U.S. Sterility Testing Market Size and Forecast – 2026-2033
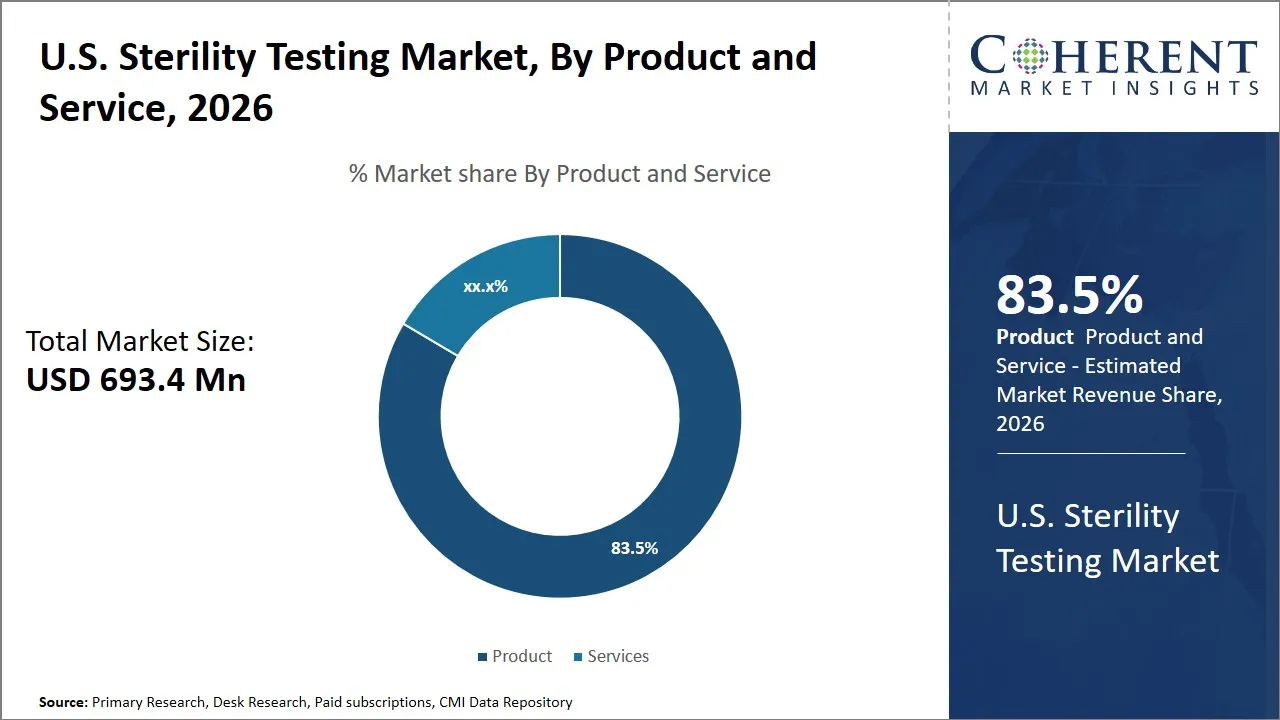
The U.S. sterility testing market is estimated to be valued at USD 693.4 Mn in 2026 and is expected to reach USD 1,120.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 12% from 2026 to 2033. This steady growth reflects the increasing emphasis on quality control and compliance in pharmaceutical and biotechnology sectors, ensuring the safety and efficacy of sterile products.
Key Takeaways of the U.S. Sterility Testing Market
- Product segment is expected to lead the U.S. sterility testing market, capturing 83.5% share in 2026.
- The membrane filtration segment is estimated to represent 33.2% of the market share in 2026.
- The outsource segment is projected to dominate with 53.2% of the U.S. sterility testing market share in 2026.
Market Overview
- In the U.S., sterility testing demand is fundamentally driven by FDA CGMP expectations for sterile drugs and biologics, including written procedures to prevent microbial contamination and adherence to aseptic-processing controls.
- Rising U.S. production of cell therapies, gene therapies, and other short-shelf-life products is increasing demand for faster contamination detection methods that work within compressed release timelines
- Growing adoption of rapid microbiological methods is accelerating market expansion as labs and manufacturers move beyond traditional long-incubation sterility workflows.
- Ongoing U.S. FDA enforcement around deficient sterility validation and batch-release controls is pushing manufacturers to invest more in validated testing, remediation, and external lab support.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
FDA tightens oversight of 503B outsourcing facilities in the U.S. |
|
|
USP advances compendial rapid sterility pathways for short-life products |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Product Segment Dominate the U.S. Sterility Testing Market in 2026?
The product segment is expected to hold the largest share of 83.5% in 2026, owing to multiple critical factors. First of all, the growing strictness of regulatory requirements made by the agencies that handle the sterility testing products, which include the U.S. FDA and the USP, has led to the escalation of the requirement of accurate and dependable sterility testing products. These laws compel pharmaceutical and biotech firms to make their products microbial contamination-free, and they have gone further to drive the need to have sophisticated kits, reagents, and instruments that can offer reliable sterility assessment.
In addition, the high rate of development of testing technologies has helped in dominating the products in this segment. More effective detection of contaminants is possible where innovations in reagent formulations and instrument sensitivity are associated with less turnaround times and more reliability. Automation and improved detection systems incorporated in instruments have also helped in increasing the consistency of the processes and this is essential in a large-scale manufacturing environment. These are technological improvement that help the companies to balance the two-fold task of ensuring compliance and maximizing operational efficiency.
For instance, in March 2025, Nelson Labs introduced a rapid sterility testing service for medical device and pharmaceutical products, designed to deliver results in as little as 6 days, improving turnaround versus traditional methods and supporting faster product release workflows.
(Source- Nelson Labs)
Why Does the Membrane Filtration Segment Dominate the U.S. Sterility Testing Market in 2026?
The membrane filtration segment is projected to capture 33.2% share in 2026, due to its accuracy, versatility, and adaptability to a variety of products. It is the popular method of membrane filtration that is especially useful to readily remove the microorganisms in liquid samples without affecting the viability, the necessary requirement to obtain the validity of sterility. The technique is particularly appropriate when you want to test large volumes of injectable solutions and other pharmaceutical liquids when the direct inoculation method is not so sensitive or labor intensive.
Its method of filtration of samples by force through a membrane and then letting the sample grow by growing it in the right media is more effective in contamination detection than other conventional methods. This accuracy satisfies the rising sterility requirements of injectable medications and ophthalmic remedies, in which the risk of contamination poses severe health consequences.
For instance, in July 2024, Rapid Micro Biosystems announced that its Growth Direct Rapid Sterility System, a membrane filtration-based test, demonstrated rapid and reliable detection across a broad challenge-organism panel, including slow-growing organisms such as C. acnes. The update highlighted the system’s ability to support faster sterility release workflows, with detection in as little as 12 hours and final results in 1–3 days.
(Source - Rapid Micro Biosystems)
Outsource Segment Dominates the U.S. Sterility Testing Market
Outsource segment is expected to capture the highest share of 53.2% in 2026, owing largely to the operational and economic advantages it offers pharmaceutical and biotechnology companies. Outsourcing enables the company to channel internal resources in their core business activities of developing and commercializing drugs, and appropriately depend on contract laboratories with the state-of-the-art equipment and well-trained personnel whose sole responsibility is sterility testing.
Regulatory and Compliance Landscape in the U.S. Sterility Testing Market
- One of the key players in the U.S. sterility testing market is regulatory and compliance activity since the U.S. FDA still holds stringent CGMP standards on sterile drugs, biologics and aseptic manufacturing. The recent warning letters and inspection measures demonstrate that the regulators continue to be concerned with the control of contamination, approved sterilization procedures, best clean room practices, and consistent sterility guarantees, which maintain high testing demand among manufacturers, CDMOs and compounding pharmacies.
- Simultaneously, the compliance levels are changing to more rapid and more validated approaches. The U.S. FDA has solidified the necessity of validated sterility testing, in line with the USP requirements, and USP revised guidance on short-life product extends the use of rapid methods of sterility testing. This is driving the market towards increased use of money in method validation, fast microbiology platforms and superior quality documentation.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In October 2025, Lonza signed an agreement to acquire Redberry SAS, a France-based company specializing in rapid microbiology testing solutions utilizing solid-phase cytometry technology. The acquisition strengthens Lonza’s bioscience testing portfolio and enhances its rapid sterility testing capabilities, supporting faster microbial detection and improved quality control solutions for pharmaceutical and biotech manufacturers.
- In October 2025, Sartorius AG announced the launch of the Cyclus dPCR Tool Box Bacteria Fungi, a digital PCR-based rapid sterility testing solution designed to enable fast and highly sensitive detection of bacterial and fungal contamination in therapeutic products. The platform supports accelerated microbial testing workflows for biopharmaceutical manufacturers, enhancing quality control efficiency and reducing time to product release in sterile production environments.
- In September 2025, Nelson Labs announced the launch of RapidCert Biological Indicator (BI) sterility testing, a rapid solution designed to significantly reduce incubation time to approximately 48 hours compared to traditional sterility testing methods. The new offering accelerates lot release and sterilization validation processes for medical device and pharmaceutical manufacturers, improving turnaround time while maintaining compliance with regulatory sterility assurance standards.
Supply Chain and Reagent Utilization Trends in the U.S. Sterility Testing Market
- Purchases of culture media, membrane-filtration consumables, sterile containers and filtration-related hardware are also frequent purchases in U.S. sterility testing supply chains due to the necessity of sterile drug and biologic manufacturing occur under highly controlled aseptic conditions. The U.S. FDA aseptic-processing directive continues the quality of suppliers, reliability of consumables, and quality control of contamination as a key aspect in the purchase decisions of the labs and their manufacturers.
- The use of reagents is currently dividing into the old-time media-based testing and high-speed molecular work flows. The traditional testing remains relying on standardized media and filtration supplies, as the emerging demanding trends are shifting to the use of qPCR and dPCR-based sterility kits to support advanced therapies and short shelf-life products to form a two-track market regarding both conventional reagents and quick testing systems.
Top Strategies Followed by U.S. Sterility Testing Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders use strong R&D budgets to develop faster, more accurate, and regulation-ready sterility testing solutions. They also strengthen their position through partnerships with pharma companies, OEMs, and technology providers, while expanding distribution and regional presence to capture demand across biotech hubs, CDMOs, and research centers. |
Nelson Labs’ RapidCert launch is a practical example of a market leader using innovation to strengthen competitive advantage by offering faster turnaround, stronger regulatory alignment, and higher-value testing support to sterile-product manufacturers. |
|
Mid-Level Players |
Mid-level players in the U.S. sterility testing market compete by offering reliable but more cost-effective solutions for smaller pharma companies, CROs, CDMOs, and healthcare labs that cannot justify premium-priced systems. Their strategy is usually to keep portfolios focused, control operating costs, and use partnerships with technology providers or distributors to widen reach without making the same capital-heavy bets as market leaders. |
Rapid Micro Biosystems is a good example of this model. In February 2025, it signed a global distribution and collaboration agreement with MilliporeSigma to expand sales of its Growth Direct systems and consumables, showing how a mid-tier player can scale market access and strengthen commercialization through partnership rather than pure size. |
|
Small-Scale Players |
Small-scale players in the U.S. sterility testing market usually compete through niche innovation rather than scale. They focus on specialized rapid-testing technologies, automation, digital data handling, or highly targeted contamination-detection tools for biotech firms, cell therapy labs, and specialized clinical or research settings. |
Redberry is a clear example of this model. Before being acquired by Lonza in October 2025, Redberry had built a niche position around rapid microbiology testing solutions including its Red One platform, showing how smaller innovators can gain traction by solving speed and sterility-assurance gaps in regulated pharma workflows. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Sterility Testing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 693.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12% | 2033 Value Projection: | USD 1,120.8 Mn |
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc, Charles River Laboratories International Inc, Nelson Laboratories LLC, SGS SA, Pace Analytical Services LLC, STERIS plc, Boston Analytical Inc, Microbac Laboratories Inc, North American Science Associates Inc, Gibraltar Laboratories, Eurofins Scientific, Merck KGaA, WuXi AppTec, bioMérieux SA, and Toxikon Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Sterility Testing Market Dynamics
To learn more about this report, Request Free Sample
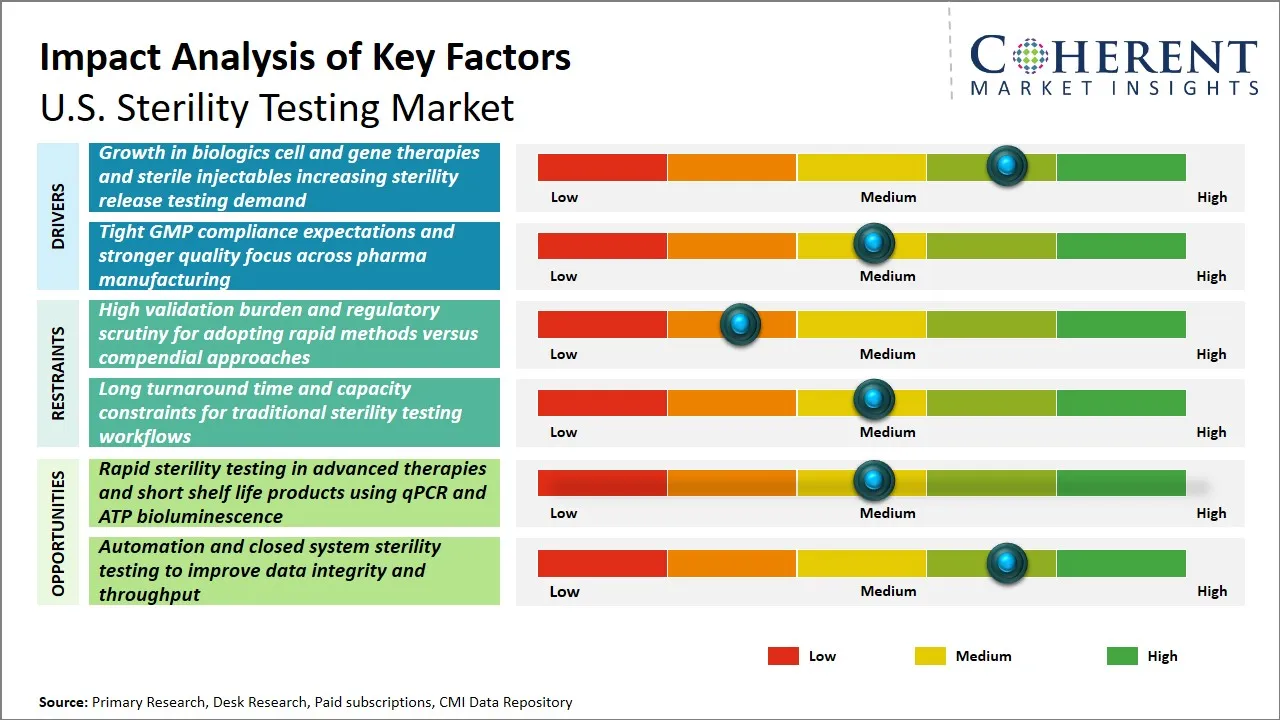
U.S. Sterility Testing Market Driver - Growth in Biologics Cell and Gene Therapies and Sterile Injectables Increasing Sterility Release Testing Demand
The increase in the number of biologics, cell, and gene therapies has led to the growth in demand of sterility release testing. The recent accelerated expansion in biologics, cell, and gene therapy and the rising use of sterile injectable drugs is greatly driving the need of sterility testing in the U.S. pharmaceutical and biotech industries. Such sophisticated therapies need a quality control system to guarantee the safety of the patients since contamination threat may cause serious side effects. The sterility release testing is important to ensure that these products are not contaminated with any microbes before they find their way to the market.
For instance, in April 2025, at its 2025 Rapid Microbial Testing Methods Workshop, NIST focused on technological advances in rapid sterility testing for advanced therapy products, reflecting growing demand for faster microbial release testing as cell and gene therapies expand in the U.S. The event also emphasized interlaboratory studies and standards work to improve confidence in rapid testing methods for these time-sensitive products.
(Source- NIST)
U.S. Sterility Testing Market Opportunity - Rapid Sterility Testing in Advanced Therapies and Short Shelf-Life Products Using qPCR and ATP Bioluminescence
The U.S sterility testing market has a big opportunity with the introduction of fast sterility testing methods especially considering the case of high-tech therapies and products that have few days shelf life. Conventional methods of sterility testing (membrane filtration, direct inoculation) can also take a long time to incubate (as long as 14 days) that is not practical when dealing with time-sensitive products (cell and gene therapies, personalized medicines, and some biologics). Rapid testing methods, such as quantitative Polymerase Chain Reaction (qPCR) and ATP bioluminescence assays, can be seen as a disruptive technology and offer a solution to this issue in that they cut down drastically on the time needed to detect microbial contamination, qPCR being able to amplify and detect microbial DNA in just a few hours, and ATP bioluminescense in real-time by detecting the presence of adenosine triphosphate as a sign of viable microbial contamination. Not only do these approaches speed up the introduction of a product and lower inventory expenses, but also, they can improve patient safety by enabling a quicker identification of contamination.
For instance, in September 2025, NIST’s published report from its collaborative workshop on rapid microbial testing for advanced therapy products included sessions on amplified ATP bioluminescence for rapid sterility testing and qPCR-based methods for rapid microbial detection. This reflects growing standardization and technical attention around non-traditional sterility methods for time-sensitive therapies.
(Source- NIST)
Analyst Opinion (Expert Opinion)
- One of the trends that is defining the U.S. sterility testing market is that speed is nearly becoming as important as compliance. There is increased demand because biologics, cell therapies and other short life sterile products demand shorter release timeframes when compared to the USP Chapter 72 and the rapid microbial testing activities of NIST are shifting the industry towards more widespread utilization of rapid systems. Validated rapid testing platforms and associated quality support is the largest opportunity, but the key issue is ensuring a regulatory defensibility as firms abandon the old sterility workflows.
- The recent industry activity supports this trend. The 2025 Rapid Microbial Testing Methods Workshop by NIST has facilitated the sharing of new knowledge and the standardization of methods, and commercial commercials like the RapidCert by Nelson Labs in September 2025 demonstrate how the market is moving to more practical, fast methods to achieve sterility assurance. All in all, the market is getting more technical, more specialized and more associated with the production of advanced therapies in the U.S.
Market Segmentation
- Product and Service Insights (Revenue, USD Mn, 2021 - 2033)
- Product
- Kits and Reagents
- Instruments
- Services
- Product
- Test Insights (Revenue, USD Mn, 2021 - 2033)
- Membrane Filtration
- Direct Inoculation
- Container closure integrity testing
- Antimicrobial effectiveness testing
- Rapid micro test
- ATP bioluminescence
- Fluorescent labeling
- Electrical resistance
- Testing Type Insights (Revenue, USD Mn, 2021 - 2033)
- Outsource
- In-house
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital
- Diagnostic Laboratories
- Biopharmaceutical Industries
- Others (Contract Manufacturing Organization, Academics and Research Institutes, etc.)
- Key Players Insights
- Thermo Fisher Scientific Inc
- Charles River Laboratories International Inc
- Nelson Laboratories LLC
- SGS SA
- Pace Analytical Services LLC
- STERIS plc
- Boston Analytical Inc
- Microbac Laboratories Inc
- North American Science Associates Inc
- Gibraltar Laboratories
- Eurofins Scientific
- Merck KGaA
- WuXi AppTec
- bioMérieux SA
- Toxikon Corporation
Sources
Primary Research Interviews
Industry Stakeholders list
- QC Microbiology Heads
- Sterility Assurance Managers
- Aseptic Processing Leads
- QA / GMP Compliance Heads
- CDMO Sterile Manufacturing Directors
- Regulatory Affairs Specialists
End Users list
- Biopharmaceutical Manufacturing Managers
- Cell and Gene Therapy Facility Leads
- Hospital Compounding Pharmacy Directors
- Clinical Microbiology Laboratory Directors
- Academic Research Center Lab Heads
- Medical Device Sterile Operations Managers
Government and International Databases
- U.S. Food and Drug Administration (FDA) Data Dashboard
- FDA Warning Letters Database
- FDA Inspection Classification Database
- ClinicalTrials.gov
- WHO IRIS Repository
Trade Publications
- Pharmaceutical Technology (PharmTech)
- BioProcess International
- American Pharmaceutical Review
- Contract Pharma
- European Pharmaceutical Review
- rapidmicrobiology
Academic Journals
- PDA Journal of Pharmaceutical Science and Technology
- AAPS PharmSciTech
- Journal of Pharmaceutical Sciences
- Pharmaceutical Research
- Journal of Microbiological Methods
- Clinical Microbiology Reviews
Reputable Newspapers
- Reuters
- Associated Press
- The Wall Street Journal
- Financial Times
- The Washington Post
- The New York Times
Industry Associations
- Parenteral Drug Association (PDA)
- International Society for Pharmaceutical Engineering (ISPE)
- American Society for Microbiology (ASM)
- American Association of Pharmaceutical Scientists (AAPS)
- Biotechnology Innovation Organization (BIO)
- American Society of Gene & Cell Therapy (ASGCT)
Public Domain Resources
- Electronic Code of Federal Regulations (eCFR)
- Federal Register
- Regulations.gov
- CDC NHSN Reports
- NIST Rapid Microbial Testing Methods Consortium Publications
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients