Acellular Dermal Matrices Market is estimated to be valued at USD 12.41 Bn in 2026 and is expected to reach USD 27.95 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 12.3% from 2026 to 2033.
Analysts’ Views on Global Acellular Dermal Matrices Market:
Rising developments and technological advancements in acellular dermal matrices can drive the global acellular dermal matrices market growth over the forecast period. Furthermore, continuous research & development (R&D) activities can improve the performance and cost-effectiveness of acellular dermal matrices, and can offer new market future opportunities.
Figure 1. Global Acellular Dermal Matrices Market Share (%), By Origin, 2026
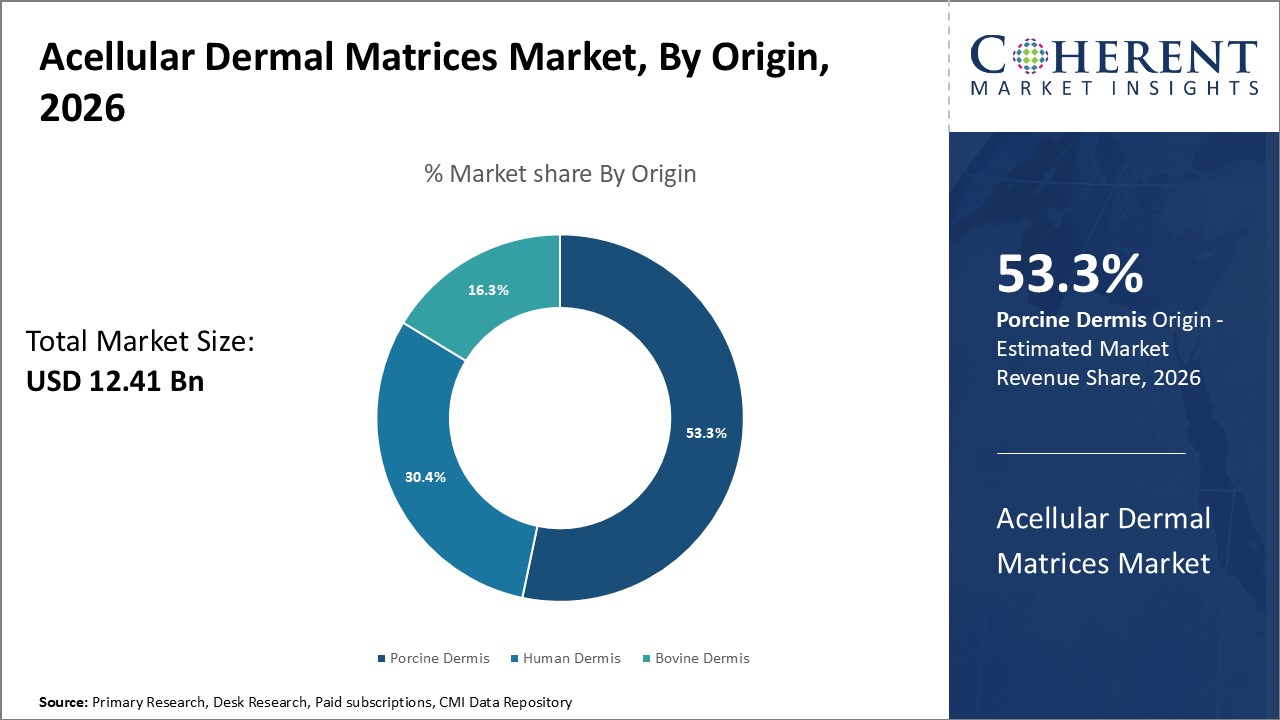
To learn more about this report, Request Free Sample
Global Acellular Dermal Matrices Market – Drivers
Figure 2. Global Acellular Dermal Matrices Market Share (%), By Region, 2026
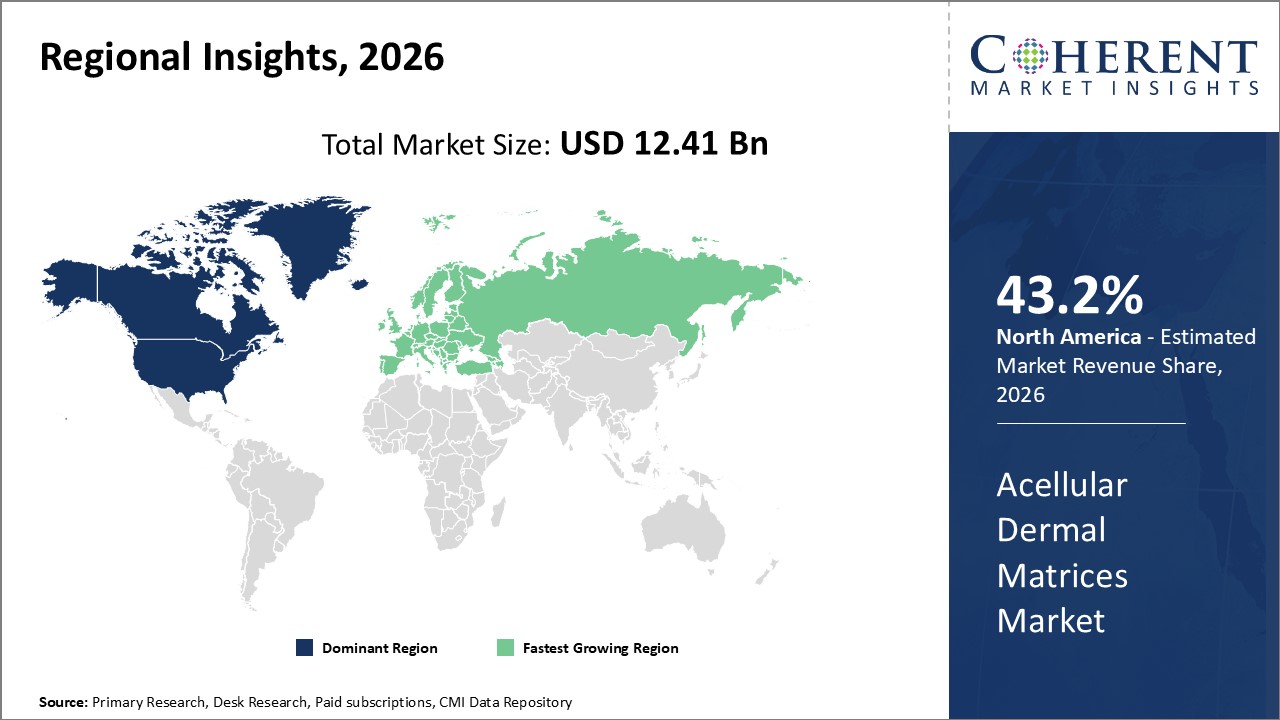
To learn more about this report, Request Free Sample
Global Acellular Dermal Matrices Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global acellular dermal matrices market over the forecast period. North America is estimated to hold 43.2 % of the market share in 2026. The market is expected to witness significant growth in the near future due to increasing product launches in the region. For instance, in April 2022, LifeNet Health, a U.S.-based biotechnology company, announced the launch of its unique allograft treatments for wounds at the 2022 Symposium on Advanced Wound Care (SAWC) Spring Conference, held from April 7-9, 2022, in Phoenix, Arizona, U.S. Its flagship biologic portfolio includes Matrion, the first full-thickness decellularized placental membrane for wounds, and Dermacell AWM, the first acellular dermal matrix clinically shown to effectively cure wounds in as little as one treatment.
Acellular Dermal Matrices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12.41 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.3% | 2033 Value Projection: | USD 27.95 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Integra LifeSciences Corporation, AbbVie Inc., Johnson & Johnson, HansBioMed, Becton, Dickinson and Company, Cook Group, Smith & Nephew Plc., Reprise Biomedical, Organogenesis Holdings Inc., Tissue Regenix, LifeNet Health, Zimmer Biomet Holdings, Inc., Stryker Corporation, MiMedx Group, PolyNovo Limited, Fidia Pharma USA Inc., Baxter International Inc., In2Bones Global, BioHorizons, Inc., Harbor MedTech, Inc., MLM Biologics Inc., Geistlich Pharma AG, Olaregen Therapeutix, Inc., EnColl Corporation, Kerecis limited, DSM, ACell Inc., 3M, Cell Constructs I, LLC, CG bio Inc., and Marine Polymer Technologies, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
This most likely resulted in decrease in demand for acellular dermal matrices for these treatments.
Global Acellular Dermal Matrices Market - Segmentation
Global acellular dermal matrices market is segmented into origin, application, place of setting, and region.
Among all the segmentation, the application segment is expected to dominate the market over the forecast period due to increasing use of ADM in reconstruction surgeries.
Global Acellular Dermal Matrices Market - Cross Sectional Analysis
Among place of setting, hospitals segment is expected to be dominant in the Middle East region, owing to increasing launch of cosmetic surgery hospitals. For instance, in July 2020, NMC Healthcare, one of the largest private healthcare company in the U.A.E., opened its new CosmeSurge Hospital in Jumeirah, U.A.E. The center, completed in December 2019, specializes in cosmetic surgery, and is situated near to the renowned Burj Al Arab. This will be the 17th facility under the CosmeSurge brand.
Global Acellular Dermal Matrices Market: Key Developments
Global Acellular Dermal Matrices Market: Key Trends
Collaborations and Partnerships: Market players are engaged in partnerships or collaborations, and this is expected to drive the global acellular dermal matrices market in the near future. In 2019, BioHorizons, a dental implant manufacturer, in partnership with Camlog, a supplier of premium dental implants, announced the CE certification of NovoMatrix, a novel acellular dermal matrix derived from porcine tissue and intended for soft tissue applications.
Global Acellular Dermal Matrices Market: Restraint
Regulatory challenges: Stringent regulatory approval processes for medical devices including acellular dermal matrices is expected to hinder the global acellular dermal matrices market growth. For instance, in March 2021, The U.S. Food and Drug Administration (FDA) released a safety communication external site pertaining to Acellular Dermal Matrices (ADM) products used in implant-based breast reconstruction. According to the U.S. FDA's review, two ADMs named FlexHD developed by MTF Biologics, a global non-profit organization and Allomax developed by BD, a medical device company may have a greater risk profile than others. Two years after surgery, there were greater incidence of serious problems, explanation, further procedures, and infections.
To counterbalance the restraint, key players should focus on increasing the safety and efficacy of ADM products.
Global Acellular Dermal Matrices Market - Key Players
Major players operating in the global acellular dermal matrices market include Integra LifeSciences Corporation, AbbVie Inc., Johnson & Johnson, HansBioMed, Becton, Dickinson and Company, Cook Group, Smith & Nephew Plc., Reprise Biomedical, Organogenesis Holdings Inc., Tissue Regenix, LifeNet Health, Zimmer Biomet Holdings, Inc., Stryker Corporation, MiMedx Group, PolyNovo Limited, Fidia Pharma USA Inc., Baxter International Inc., In2Bones Global, BioHorizons, Inc., Harbor MedTech, Inc., MLM Biologics Inc., Geistlich Pharma AG, Olaregen Therapeutix, Inc., EnColl Corporation, Kerecis limited, DSM, ACell Inc., 3M, Cell Constructs I, LLC, CG bio Inc., and Marine Polymer Technologies, Inc.
Definition: Acellular dermal matrices (ADMs) are surgical and medical devices that are used principally in the domains of plastic and reconstructive surgery, wound care, and tissue regeneration. These matrices are generated from human or animal sources, and go through a procedure that removes cellular components while keeping the extracellular matrix's (ECM) structure and functional features. Acellular dermal matrices have become indispensable tools in modern medicine by assisting in tissue restoration and reconstruction, particularly when natural tissue repair and regeneration are insufficient. The type of ADM utilized and how it is used are defined by a patient's individual medical condition and need, and are often done in collaboration with surgeons, healthcare practitioners, and tissue engineering specialists.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients