS Coverage with Evidence Development (CED) – Leadless Pacemakers |
Coverage is provided for innovative devices such as leadless pacemakers under clinical study frameworks, encouraging adoption while collecting real-world evidence. |
Facilitates the early adoption of next-generation technologies while managing clinical risk |
|
Remote Monitoring Reimbursement |
CMS Remote Patient Monitoring (RPM) Codes (99453, 99454, 99457, 99458) |
Enables providers to bill for the continuous monitoring of cardiac data transmitted from implantable devices, including arrhythmia tracking and device diagnostics. |
Accelerates the integration of digital health and increases the utilization of connected CIEDs |
|
Device Surveillance & Follow-up |
Medicare Coverage for ICD Monitoring Services |
Medicare covers both in-person and remote monitoring of implanted defibrillators for device performance and patient condition assessment. |
Supports long-term device usage and recurring revenue streams through follow-up services |
|
Recent Policy Development (2026) |
CMS–FDA RAPID Coverage Pathway |
A new initiative to align U.S. FDA approval with Medicare coverage decisions, reducing reimbursement timelines to 60–90 days for breakthrough devices. |
Accelerates the commercialization of innovative CIED technologies and improves market access |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the expansion of remote patient monitoring and digital health integration in cardiac care creating new growth opportunities in the U.S. cardiac implantable electronic device market?
The expansion of remote patient monitoring and digital integration is presenting substantial opportunities for the growth of the U.S. cardiac implantable electronic device market as they facilitate constant, real-time cardiac management beyond conventional clinical environments, which improve patient outcomes as well as reduce the number of hospitalizations. In the U.S., the Centers for Medicare & Medicaid Services has introduced new reimbursement programs for remote patient monitoring to promote the use of cardiac implantable electronic devices through digital integration.
For instance, Remote Patient Monitoring (RPM) reimbursement program (CPT codes 99453, 99454, 99457, 99458) provides support for the development, data transmission, and clinical management of remote patient monitoring for patients wearing connected devices. The CPT codes allow physicians to charge for the ongoing monitoring of vital signs such as cardiac rhythms and functions of the devices themselves. This reimbursement program encourages the use of connected cardiac implantable devices due to the opportunity to receive payments for remote health services. (Source: American College of Physicians, Inc.)
Concurrently, the U.S. Food and Drug Administration has approved a number of remote monitoring technologies for use in device-integrated platforms, including apps designed to transmit secure information about patients wearing pacemakers. For instance, myMerlinPulse app was launched by Abbott, offering remote monitoring capabilities that transmit patients’ data securely to their physicians on the go. The mobile-based approach makes it possible to monitor the cardiac rhythms of patients in real time and take prompt action in case of any deviations without requiring them to visit medical facilities often (Source: Abbott). These trends demonstrate the increasing move towards interconnected, patient-oriented ecosystems of cardiovascular care, thus creating an impetus for a greater demand for digitally-enabled cardiac implantable electronic devices in the U.S.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 23, 2026, BIOTRONIK received the U.S. FDA approval for its next generation devices – the Acticor Sky ICD and Rivacor Sky CRT-D, which provide better longevity, MRI compatibility, and improved monitoring features. This approval will further reinforce BIOTRONIK’s product range in the U.S. and is indicative of innovations being made in cardiac rhythm management technologies.
- On April 23, 2026, Philips launched the Bridge Plus Occlusion Balloon that is used to deal with superior vena cava (SVC) tears while extracting leads from patients. The device makes it safer to remove leads for patients who have their CIED leads removed or replaced. Such developments make CIEDs better as they help solve the problems that arise during their management. This should improve implantable cardiac device management in the future.
Competitive Landscape
The U.S. cardiac implantable electronic device market is extremely competitive due to constant innovations and high demand for cardiac rhythm management solutions. The main market players include Medtronic, Abbott, Boston Scientific, and BIOTRONIK that dominate the market owing to product innovation and technology used by these players. The current trend towards development in the market is making leadless devices, devices compatible with MRI scans, and increased battery life in order to ensure maximum effectiveness and better results for patients. Key focus areas include:
- Development of leadless and minimally invasive cardiac implantable devices
- Integration of remote patient monitoring and digital health ecosystems
- Use of artificial intelligence for predictive cardiac care and device optimization
- Strengthening reimbursement positioning through clinical evidence and outcomes data
Market Report Scope
U.S. Cardiac Implantable Electronic Device Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12,294.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.7% | 2033 Value Projection: | USD 20,643.6 Mn |
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Abbott Laboratories, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, LivaNova PLC, MicroPort Scientific Corporation, Koninklijke Philips N.V., GE Healthcare Technologies Inc., Nihon Kohden Corporation, and ZOLL Medical Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The further trajectory of the U.S. cardiac implantable electronic device market would involve going for smaller, leadless and connected devices. Advancements within the field of AI diagnostics and increased battery life would see the transformation from devices that are only used in reacting to situations into devices of proactive management of heart conditions. Rising trend towards minimally invasive procedures and the use of outpatient facilities would affect future development too.
- The most appealing avenue for growth in this industry is the development of leadless pacemakers used in the treatment of arrhythmias in the U.S., fuelled by an increase in the incidence of cardiac rhythm abnormalities. As a result of an increase in the incidence of atrial fibrillation and other rhythm problems, along with a necessity to minimize the number of complications caused by traditional transvenous leads, there will be an increased demand for leadless systems.
- Indeed, the focus of market players should be put on developing the next generation of devices with remote monitoring and artificial intelligence-based analytics. The key to success lies in close cooperation between the stakeholders mentioned above in building a holistic cardiac care ecosystem. Additionally, it is crucial for manufacturers to make their devices affordable for patients and conduct clinical studies so as to ensure proper reimbursement of expenses.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Pacemakers
- Defibrillators
- Loop Recorders
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Abnormal Cardiac Rhythms/Arrhythmia
- Heart Block
- Heart Failure
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Centers
- Others
- Key Players Insights
- Medtronic
- Abbott Laboratories
- Boston Scientific Corporation
- BIOTRONIK SE & Co. KG
- LivaNova PLC
- MicroPort Scientific Corporation
- Koninklijke Philips N.V.
- GE Healthcare Technologies Inc.
- Nihon Kohden Corporation
- ZOLL Medical Corporation
Sources
Primary Research Interviews
- Chief of Cardiology / Electrophysiologists – CIED implantation procedures (U.S. hospitals & cardiac centers)
- Director – Cardiac Catheterization Labs / Electrophysiology Labs (U.S. hospital networks)
- Clinical Specialists – Cardiac rhythm management devices (CIED manufacturers)
- Ambulatory Surgical Center (ASC) Administrators – outpatient cardiac device implantation trends
- Biomedical Engineers – device performance, monitoring systems, and post-implant care
Stakeholders
- Cardiac Implantable Electronic Device Providers: (e.g., Medtronic, Abbott, Boston Scientific, BIOTRONIK)
- End-use Sectors
- Hospitals (tertiary care & specialized cardiac centers)
- Ambulatory Surgical Centers (ASCs)
- Specialty cardiac clinics / electrophysiology centers
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – approval pathways (PMA/510(k)), safety monitoring, and post-market surveillance for pacemakers, ICDs, and CRT devices, Centers for Medicare & Medicaid Services (CMS) – reimbursement policies, National Coverage Determinations (NCDs), and ASC payment frameworks for CIED procedures, American College of Cardiology (ACC) – clinical guidelines, registries (e.g., NCDR ICD Registry), and best practices for cardiac device implantation, Heart Rhythm Society (HRS) – electrophysiology guidelines, device programming standards, and clinical consensus statements, Centers for Disease Control and Prevention (CDC) – cardiovascular disease burden, arrhythmia prevalence, and mortality statistics influencing device demand, National Institutes of Health (NIH) – funding and research initiatives in cardiac rhythm management and implantable technologies
Databases
- Centers for Medicare & Medicaid Services (CMS) – procedure volumes, reimbursement, and coverage decisions for cardiac devices
- Healthcare Cost and Utilization Project (HCUP) – cardiovascular hospitalization and procedure data
- National Center for Health Statistics (NCHS) – cardiac disease prevalence and mortality trends
- ClinicalTrials.gov – ongoing trials on pacemakers, ICDs, CRT devices
- FDA MAUDE Database – device safety, adverse events, and performance reports
Magazines
- FDA Medical Device Safety Communications – recalls and safety alerts for pacemakers and ICDs
- NIH Research Matters – innovations in cardiac implantable technologies
- AHRQ Patient Safety Network – outcomes and safety of cardiac implant procedures
- Cardiology Today / MedTech Dive – device innovation and market trends
Journals
- Journal of the American College of Cardiology (JACC) – clinical outcomes and device efficacy
- Circulation (American Heart Association) – cardiovascular disease burden and device usage
- Heart Rhythm Journal – electrophysiology and CIED advancements
- Europace – cardiac rhythm management and implantable device studies
Newspapers
- U.S. FDA press announcements – approvals and recalls of cardiac implantable devices
- NIH news releases – funding and research developments in cardiac technologies
- HHS press releases – healthcare policy and device adoption trends
- Major hospital system announcements (e.g., Mayo Clinic, Cleveland Clinic) on electrophysiology advancements
Associations
- American College of Cardiology (ACC)
- Heart Rhythm Society (HRS)
- American Heart Association (AHA)
- Medical Device Innovation Consortium (MDIC)
Public Domain Sources
- U.S. Food and Drug Administration (FDA) – device approvals, regulatory pathways, and safety monitoring
- Centers for Medicare & Medicaid Services (CMS) – reimbursement frameworks and national coverage decisions
- Centers for Disease Control and Prevention (CDC) – cardiovascular disease burden and procedure trends
- National Institutes of Health (NIH) – cardiac research funding and innovation initiatives
- Agency for Healthcare Research and Quality (AHRQ) – patient safety and procedural outcomes
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients